Introduction
The grafting in the plant is an asexual propagation approach that has been widely used to improve crop cultivation. In general, woody plants are grafted to enable dwarfing and/or sturdiness, while herbaceous plants are grafted to increase productivity and/or to control the damages caused by biotic or abiotic stresses (Muneer et al., 2015). Rootstock and scion grafts also improve biotic or abiotic stress tolerance (Kim et al., 2001). Among the horticultural crops, tomatoes, cucumbers, sweet cherries, and melons are the major crops that are commonly grafted (Estañ et al., 2005).
Grafting has been extensively used for understanding the signaling pathways and has led to the discovery of numerous plant processes (Corbesier et al., 2007), the transcriptional changes related to graft interface-specific genes (Cookson et al., 2013), and recently, the rootstock/scion proteins involved in various plant processes such as the transport activity of vascular connections (Muneer et al., 2015). The development of graft union is a complex process during which several histological and physiological alterations such as regeneration of organs and exchange of genetic materials between the rootstocks and scions occurs (Stegemann and Bock, 2009). Numerous imaging techniques and common histological methods have been predominately used for studying the physiological changes that occur during the development of graft unions (Muneer et al., 2015; Soumelidou et al., 1994). Moreover, several studies have characterized the morphological and physiological variations between graft unions (Kollmann and Glockmann, 1985), and a few recent studies have investigated the molecular modifications associated with graft unions (Yin et al., 2012). However, the physiological and biochemical modulations of graft unions in response to relative humidity of the air have not been established well. Therefore, in the present study the effect of different relative humidity regimes on the physiology and antioxidant metabolism in two cultivars of grafted watermelon was investigated.
Materials and Methods
1. Plant materials and relative humidity treatments
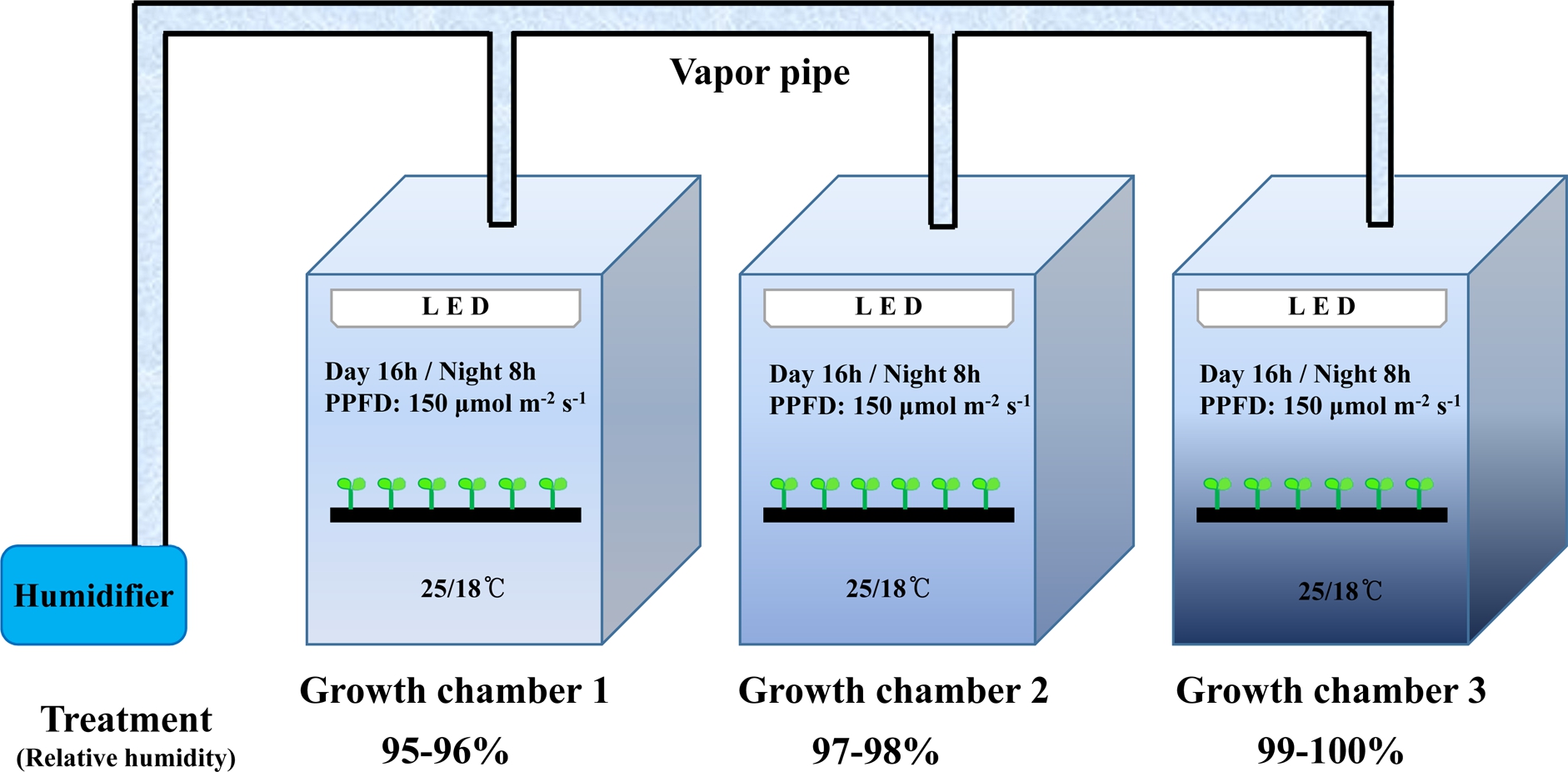
In this experiment, ‘Sambok Honey’ and ‘Speed’ watermelon (Citrullus vulgaris Schrad.) were grafted onto the ‘RS-Dongjanggun’ bottle gourd (Lagenaria siceraria Stanld.). The grafted seedlings were acquired from Green Plug Seedling Nursery (Haman, Korea) and they were grown in square plug trays containing a commercial medium (Super Mix, Nong Kyung Co., Jincheon, Korea). The grafted plants were grown in an environment-controlled chambers (KGC- 175V, Koencon Co. Ltd., Hanam, Korea) and were maintained at a relative humidity (RH) of, 95-96% [1.1-0.8 (day) or 0.8-0.6 (night) g·m-3 (VPD)], 97-98% [ 0.7-0.4 (day) or 0.5-0.3 (night) g·m-3 (VPD)], or 99-100% [0.3-0.0 (day) or 0.2-0.0 (night) g·m-3 (VPD)] according to the Mollier diagram based on air temperature of 25°C day/18°C night with 16h photoperiod per day as shown in Fig. 1. The RH was measured using a precision psychrometer (Almemo-2890- 9, Ahlborn Messund Regelungstechnik GmbH, Holzkirchen, Germany). After six days of subjection to the relative humidity treatment, all grafted plants were transferred to a greenhouse condition and maintained for six days. For experimental measurements, the grafted plants were continuously observed from day 1 of grafting up to day 12. The time points for physiological observations (first day to 12th days) were selected because physiological changes occur within 24 h of grafting.
2. Measurements of physiological parameters, hardness of grafted plants, and vascular staining
For the measurements of physiological parameters, the rootstocks and scions were constantly weighed from the first day to the 12th day after grafting. Hardness of the grafted scion and rootstock was measured using a pressure tester (IT/DFT-01, Proem Co., Forlì, Italy). For vascular staining, the rootstocks and scions were fixed in 0.1% absorbable flower dye red (Absorbable Flower Dye Purper, Centimo Co., RP De Kwakel, The Netherlands) for 20-30 min and rinsed with water according to the method of Olmstead et al. (2006). The stained rootstocks and scions were segmented into transverse and longitudinal sections and observed under a light microscope (Eclipse Ci-S/Ci-L, Nikon, Tokyo, Japan).
3. Analysis of biochemical stress parameters
The content of hydrogen peroxide (H2O2) was estimated by following the protocol of Christou et al. (2014). The antioxidant enzyme extracts were prepared according to the method of Manivannan et al. (2015). The graft junctions (20-30mm long, 0.1g) were used for determination of activities of antioxidant enzymes. The activities of sodium dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX), and guaiacol peroxidase (GPX) were estimated by following the protocols of Shekhawat et al. (2010). The total protein content was estimated according to the Bradford method (Bradford, 1976).
The native staining of the antioxidant enzymes was performed according to the protocol of Shah and Sareeta (2012). All the antioxidant enzymes (30μg protein) were electrophoresed in 10% separating and 4% stacking gel in 4°C for 4h at 80 volts with a tris-glycine (pH 8.3) running buffer. The GPX isomers were detected after the incubation of gels in a 50mM tris-HCl buffer (pH 6.8) with 20mM guaiacol and 20mM H2O2 for 20 min. The SOD isozyme was identified by immersing the gel in a reaction solution containing 0.5mM nitro blue tetrazolium (NBT) and 2mM riboflavin for 15 min in dark followed by light irradiation for 30 min. The APX was localized by incubating the gel in a 50mM phosphate buffer (pH 6.4) containing 4mM ascorbic acid and 4mM H2O2 for 15 min. Subsequently, the gel was immersed in 0.1% ferrocyanide and 0.1% ferricchloride prepared in 1.25 N HCl for 5 min. The CAT enzyme was observed by soaking the gel in a 50mM phosphate buffer (pH 7.0) with 5mM H2O2. After 10 min, the gel was stained with 2% ferricchloride and 2% potassium ferriccyanide.
4. Statistical analysis
The experiment was laid out in a completely randomized design with three replications per treatment containing 40 grafted plants in each plug tray. All the assays were performed in triplicates and the results were averaged. Significant differences among the treatments were determined by analysis of variance (ANOVA) followed by the Duncan multiple range test at a significant level of P=0.05 using a statistical analysis computer package (V. 6.12, SAS Institute Inc., Cary, NC, USA).
Results and Discussion
1. Influence of RH on the physiological indices of the rootstocks and scions
The biomass and hardness of grafted watermelon seedlings grown under different RH treatments are shown in Figs. 2 and 3. Plant height of ‘Speed’ watermelon was significantly increased in the 97-98% RH treatment. Similarly, the 97-98% RH treatment improved the fresh weight of the scions and rootstocks in comparison with other treatments after nine days. Likewise, the hardness of the graft union was enhanced after nine days of grafting under the 97-98 and 99-100% RH treatments, whereas it was less favored in the 95-96% RH treatment. The thickness of both the scions and rootstocks was not significantly influenced by the RH treatments.
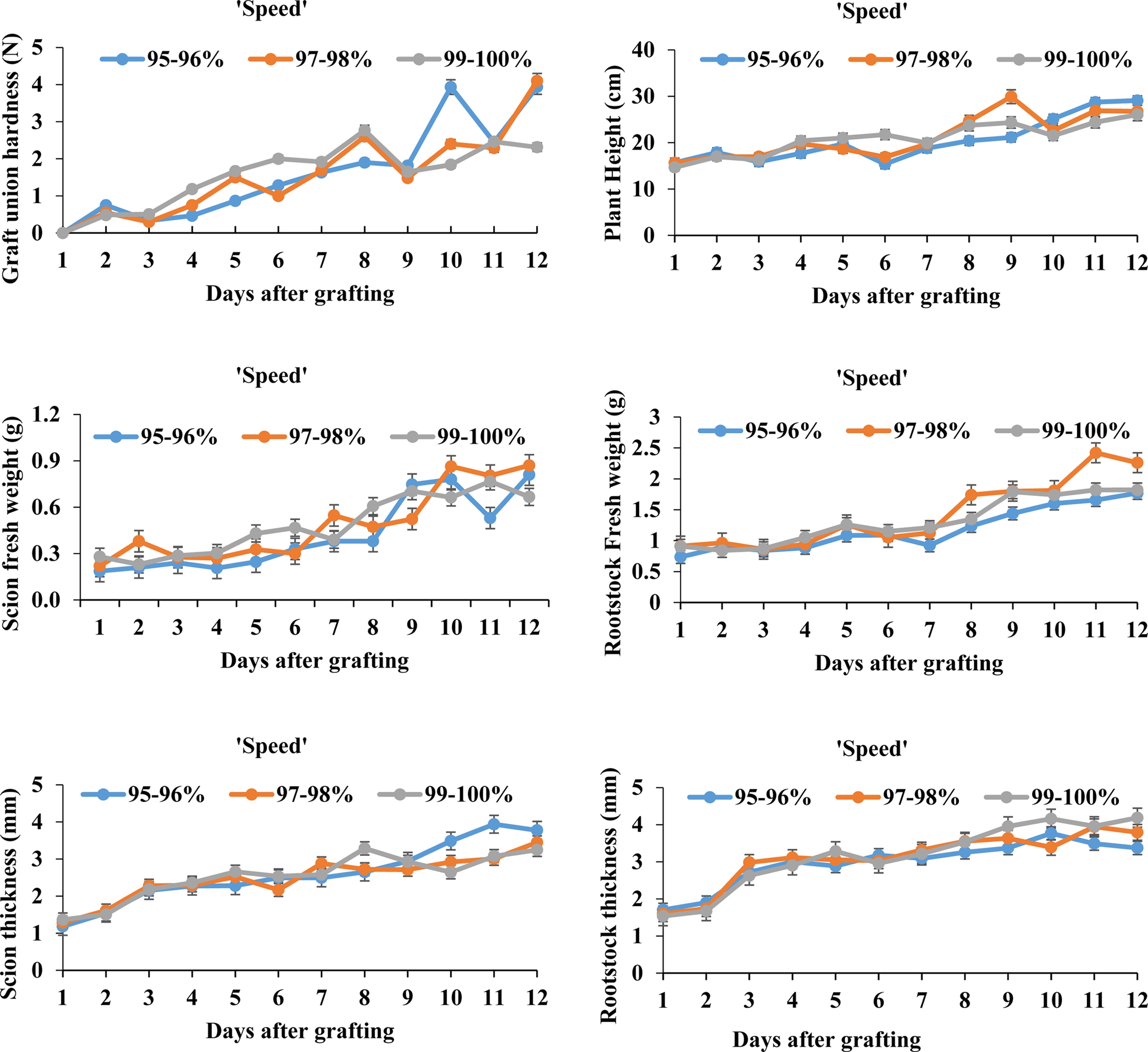
Fig. 2.
Physiological changes of watermelon ‘Speed’ scion and bottle gourd ‘RS-Dongjanggun’ rootstock grown under different relative humidity treatments (95-96, 97-98, and 99-100%). Line diagrams among different treatments represent mean ± SE of five biological replicates (n = 5).
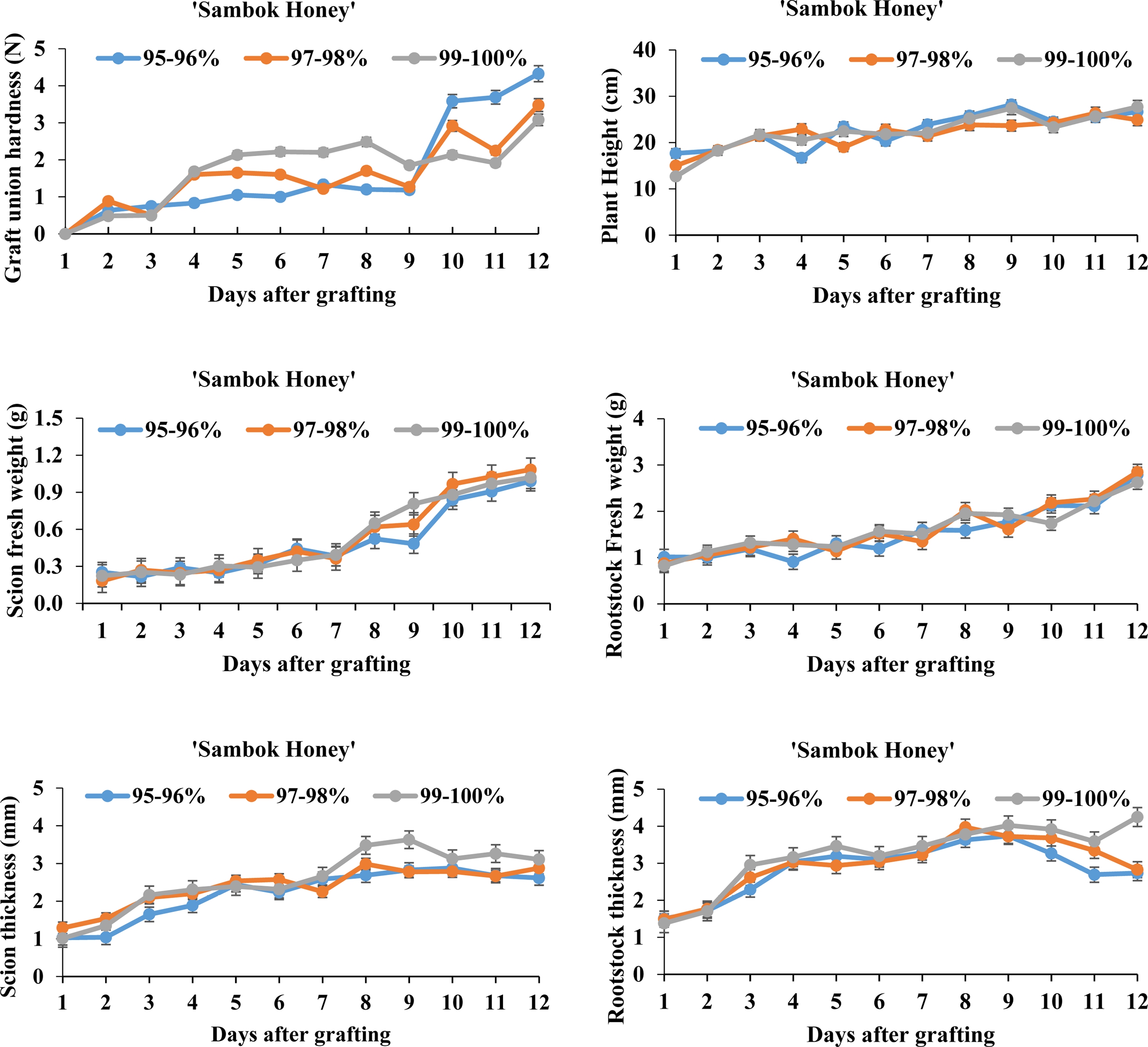
Fig. 3.
Physiological changes of watermelon ‘Sambok Honey’ scion and bottle gourd ‘RS-Dongjanggun’ rootstock grown under different relative humidity treatments (95-96, 97-98, and 99-100%). Line diagrams among different treatments represent mean ± SE of five biological replicates (n = 5).
On contrary to ‘Speed’ watermelon, plant height of ‘Sambok Honey’ watermelon was smallest under the 97- 98% RH and increased in the 99-100% RH treatment after nine days of grafting. Furthermore, fresh weight of the scions significantly increased after seven days in all RH treatments. Among the RH treatments, hardness of the graft unions was enhanced under the 95-96 and 97-98% RH regimes. In a similar manner as with the ‘Speed’ watermelon, thickness of the scions and rootstocks was less affected by the RH treatment. According to Ostendorp et al. (2016), the high relative humidity during the early phase of grafting enhanced the survival of grafted plants. The standard RH regime is necessary for the optimal growth and development of grafted plants, thus physiological results illustrate that the 97-98% RH regime could be appealing for the improvement of grafted watermelon in this study.
2. Vascular transport activity
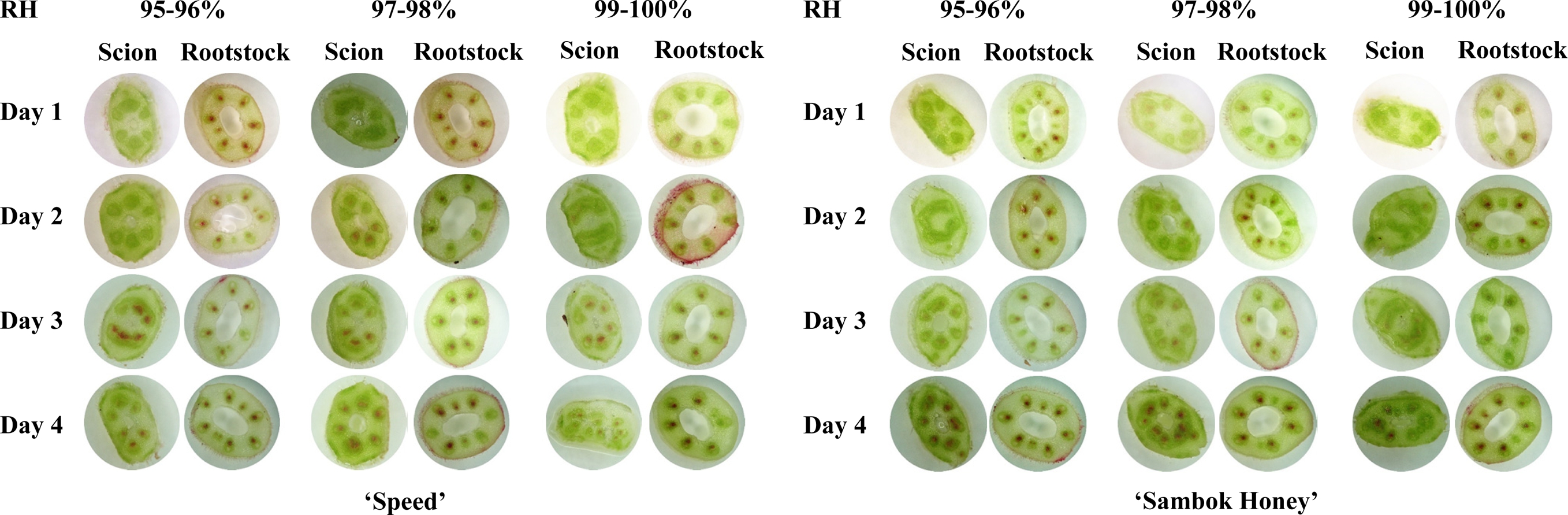
The red coloration in the vascular tissues of all grafted plants was observed in both longitudinal and transverse cross sections (Fig. 4). The higher absorption of the dye in the scion region was observed after two days of grafting under the 97-98% RH treatment in both cultivars. This denoted that the 97-98% RH regime most positively influenced the vascular connection between the scions and rootstocks in both cultivars. The determination of vascular transport activity in the rootstocks and scions by staining was also used previously in grafted sweet cherry (Olmstead et al., 2006) and arabidopsis (Yin et al., 2012). (Fig. 5).
3. Influence of RH treatment on biochemical stress parameters
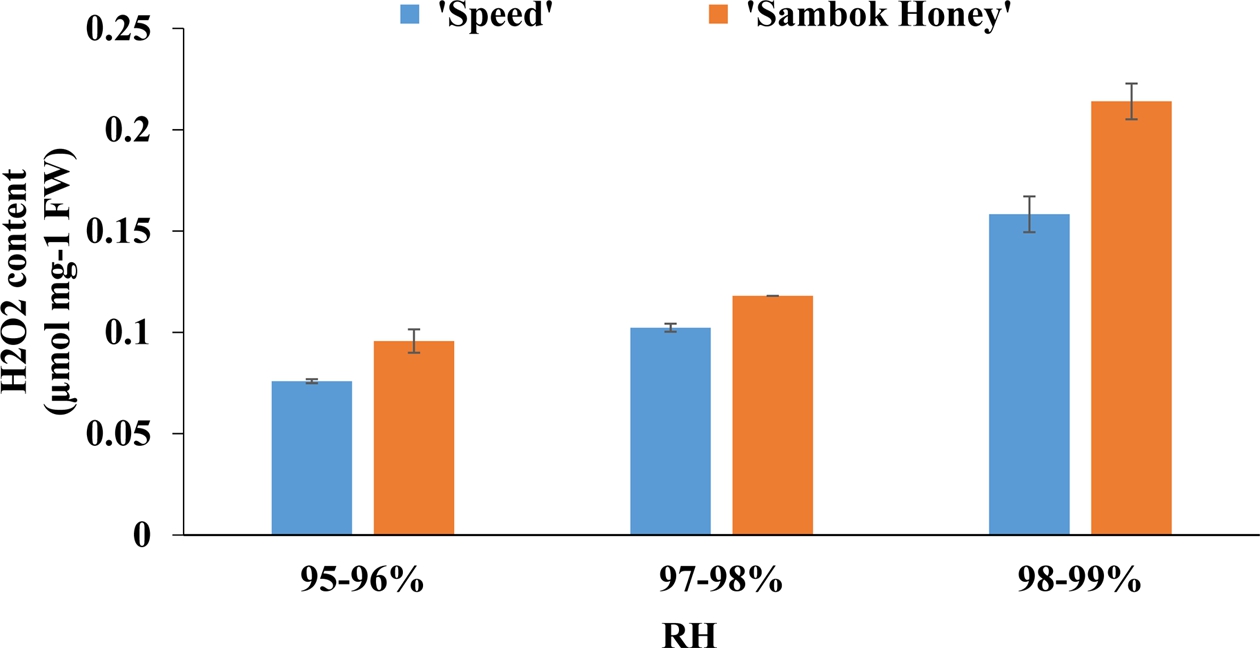
Based on the physiological results, it was highly evident that the grafted plants displayed fluctuations under different RH treatments. According to Muneer et al. (2015), the generation of the harmful reactive oxygen species (ROS) acted as one of the important factors for the occurrence of changes in physiology, such as plant height, hardness of graft union, and biomass, of the grafted tomato seedlings. In the present study, the highest endogenous levels of H2O2 determined in grafted watermelon seedlings was found in the 99-100% treatment in both cultivars. Considering that the H2O2 is a primary signaling molecule, generated highly under an oxidative stress condition, the high RH treatment used in this study induced oxidative stress in the grafted watermelon seedlings. (Fig. 6).
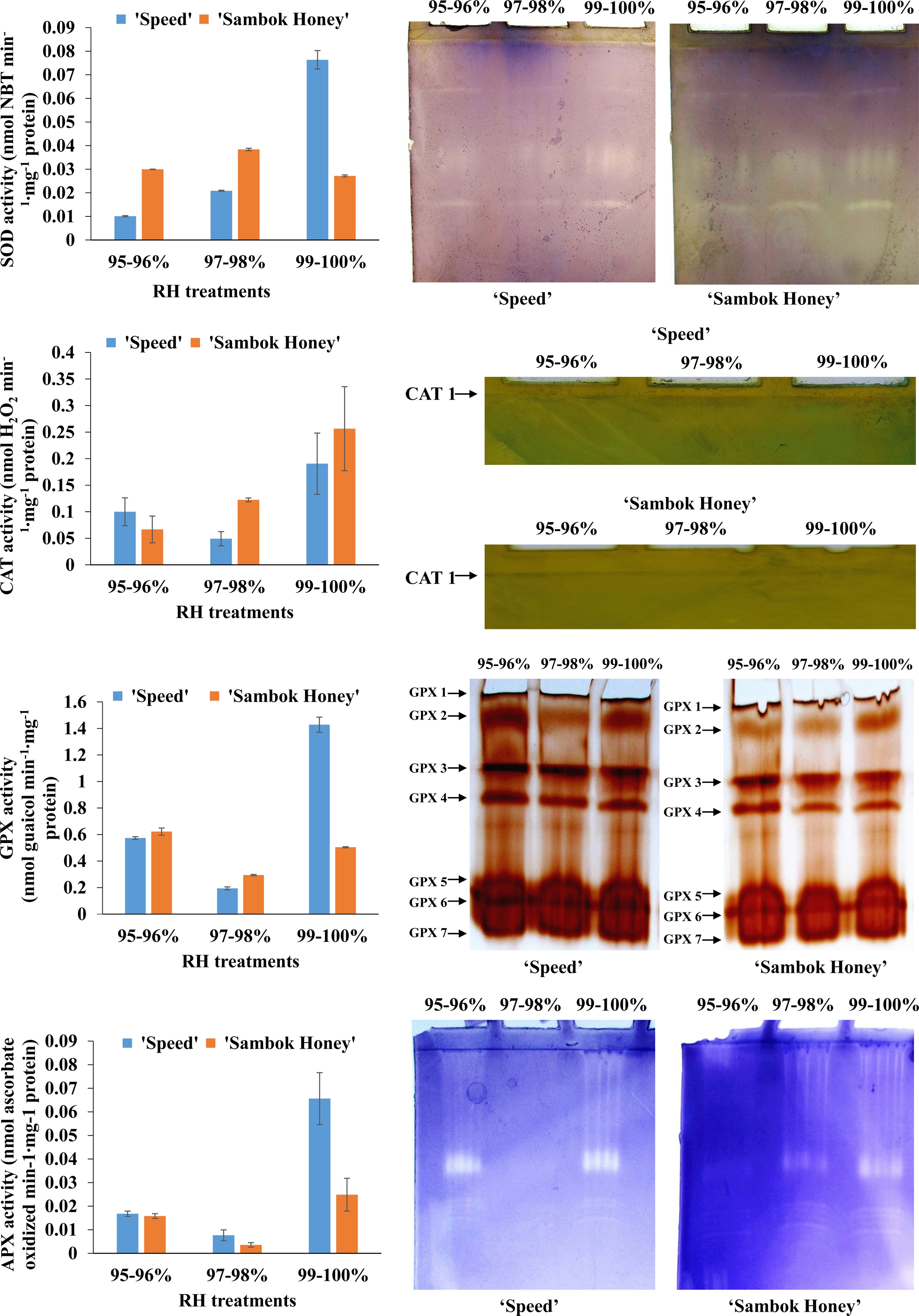
Fig. 6.
Effect of different relative humidity regimes on the activities and expressions of antioxidant enzymes of grafted seedlings. Data are represented as means of three replicates.
In addition, activities and isomeric expressions of antioxidant enzymes, such as SOD, CAT, GPX and APX, were significantly increased in the 99-100% RH treatment. In general, SOD is the primary enzyme involved in the first line of defense to regulate high levels of ROS generation in plants. This enzyme catalyzes the conversion of superoxide radical into hydrogen peroxide and molecular oxygen (Giannopolitis and Ries, 1977). Further cascade of antioxidant enzymes, such as APX, GPX and CAT, scavenges and detoxifies the H2O2 molecule (Gill and Tuteja, 2010). Taken together, the grafted seedlings combated the oxidative burst occurred in the 99- 100% RH regime by the internal regulation of antioxidant metabolism. Moreover, the maintenance of minor levels of ROS is necessary for the proper functioning of signal transduction pathways involved in the cellular differentiation and tissue development (Foreman et al., 2003; Gechev and Hille, 2005). Thus, the occurrence of lower levels of H2O2 and activities of antioxidant enzymes in the 95-96 and 97-98% RH treatments could improve the growth of the grafted seedlings. Overall, the healing process in the grafted watermelon seedlings can be significantly enhanced by the optimization of RH which influenced the physiology and biochemical antioxidant metabolism.
Conclusions
Overall, the RH regimes significantly influenced the graft healing process by regulating the physiological and biochemical mechanisms in grafted watermelon seedling of both cultivars. Among the RH treatments, 97-98% RH treatment enhanced the vascular connection and biomass of the grafted plants. Thus, the outcomes of the present endeavor can help in the selection of optimal humidity condition for the successful healing process in graft watermelon seedlings.