Introduction
Materials and Methods
1. Experimental materials and growing conditions
2. Drought treatment and drought response assessment of kimchi cabbage seedlings
3. Growth measurement and photosynthetic parameters
4. CF Imaging Parameters
5. Statistical Analysis
Results
1. Morphological characteristics during water deficit treatment and recovery period
2. Photosynthetic and recovery responses to drought stress
3. Changes in CF parameters and their correlation with physiological and morphological characteristics during drought stress
Discussion
1. Growth and development responses to drought stress and recovery
2. Cultivar variation in recovery dynamics after re-irrigation
3. Photosynthetic responses and recovery
4. CF as a stress and recovery indicator
5. Implications on breeding and crop management
Conclusion
Introduction
Kimchi cabbage (Brassica rapa ssp. pekinensis) is one of the most widely cultivated leafy vegetables in South Korea, primarily used for kimchi production. It plays a crucial role in Korean agriculture but remains highly susceptible to abiotic stresses such as drought and heat, particularly during the heading stage after transplantation (Jang et al., 2024; Patra et al., 2016). The increasing frequency of extreme weather events associated with global climate change has further elevated drought risk, especially in spring cultivation systems (Jang et al., 2024; Lee et al., 2025).
Drought stress negatively affects both yield and quality, making the breeding of drought-tolerant cultivars a key priority for sustainable production. However, conventional evaluation methods rely largely on destructive measurements and subjective visual assessments, which limit their reproducibility and scalability. Most previous studies on drought stress in plants have focused mainly on physiological, morphological, biochemical, photosynthetic, and molecular responses (Chen et al., 2022; Chen et al., 2024; Eom et al., 2018; Jeong et al., 2013; Kaur et al., 2021; La et al., 2019; Li et al., 2023; Shawon et al., 2018). Although these approaches have advanced our understanding of drought-induced changes, they often overlook the equally important recovery phase following re-irrigation.
The ability of a plant to recover, also referred to as drought resilience, is critical for survival and yield stability under fluctuating water availability. Recovery depends on the restoration of photosynthetic activity, water status, and metabolic functions, serving as an important indicator for breeding drought-resilient cultivars (Antunović Dunić et al., 2023; Chen et al., 2022; Illouz-Eliaz et al., 2024; Jang et al., 2024; Levitt, 1986; Weisz et al., 1989; Wu et al., 2012). Despite its importance, comprehensive evaluations of drought recovery in kimchi cabbage, particularly during the seedling stage, remain limited.
Non-destructive and quantitative screening tools are essential for assessing drought responses and resilience in seedlings. Traditional visual evaluation of leaf wilting provides a common approach to distinguish tolerant genotypes from sensitive ones (Jang et al., 2024; Kilpeläinen et al., 2020). However, it is prone to subjectivity, human error, delayed detection, and inconsistency. To overcome these limitations, recent advances in high-throughput phenotyping (HTP) technologies, RGB imaging, hyperspectral reflectance, and chlorophyll fluorescence (CF) imaging have enabled objective and repeatable assessment of plant stress responses (Briglia et al., 2020; Orka et al., 2023; Pérez-Patricio et al., 2024; Shin et al., 2021; Ye et al., 2023).
Among these tools, CF imaging provides a measurement of photosystem II (PSII) efficiency and photosynthetic performance under stressful conditions. Numerous studies have demonstrated that the maximum quantum yield of photosystem II (Fv/Fm) is a sensitive indicator of stress-induced photoinhibition (Arief et al., 2023; Bartold and Kluczek, 2024; Jumrani and Bhatia, 2019; Lin et al., 2022; Shin et al., 2019; Shin et al., 2020). Drought reduces photosynthetic activity through cellular dehydration, stomatal closure, and osmotic imbalance, all of which can be detected through CF-derived parameters (Guo et al., 2022; Jumrani and Bhatia, 2019; Shin et al., 2019; Shin et al., 2021; Zait et al., 2024). However, the application of CF imaging parameters for evaluating post-drought recovery in kimchi cabbage seedlings has not yet been comprehensively studied.
Therefore, we evaluated the applicability of CF imaging parameters as indicators of drought stress and subsequent recovery in kimchi cabbage seedlings. By integrating CF-derived indices with morphological and physiological traits, we developed reliable and non-destructive indices for assessing drought resilience, thereby supporting the selection and breeding of drought-tolerant genotypes.
Materials and Methods
1. Experimental materials and growing conditions
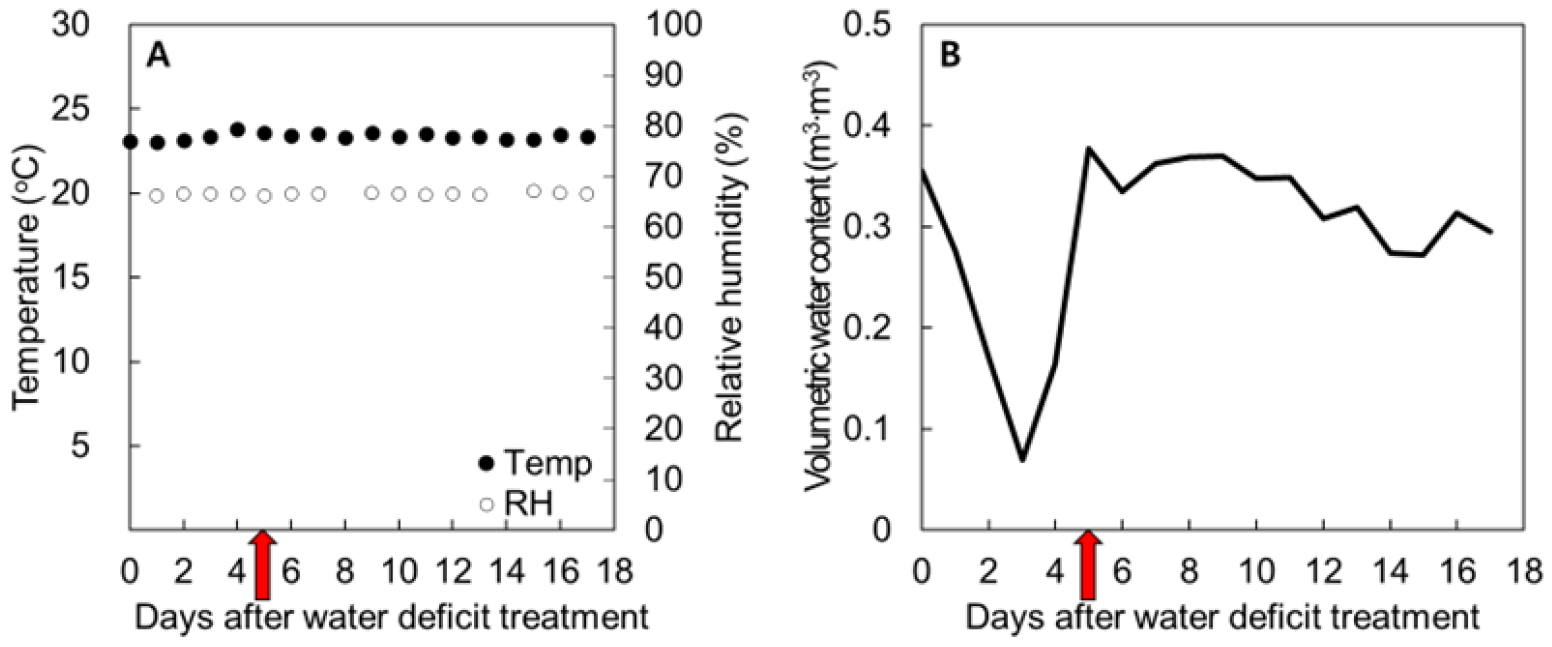
An experiment was carried out in a walk-in plant growth chamber (Gaooze, Suwon, Republic of Korea) at the plant phenotyping research center of the National Institute of Horticultural and Herbal Science, Wanju, republic of Korea. Environmental conditions were maintained at 23.4 ± 0.2℃ and 66.5 ± 0.2% relative humidity (RH) under a 14 h light/10 h dark photoperiod using a light emitting diode (LED) system that provided 410 ± 38.5 µmol·m-2·s-1 photosynthetic photon flux density (PPFD) at the shoot apex (Fig. 1A).
The volumetric water content (VWC, m3·m-3) of the potting soil was monitored every 10 min using a soil moisture sensor (MX2307, Onset Computer Corp., ME, USA; Fig. 1B). The sensor measures the dielectric permittivity of the substrate at 70 MHz and converts it internally to VWC based on the factory calibration for potting soil.
Two kimchi cabbage cultivars, ‘Bulam3ho’ (Farm Hannong, Republic of Korea) and ‘Hwipalam’ (Sakata Korea, Republic of Korea), were evaluated at the seedling stage. The experiment followed a randomized complete block design, with each cultivation bench serving as a block. All cultivars were grown on every bench, with their positions within each bench randomized. Seeds (18 per tray) were sown in two 72-cell trays (280 × 540 × 48 mm; 6 × 12 cells, Bumnong Co., Republic of Korea) filled with potting soil (Bio Sangto No. 1, Heungnong Seeds, Pyeongtaek, Republic of Korea). After germination, seedlings were irrigated regularly using a liquid fertilizer (Mulpure, Daeyu Co., Seoul, Republic of Korea).
2. Drought treatment and drought response assessment of kimchi cabbage seedlings
At 17 days after sowing (DAS; equivalent to 0 days after water-deficit treatment, 0 DAW), when seedlings had developed four to five true leaves, drought stress was imposed by withholding irrigation for 5 days. As shown in Fig. 1B, the substrate VWC decreased from 0.35 to 0.07 m3·m-3 during the drought period and recovered to 0.38 m3·m-3 after re-watering, remaining stable thereafter. Irrigation during the experimental period was adjusted according to substrate moisture status and seedling growth.
Drought and recovery responses were visually evaluated using three indices as wilting score, survival of leaves, and survival of plants (Jang et al., 2024; Kilpeläinen et al., 2020; Lee et al., 2024). The wilting score was classified into six categories: 0 (completely dried or dead), 1 (>80% of leaves wilted), 2 (60-80%), 3 (40-60%), 4 (20-40%), and 5 (no visible symptoms). The average wilting score was calculated across all seedlings within each treatment. Survival of leaves was defined as the ratio of living leaves to the total number of leaves per plant, and survival of plants as the proportion of surviving plants among the 36 seedlings per cultivar. Although visual assessments are limited in reproducibility, they were included for correlation analysis with drought-related indicators such as CF parameters.
During the recovery phase, the leaf water content (LWC) was calculated following the equation described in (Guo et al., 2022; Wang et al., 2021):
3. Growth measurement and photosynthetic parameters
Both non-destructive and destructive measurements were conducted on 12 plants at 0 DAW and at 0, 2, 4, 8, and 12 days after re-irrigation (DAR; corresponding to 5, 7, 9, 13, and 17 DAW, respectively). The measured morphological characteristics included the number of leaves, leaf area, fresh weight, and dry weight per plant. Leaves longer than 1 cm were counted manually, and the leaf area was measured using a leaf area meter (Li-3100C, Li-Cor, NE, USA). Fresh weight was measured immediately after sampling using a precise balance (FX-2000i, A&D, Japan), and dry weight was determined after oven-drying the plants at 70℃ for more than 72 h (DS-80-3, DASOL SCIENTIFIC, Hwaseong, Republic of Korea). Measurements were performed on 12 plants per cultivar, excluding the border rows on each side of the bench.
Growth and development dynamics were analyzed relative growth rate (RGR), relative leaf expansion rate (RLER), and relative leaf appearance rate (RLAR), according to Hunt (1978).
Where X1 and X2 are the measured trait values at times t1 and t2 (DAR). RGR was calculated based on plant fresh weight (RGRtfw, g·g-1·d-1) and dry weight (RGRtdw, g·g-1· d-1). The relative leaf expansion rate (RLER, cm2·cm-2·d-1) was calculated from leaf area, and the relative leaf expansion rate (RLAR, leaves·leaves-1·d-1) was calculated from number of leaves.
Photosynthetic parameters were measured in six seedlings per treatment on the same days as the morphological trait measurements (0 DAW and 0, 2, 4, 8, and 12 DAR; corresponding 5, 7, 9, 13 and 17) using a portable photosynthesis system (Li-6800, Li-Cor Co., Inc., USA). The LED light source was set to a red:blue ratio of 75:25. The leaf chamber conditions were set at 23℃, 60% RH, 400 µmol·m-2·s-1 PPFD, and 400 ppm CO2 concentration. Measurements were taken from the third to fourth fully expanded leaves, counted from the youngest unfolded leaf, after the leaf had acclimated to the leaf chamber. The net photosynthetic rate (Pn), stomatal conductance (Gs), intercellular CO2 concentration (Ci), and transpiration rate (Tr) were recorded.
4. CF Imaging Parameters
CF imaging was conducted from a top view of the 12-18 seedlings using a CF imaging system (Open FluorCam 800-O/3535, Photon System Instruments, Czech Republic). Before measurement, seedlings were dark-adapted for 20 min. The distance between the camera and the plants was maintained at more than 30 cm. Among various CF parameters, the maximum quantum yield of photosystem II (Fv/Fm), a widely used indicators of overall photosynthetic performance or damage in plants, was analyzed. Measurements were carried out on the same days as the growth and photosynthetic investigation. System settings, including the flash light level, super light level, detector sensitivity, and shutter speed, were calibrated to ensure accurate and consistent fluorescence detection across the upper surface of true leaves. These measurement settings were kept constant throughout the experiment. The CF imaging parameters used in this study are presented in Table 1.
Table 1.
Chlorophyll fluorescence imaging parameters used in this study.
5. Statistical Analysis
All statistical analyses were performed using SPSS Statistics 29 software (IBM, IL, USA). Differences between 0 and 5 DAW within each cultivar were evaluated using independent t-tests. Comparisons across recovery stages were conducted using one-way analysis of variance followed by Duncan’s multiple test at the 95% confidence level. Pearson’s correlation analysis was used to examine relationships among growth and development characteristics, photosynthetic parameters, and CF-derived indices under drought stress and during recovery.
Results
1. Morphological characteristics during water deficit treatment and recovery period
The morphological characteristics of kimchi cabbage seedlings (‘Bulam3ho’ and ‘Hwipalam’) to water deficit and subsequent recovery shown in Fig. 2. Both cultivars exhibited similar overall responses under drought stress. In ‘Bulam3ho’, the number of leaves increased slightly (p ≤ 0.01), whereas the plant leaf area decreased markedly by 30.5% (p ≤ 0.01) during the drought period (Figs. 2A and 2B). The plant fresh weight and LWC decreased significantly (p ≤ 0.01, Figs. 2C and 2F). In contrast, plant dry weight did not change significantly (ns, Fig. 2D), while dry matter content (DMC) increased from 0.12 to 0.31 (p ≤ 0.01), indicating relative water loss (Fig. 2E).
Similar patterns were observed in ‘Hwipalam.’ The number of leaves increased slightly (4.42 to 5.00 leaves, p ≤ 0.05, Fig. 2A), whereas the plant leaf area decreased by 39.4% (p ≤ 0.01, Fig. 2B). The plant fresh weight decreased sharply by 46.6% (p ≤ 0.001, Fig. 2C), and LWC declined from 0.89 to 0.57 (p ≤ 0.001, Fig. 2F). In contrast to ‘Bulam3ho’, plant dry weight increased from 0.20 g to 0.28 g (p ≤ 0.05, Fig. 2D), accompanied by a 3.9 fold increase in DMC (p ≤ 0.001, Fig. 2E). Overall, both cultivars showed reduced plant leaf area, plant fresh weight, and LWC, along with increased DMC, indicating that drought primarily induced leaf dehydration and biomass reduction. However, ‘Hwipalam’ exhibited a greater reduction in plant fresh weight (Fig. 2C).
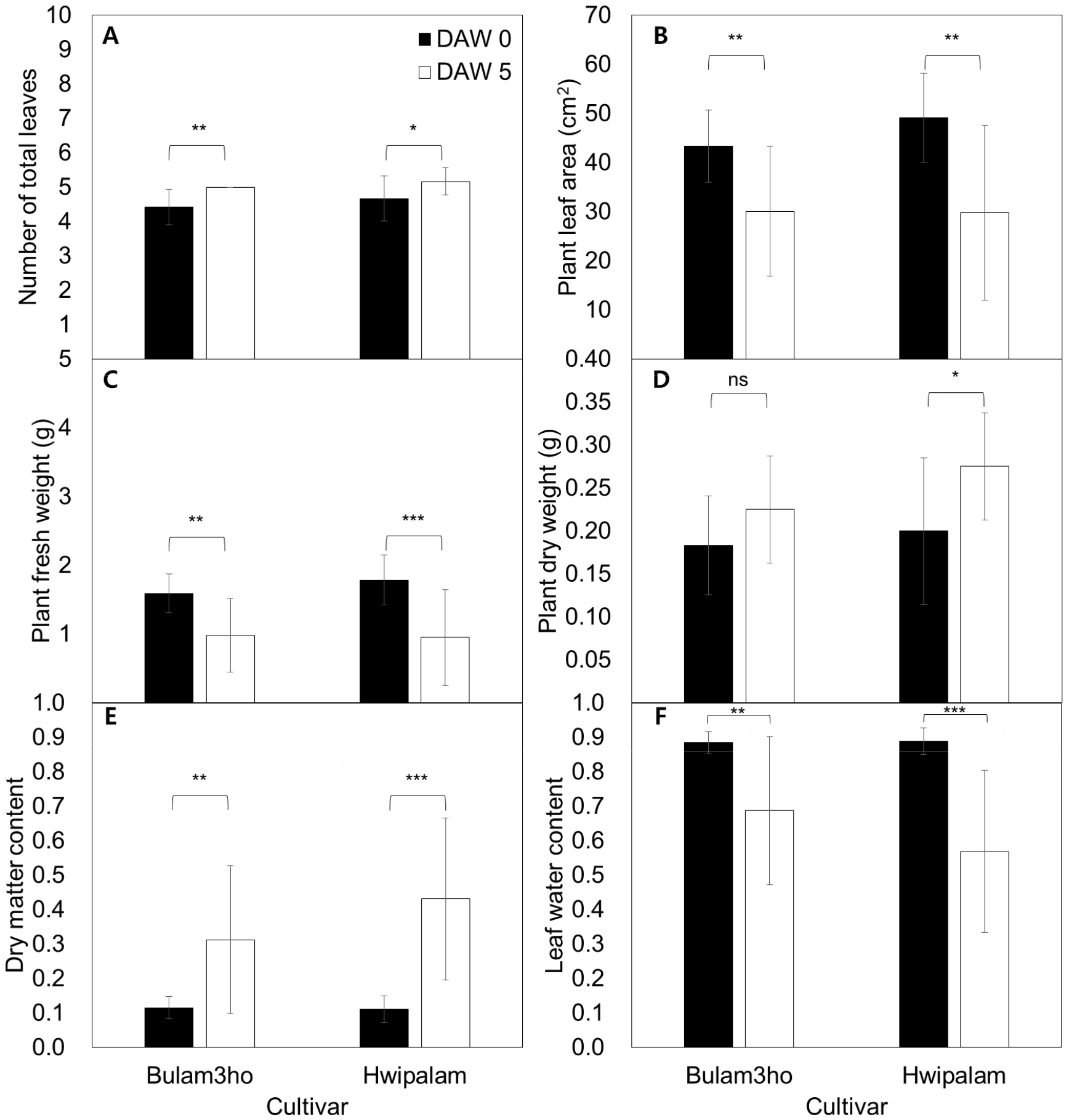
Fig. 2
Comparison of number of leaves (A), plant leaf area (B), plant fresh weight (C), plant dry weight (D), dry matter content (E), and leaf water content (F) in kimchi cabbage ‘Bulam3ho’ and ‘Hwipalam’ measured at 0 and 5 days after water deficit treatment (DAW) respectively. Bars represent mean values ± standard deviation (n = 12). Statistical significance was determined using independent t-tests; different asterisks indicate significant differences between cultivars at each time point (ns; non-significant, *; p ≤ 0.05, **; p ≤ 0.01, ***; p ≤ 0.001).
Following re-irrigation, both cultivars showed gradual recovery (Fig. 3). The number of leaves, plant leaf area, plant fresh weight, and plant dry weight increased progressively. The RLAR increased immediately after re-watering, remaining consistently higher in ‘Bulam3ho’. During the early recovery phase (0-1 DAR), the RLAR of ‘Bulam3ho’ was 2.4 fold higher, indicating a more rapid initiation of leaf emergence (Figs. 3A and 3B). Similarly, RLER increased in both cultivars, but ‘Bulam3ho’ showed earlier and wider rate, whereas ‘Hwipalam’ exhibited delayed expansion (Figs. 3C and 3D). RGRtfw peaked in ‘Bulam3ho’ during 0-1 DAR (0.214 d-1), reflecting rapid rehydration and turgor recovery. In contrast, the ‘Hwipalam’ showed a gradual increase (Figs. 3E and 3F). RGRtdw was lower than RGRtfw but increased steadily, with ‘Bulam3ho’ showing more efficient dry matter recovery (Figs. 3G and 3H). DMC decreased initially during early recovery (2-4 DAR; Fig. 3I), whereas LWC increased at 4 DAR before declining slightly toward the end of the recovery period (Fig. 3J). Overall, these results indicate that ‘Bulam3ho’ recovered leaf expansion and biomass accumulation more rapidly than ‘Hwipalam’, revealing distinct genotypic differences in post-drought recovery strategies.
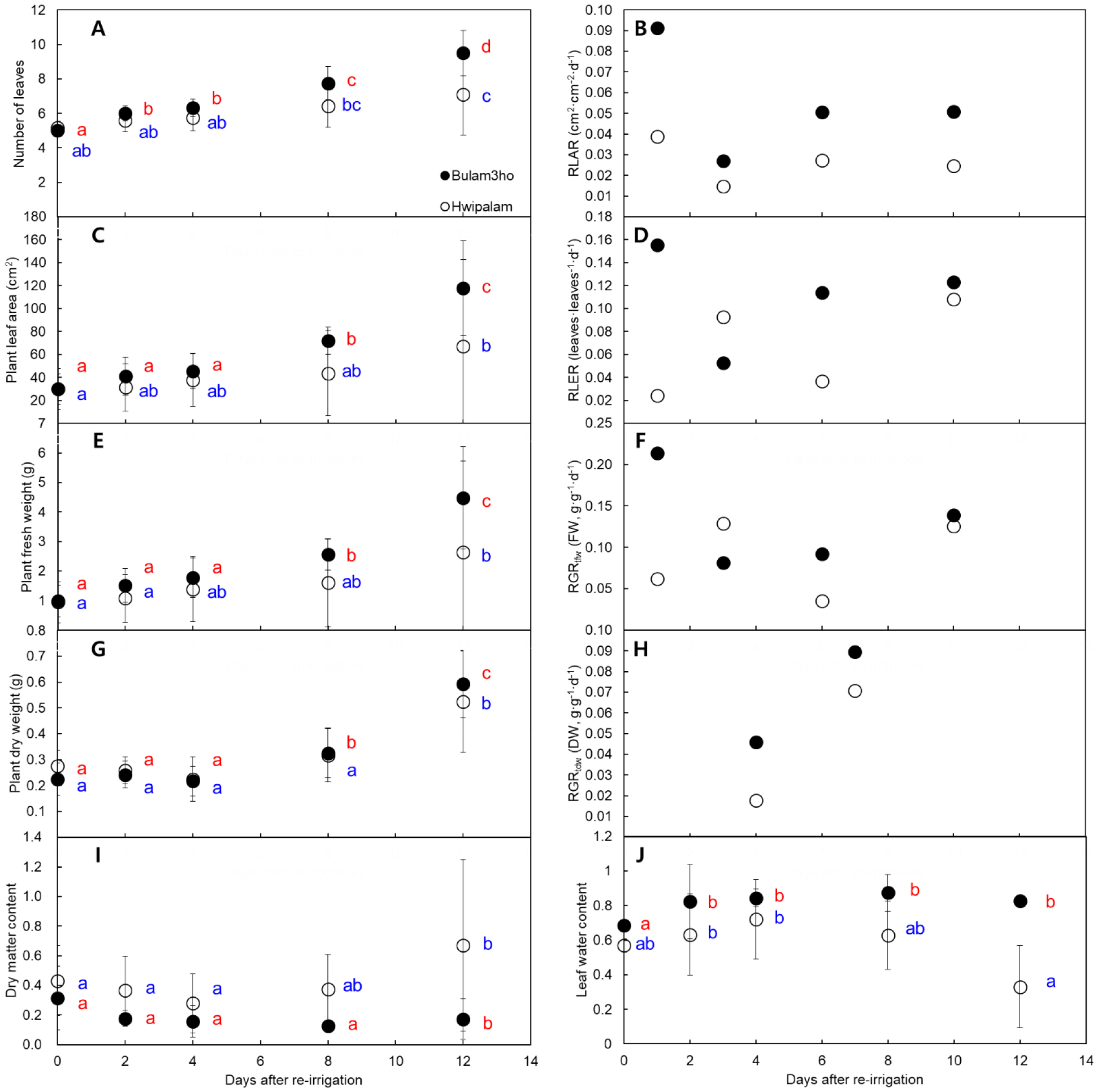
Fig. 3
Changes in the number of leaves (A), relative leaf appearance rate (B), plant leaf area (C), relative leaf expansion rate (D), plant fresh weight (E), relative growth rate based on plant fresh weight (F), plant dry weight (G), and relative growth rate based on plant dry weight (H) of Kimchi cabbage cultivars ‘Bulam3ho’ (●) and ‘Hwipalam’ (○) during recovery period following water deficit treatment. For each cultivar, Duncan’s multiple range test results for measurements are indicated in blue for ‘Bulam3ho’ and red for ‘Hwipalam.’ Means followed by different letters are significantly different at p ≤ 0.05 according to Duncan’s multiple range test. Data in (A, B, C, D, E, F, G, and H) represent the mean ± standard deviation (n = 12).
2. Photosynthetic and recovery responses to drought stress
Drought stress induced reductions in photosynthetic parameters in both cultivars after 5 DAW (Fig. 4). Stomatal closure limited transpiration and CO2 uptake, thereby restricting photosynthesis. In ‘Bulam3ho’, Pn decreased from 3.80 to -0.73 µmol CO2·m-2·s-1 (p ≤ 0.01, Fig. 4A). Tr and Gs declined by 85.6% and 86.9%, respectively (p ≤ 0.001, p ≤ 0.01, Figs. 4B and 4C). In contrast Ci increased 1.3 fold (Fig. 4D). Similar patterns were observed in ‘Hwipalam’, where Pn, Tr, and Gs decreased significantly (p ≤ 0.01), whereas Ci increased from 342.9 to 484.0 µmol·mol-1 (p ≤ 0.001).
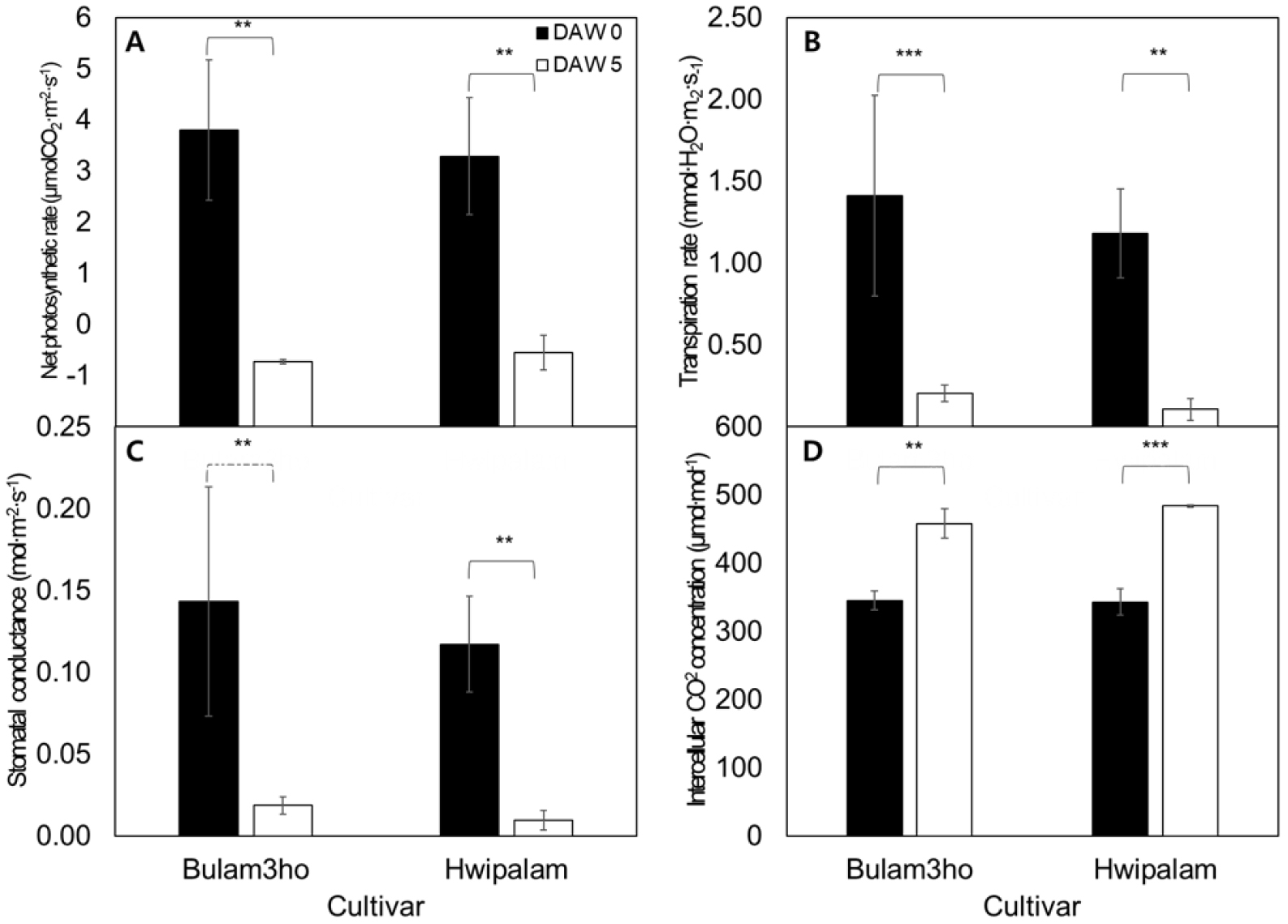
Fig. 4
Comparison of net photosynthetic rate (Pn; A), transpiration rate (Tr; B), stomatal conductance (Gs; C), and Intercellular CO2 concentration (Ci; D) in kimchi cabbage ‘Bulam3ho’ and ‘Hwipalam’ measured at 0 and 5 days after drought stress treatment (DAW), respectively. Bars represent mean values ± standard deviation (n = 6). Statistical significance was determined using independent t-tests; different asterisks indicate significant differences between cultivars at each time point (ns; non-significant, *; p ≤ 0.05, **; p ≤ 0.01, ***; p<0.001).
During the recovery phase (Fig. 5), photosynthetic activity improved markedly in both cultivars. Immediately after re-irrigation (0 DAR), Pn remained negative, indicating severe inhibition of photosynthesis. However, Pn increased rapidly, reaching 9.37-9.47 µmol CO2·m-2·s-1 by 8-12 DAR (Fig. 5A). Both Tr and Gs increased steadily throughout recovery (Figs. 5B and 5C), increasing 23.2 fold and 35.7 fold in ‘Bulam3ho’ and ‘Hwipalam’, respectively. Conversely, Ci, which was increased at 0 DAR (358.9-458.3 µmol·mol-1), declined substantially during early recovery, reaching minimum values of 330.6 µmol·mol-1 in ‘Bulam3ho’ and 336.8 µmol·mol-1 in ‘Hwipalam’ at 8 DAR (Fig. 5D). Overall, re-irrigation restored photosynthetic capacity within 8-12 days. The increases in Pn, Tr, and Gs and the decrease in Ci closely linked to stomatal reopening and improved CO2 assimilation.
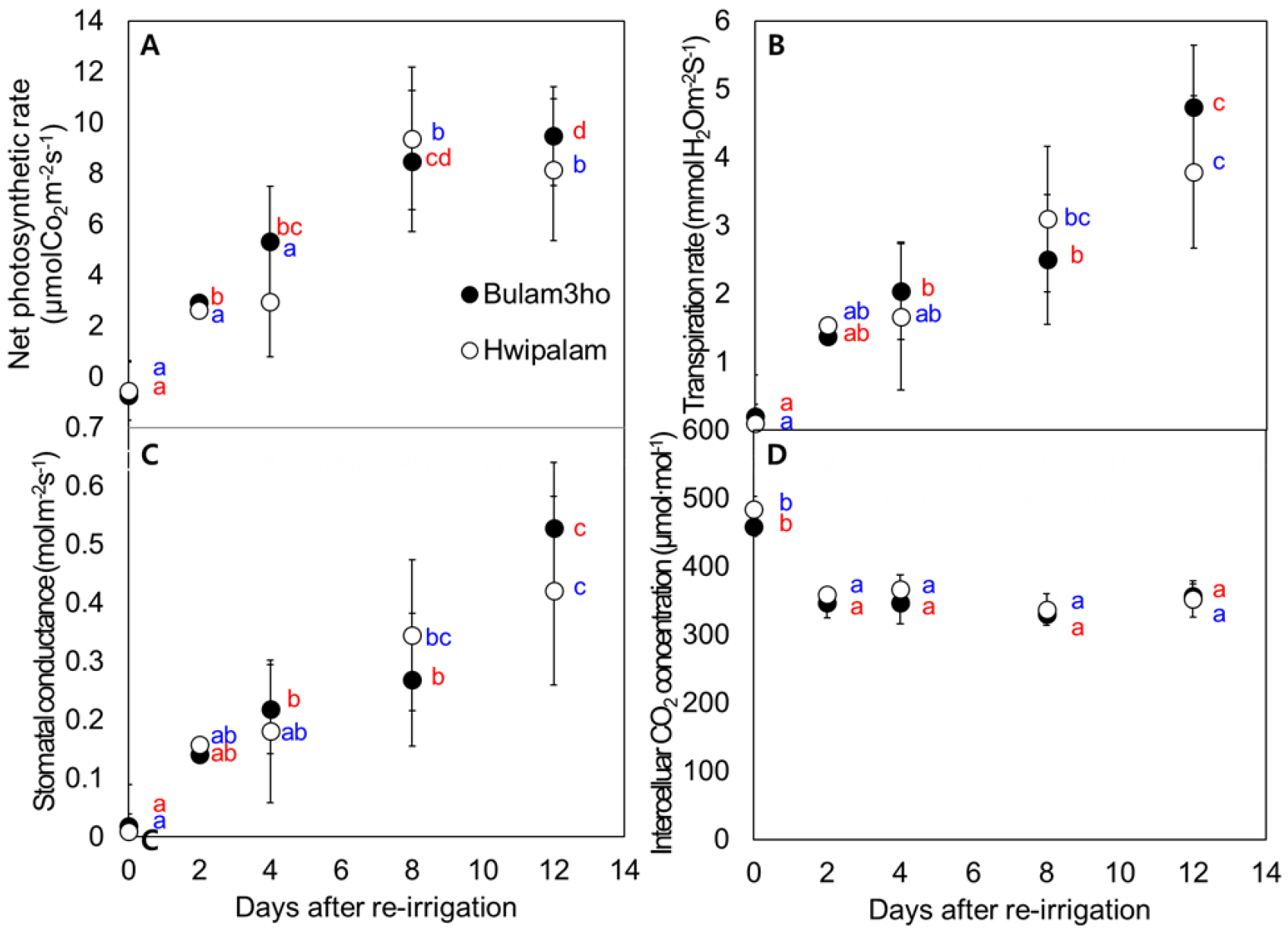
Fig. 5
Changes in the net photosynthetic rate (Pn; A), transpiration rate (Tr; B), stomatal conductance (Gs; C), and intercellular CO2 concentration (Ci; D) of kimchi cabbage cultivars ‘Bulam3ho’ (●) and ‘Hwipalam’ (○) during recovery period following water deficit treatment. For each cultivar, Duncan’s multiple range test results for measurements are indicated in blue for ‘Bulam3ho’ and red for ‘Hwipalam’. Means followed by different letters are significantly different at p ≤ 0.05 according to Duncan’s multiple range test. Data in (A, B, C, and D) represent the mean ± standard deviation (n = 6).
3. Changes in CF parameters and their correlation with physiological and morphological characteristics during drought stress
The projected fluorescence area was significantly lower at 5 DAW than at 0 DAW in both cultivars (Figs. 6 and 7A). During the recovery period, F0 showed smaller variation than the other parameters (Fig. 7B). Fm and Fv patterns similar to that of Fv/Fm, reaching their lowest values on 5 DAW. Thereafter, significant recovery was observed in ‘Bulam3ho’ at 4 DAR and in ‘Hwipalam’ at 8 DAR (Figs. 7C and 7D). Fv/Fm decreased by 40.8% in ‘Bulam3ho’ and 65.0% in ‘Hwipalam’ between 0 and 5 DAW, followed by a gradual increase during re-irrigation. In ‘Bulam3ho’, Fv/Fm recovered significantly by 4 DAR, whereas in ‘Hwipalam’ recovery was slower, and values stabilized after 12 DAR (Fig. 7E). The Fv/F0, representing the maximum potential PSII efficiency, was highly susceptible to drought, decreasing at 5 DAW by 14.2% and 22.9% from 0 DAW values in ‘Bulam3ho’ and ‘Hwipalam’, respectively (Fig. 7F). Both indices gradually recovered, with ‘Hwipalam’ maintaining higher values at later stages. These results indicate that drought induced substantial but reversible photoinhibition of PSII, with ‘Hwipalam’ showing relatively greater photochemical stability despite slower recovery.
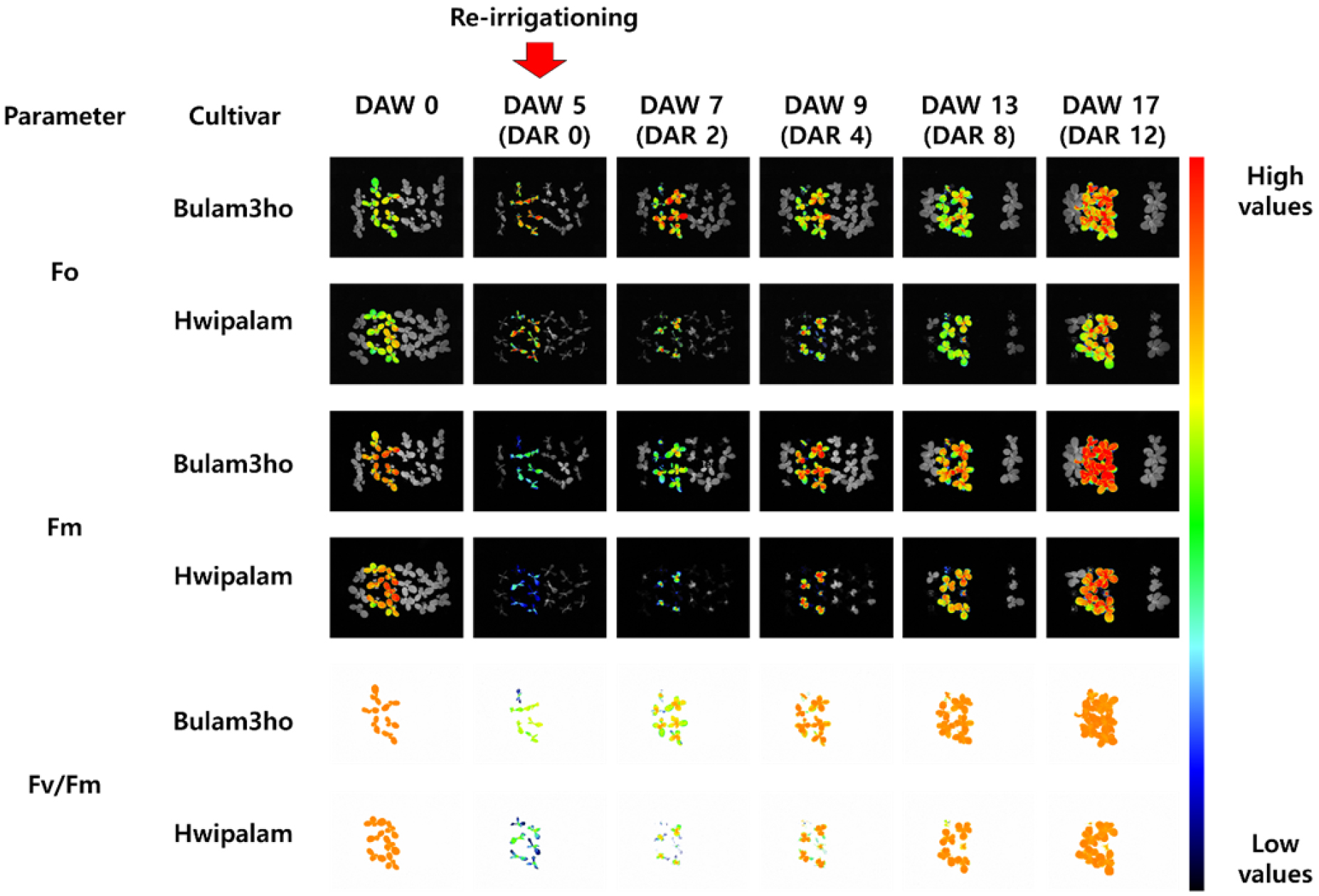
Fig. 6
Changes in chlorophyll fluorescence parameters including the minimum fluorescence in the dark-adapted state (Fo), maximum fluorescence in the dark-adapted state (Fm), and maximum PSII quantum yield (Fv/Fm) of kimchi cabbage cultivars ‘Bulam3ho’, and ‘Hwipalam’ as a function of days after water deficit treatment (DAW). The red arrow indicates the time point of re-irrigation.
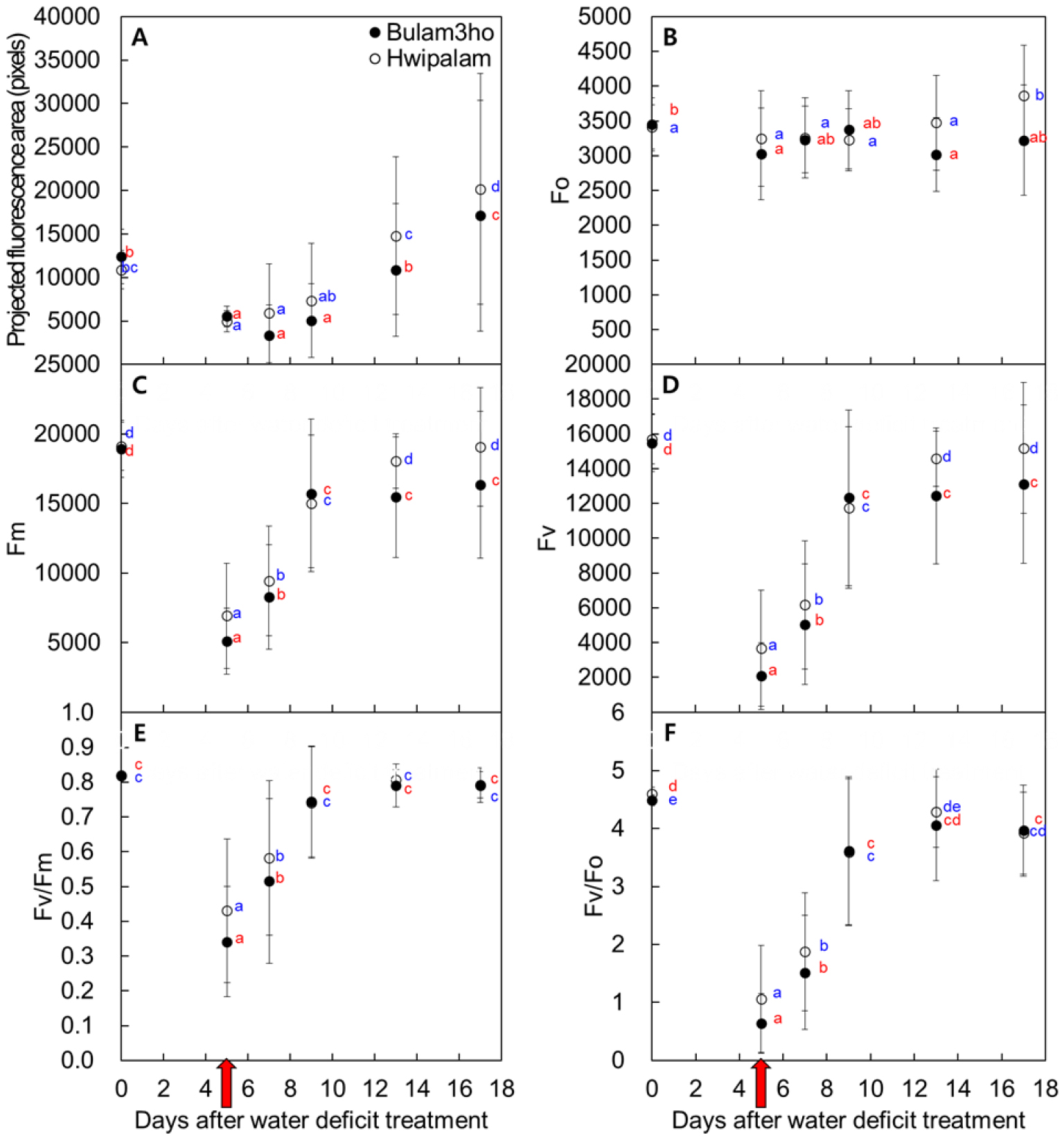
Fig. 7
Changes in chlorophyll fluorescence parameters of kimchi cabbage cultivars ‘Bulam3ho’ and ‘Hwipalam’ throughout the entire experimental period following the initiation of water deficit treatment. The parameters include the projected fluorescence area (A), minimum fluorescence in the dark-adapted state (Fo; B), maximum fluorescence in the dark-adapted state (Fm; C), variable fluorescence (Fv; D), maximum PSII quantum yield (Fv/Fm; E), and maximum potential efficiency of PSII (Fv/Fo; F). According to Duncan’s multiple range test at the p ≤ 0.05 level, means followed by different letters are significantly different. 'Bulam3ho' is indicated in blue letters, and 'Hwipalam' in red letters. Data in (A, B, C, and D) represent the mean ± standard deviation (n = 24).
Pearson’s correlation analysis revealed strong associations among CF, gas exchange, and morphological traits (Figs. 7 and 8). Fv/Fm showed highly significant correlations with Fm (r = 0.855, p ≤ 0.01), Fv (r = 0.864, p ≤ 0.01), Fv/F0 (r = 0.860, p ≤ 0.01), Tr (r = 0.791, p≤ 0.01), and Gs (r = 0.785, p ≤ 0.01). Moreover, Fv/Fm was positively correlated with the number of leaves, plant leaf area, and plant fresh weight (p ≤ 0.05), suggesting that higher PSII efficiency promotes carbon assimilation and biomass accumulation. In contrast, Fv/Fm was negatively correlated with Ci (r = -0.825, p ≤ 0.01), indicating that enhanced PSII efficiency was associated with reduced internal CO2 accumulation. No significant correlations were observed for DMC or LWC. Although Fv/Fm was not significantly correlated with overall survival of plants, it showed a moderate positive association with survival of leaves (p = 0.09 Fig. 8). Collectively, these findings demonstrate that Fv/Fm and Fv/F0 serve as reliable indicators of photosynthetic performance and post-drought recovery, supporting their potential as predictive indices of drought tolerance.
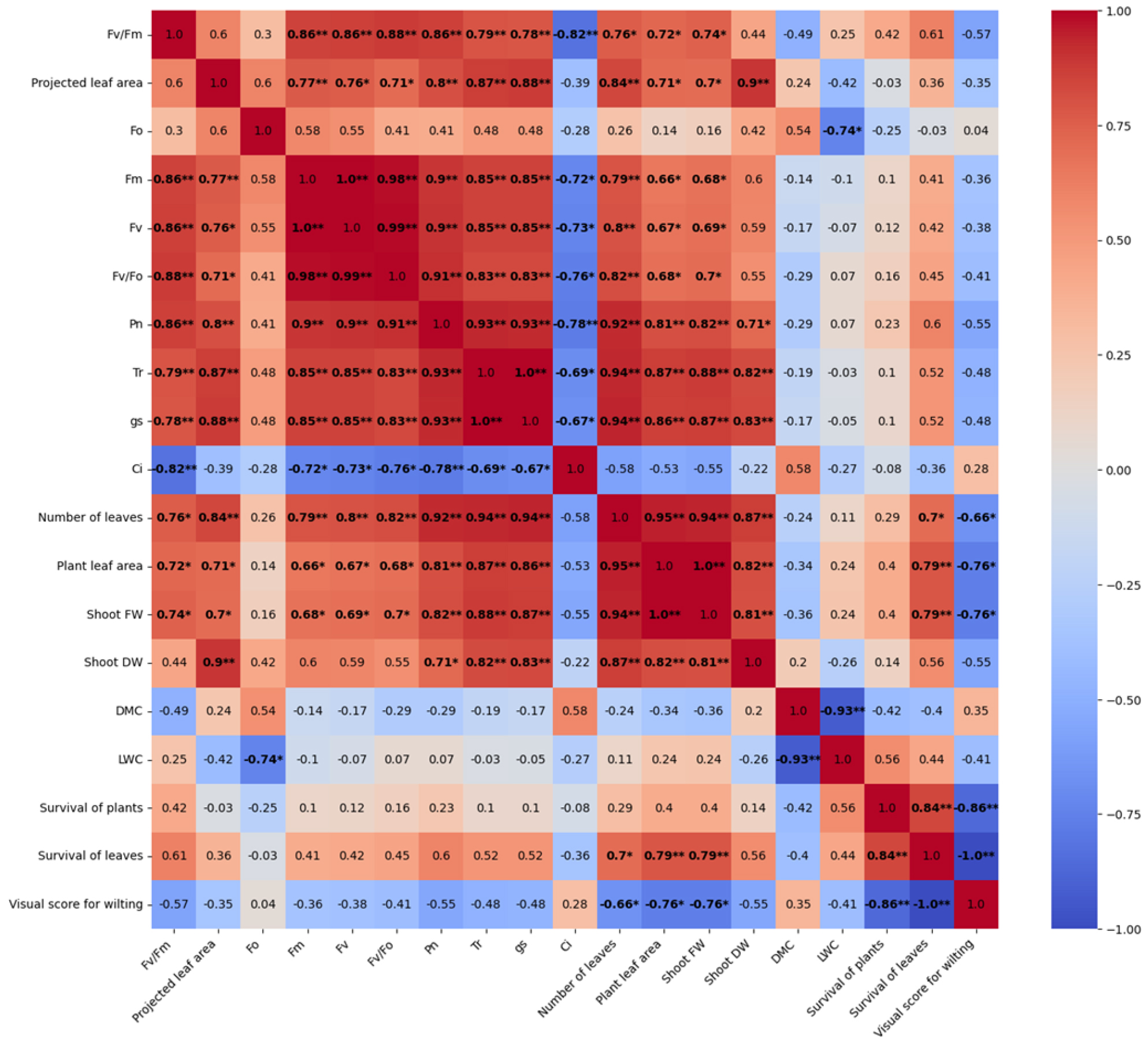
Fig. 8
Pearson correlation analysis between the chlorophyll fluorescence parameters (Fv/Fm, Fo, Fm, Fv, and Fv/Fo), growth characteristics (number of leaves, plant leaf area, shoot FW, shoot DW, DMC, and LWC), photosynthetic parameters (Pn, Tr, Gs, and Ci), and visual assessments of kimchi cabbage cultivars during the recovery period following the water deficit treatment. Positive and negative correlations are indicated in red and blue, respectively, and the color intensity reflects the magnitude of the correlation coefficients. Asterisks indicate significance levels (*, **, and *** for p ≤ 0.05, p ≤ 0.01, and p ≤ 0.001, respectively).
Discussion
In this study, we investigated the morphological, physiological, and CF responses of two kimchi cabbage cultivar (‘Bulam3ho’ and ‘Hwipalam’) to drought stress and the subsequent recovery after re-irrigation. The results revealed both common and cultivar-specific resilience strategies, providing insights into the mechanisms underlying drought tolerance and recovery in kimchi cabbage.
1. Growth and development responses to drought stress and recovery
Water deficit treatment significantly reduced the plant leaf area, plant fresh weight, and LWC in both cultivars, whereas DMC increased. Such reductions are typical of drought stress, during which decreased turgor pressure limits cell expansion and leading to smaller leaves and reduced canopy size (Medyouni et al., 2021; Yang et al., 2021). Compared with the morphological parameters at 0 DAW, plant leaf area decreased by 30.6% in ‘Bulam3ho’ and 39.4% in ‘Hwipalam’, indicating that growth and development inhibition in both cultivars, with slightly stress-induced damage in ‘Hwipalam.’ Previous studies reported that drought stress induces leaf curling, scorching, and wilting, which reduce leaf area (Chauhan et al., 2023; Jang et al., 2024; Li et al., 2023; Seleiman et al., 2021; Shawon et al., 2018). The observed increase in DMC and the stable plant dry weight of ‘Hwipalam’ during recovery suggest that biomass changes under drought were primarily due to water loss rather than degradation of dry matter. As leafy vegetables contain a high proportion of water, their internal water status is strongly affected by environmental conditions. Hence, drought mainly disrupts the water balance before structural biomass loss occurs (Antunović Dunić et al., 2023; Yang et al., 2021). In addition, re-irrigation can temporarily enhance water use efficiency, allowing greater biomass gain accumulation per unit water, as reported in rice (Chatterjee et al., 2025).
During the drought period, the number of leaves continued to increase in both the cultivars. This suggests that leaf initiation and appearance were not directly inhibited, even though leaf expansion was restricted. This observation is consistent with previous reports indicating no significant difference in number of leaves between control and drought-treated Chinese cabbage plants (Jang et al., 2024). The rate of leaf appearance is mainly governed by temperature, with water availability exerting secondary effects (Jamieson et al., 1995; Jang et al., 2024; Shin et al., 2021; Wi et al., 2020). In this experiment, the relatively high temperatures were may have accelerated leaf emergence. Overall, these results suggest that drought stress primarily limits leaf expansion rather than the rate of leaf appearance in kimchi cabbage.
2. Cultivar variation in recovery dynamics after re-irrigation
Following re-irrigation, both ‘Bulam3ho’ and ‘Hwipalam’ resumed growth and development. However, ‘Bulam3ho’ consistently exhibited faster recovery across several parameters. During the early recovery phase (0 to 1 DAR), the RLAR of ‘Bulam3ho’ was 2.4 times higher than ‘Hwipalam’, indicating a more rapid initiation of new leaf emergence. Similarly, the RLER and RGRtfw were higher in ‘Bulam3ho’ throughout most of the recovery period, reflecting earlier restoration of cell expansion and turgor pressure (Taiz et al., 2023; Yang et al., 2021). In contrast, ‘Hwipalam’ showed slower increases in both fresh and dry weights of the plant, suggesting a delayed resumption of assimilate accumulation. These results demonstrate that recovery dynamics differ substantially among kimchi cabbage genotypes (Jang et al., 2024), reflecting the fundamental differences in their stress response mechanisms.
Plants adopt diverse strategies to cope with drought, including tolerance, recovery, avoidance, and escape (Haghpanah et al., 2024). The early and consistent restoration of growth observed in ‘Bulam3ho’ corresponds to a ‘rapid recovery’ strategy, characterized by fast restoration of leaf area expansion and biomass accumulation once water availability improves. A similar study has been observed in rice, which maximized light interception after exposure to stress (Okami et al., 2015). In contrast, ‘Hwipalam’ exhibits a relatively ‘slow recovery’ strategy, maintaining photochemical stability while delaying biomass partition until turgor and photosynthetic capacity were fully restored. This strategy reflects an avoidance or tolerance strategy, rather than a recovery mechanism.
More broadly, drought adaptation involves the interaction between avoidance, tolerance, and recovery processes (Chen et al., 2015; Seleiman et al., 2021). The contrasting strategies of the two cultivars, rapid recovery with high growth resumption in ‘Bulam3ho’ and conservative photoprotective behavior in ‘Hwipalam’, illustrate distinct adaptive frameworks that can be leveraged for breeding drought-resilient kimchi cabbage cultivars.
3. Photosynthetic responses and recovery
Drought stress reduces the photosynthetic capacity of both cultivars by restricting CO2 assimilation and gas exchange processes. Under water deficit, the limitation of gas exchange and photochemical activity was accompanied by reductions in chlorophyll content and damage to both PSII and PSI (Abbas et al., 2023; Chauhan et al., 2023; Issarakraisila et al., 2007; Ke et al., 2025; Mohan et al., 2025; Yang et al., 2021). In both ‘Bulam3ho’ and ‘Hwipalam’, significant decreases were observed in Pn, Tr, and Gs, whereas Ci increased markedly. The decline in Pn to negative values at 5 DAW indicates that respiration exceeded carbon fixation under severe drought stress (Chevilly et al., 2021; Jang et al., 2024; Rabha and Uprety, 1998; Singh et al., 2009; Wu et al., 2012). Similar responses have been reported in Brassica species. When drought prolonged for more than six days, net assimilation approached zero or became negative even when the soil moisture was approximately 20% (Guadagno et al., 2017).
The simultaneous decline in Tr and Gsby more than 85% reflects stomatal closure, a primary defense mechanism that limits transpiration water loss (Ejaz et al., 2023). Such reductions in Gs have also been documented in rapeseed, spring wheat, Chinese cabbage, tomato, and strawberry (Ahmad Lone et al., 2022; Ejaz et al., 2023; Li et al., 2023; Li et al., 2025). However, stomatal closure also restricts CO2 uptake, thereby decreasing photosynthetic carbon assimilation (Shin et al., 2019).
Both cultivars showed an increase in Ci despite the decrease in Gs and Pn. This suggests that CO2 entering the leaf was not efficiently assimilated, implying a transition from stomatal to non-stomatal limitation of photosynthesis (Mohan et al., 2025). Similar responses have been reported in Brassica species, where drought-tolerant cultivars exhibit smaller reductions in Pn, Gs, and Tr while maintaining relatively high Ci compared with sensitive ones (Li et al., 2023; Li et al., 2025). Elevated Ci values thus indicate impaired mesophyll conductance and decreased biochemical activity of the Calvin cycle, leading to internal CO2 accumulation (Issarakraisila et al., 2007). Overall, these results demonstrate that prolonged drought stress suppresses photosynthesis through both stomatal and non-stomatal mechanisms.
Following re-irrigation, both cultivars exhibited rapid restoration of photosynthetic function. Pn, Tr, and Gs increased sharply during early recovery and reached near-maximum values by 8-12 DAR. The reopening of stomata enhanced CO2 diffusion and transpiration, promoting carbon assimilation as water status was restored (Arief et al., 2023). These observations confirm that the photosynthetic apparatus of kimchi cabbage can recover efficiently from short-term drought once water availability is restored, although the speed and extent of recovery differ between cultivars.
4. CF as a stress and recovery indicator
CF is a reliable and non-destructive indicator for monitoring photosynthetic efficiency and stress-induced photoinhibition in plants (Maxwell and Johnson, 2000; Baker, 2008). In particular, CF parameters provide quantitative information into the functional status of PSII, enabling the early detection of physiological impairment and the assessment of recovery following rehydration (Guo et al., 2022; Shin et al., 2019; Yao et al., 2018).
Both Fv/Fm and Fv/F0 decreased substantially at 5 DAW, indicating transient photoinhibition and reduced maximum PSII efficiency under severe drought stress. Both cultivars demonstrated a larger decline in Fv/Fm and Fv/F0, with 58.2% and 85.8% in ‘Bulam3ho,’ and 47.5% and 77.1% in ‘Hwipalam,’ suggested higher photodamage during severe drought stress. However, the relative results differed, suggesting genotype-specific differences in photochemical sensitivity. ‘Hwipalam’ exhibited a smaller reduction in Fv/Fm, indicating relatively greater photochemical stability, whereas ‘Bulam3ho’ showed stronger suppression during stress but a more rapid recovery thereafter. Similar PSII impairment has been widely documented across species, including tomato, strawberry, and grafted watermelon, where drought stress leads to a pronounced decline in Fv/Fm (Yao et al., 2018).
During the early recovery phase (5 to 7 DAW, same as 0 to 2 DAR), ‘Hwipalam’ maintained a slightly higher Fv/Fm, showing a more effective restoration of PSII efficiency once the water status was restored. The response of CF parameters varies considerably depending on the type, intensity, and duration of stress, as well as the plant genotype (Shin et al., 2021). In grafted watermelon seedlings, the Fv/Fm index decreased during severe drought stress, whereas no significant differences were observed under mild drought stress. Therefore, Fv/Fm has been identified as the most reliable parameter for assessing recovery following re-irrigation (Shin et al., 2019). Because Fv/Fm represents the maximum quantum yield of PSII, it serves as an integrative indicator of both stress tolerance and recovery potential across genotypes (Guo et al., 2022).
In addition to Fv/Fm, Fv/F0 represents the quantum yield of primary PSII photochemistry and serves as another important indicator of its functional state (Medyouni et al., 2021; Zhang et al., 2018). In tomato, Fv/F0 values decrease under water deficit, from 4.865 ± 0.180 in control plants to 4.492 ± 0.219 under stress, indicating the sensitivity of this parameter to PSII damage (Medyouni et al., 2021). Furthermore, similar to the results of the current study, drought stress decreases both minimal and maximal fluorescence intensities (Medyouni et al., 2021). Collectively, Fv/Fm and Fv/F0 provide integrative and complementary insights on PSII efficiency, making them effective indicators for evaluating drought-induced damage and subsequent recovery in kimchi cabbage.
The observed correlation between Fv/Fm, gas-exchange parameters (Pn, Tr, Gs), and morphological traits further confirms the connection between PSII function and overall photosynthetic capacity during stress and recovery. As stomatal reopening restores CO2 assimilation, Fv/Fm simultaneously increases, indicating the reactivation of photochemical processes. Under severe drought, when non-stomatal factors dominate, PSII impairment becomes the principal constraint on photosynthesis, as evidenced by the concurrent decline in Fv/Fm and Pn and the transient increase in Ci (Flexas and Medrano, 2002). This transitional pattern, from stomatal to non-stomatal limitation, was evident in the simultaneous changes of Ci and Fv/Fm, supporting the use of CF parameters as sensitive indicators of photosynthetic recovery. A decline in Fv/Fm indicates a reduction in the maximum photochemical efficiency of PSII, whereas decreased Fv/Fo reflects impaired primary photochemistry (Baker, 2008; Murchie and Lawson, 2013). Thus, the integration of CF parameters with gas exchange and morphological traits offers a quantitative indices for evaluating both the extent of drought-induced damage and the efficiency of recovery processes (Maxwell and Johnson, 2000).
5. Implications on breeding and crop management
The contrasting recovery patterns observed in ‘Bulam 3ho’ and ‘Hwipalam’ suggest different drought-resilience strategies. ‘Bulam3ho’ showed rapid canopy expansion and biomass restoration, following re-irrigation, suggesting an adaptive ‘rapid recovery’ mechanism that supports early resumption of photosynthetic activity and carbon assimilation under fluctuating water availability. In contrast, ‘Hwipalam’ demonstrated slower recovery, which may protect the PSII system more effectively during the drought period, potentially providing advantages under repeated drought stress conditions.
From a breeding perspective, these results suggest that both early recovery capacity and photochemical stability are important. Although traditional visual assessment is a simple way to assess stress symptoms, it is subjective and has limited temporal resolution, which reduces its reliability for quantitative screening. In contrast, CF parameters, particularly Fv/Fm and Fv/Fo, provide quantitative, reproducible, and non-destructive measurements of PSII efficiency that can enable consistent evaluation of both stress severity and recovery capacity across genotypes. Nevertheless, the present study assessed a limited set of CF parameters. Expanding future analyses to include additional variables, such as qP, qL, Rfd, and NPQ, would provide a more comprehensive view of PSII photochemical regulation and energy dissipation mechanisms under fluctuating water conditions.
The combination of CF parameters with morphological and physiological traits provides a comprehensive index for identifying genotypes with an effective drought strategy, characterized by both high drought tolerance and rapid post-stress recovery. Such integration is now technically feasible through advances in high-throughput phenotyping (HTP) systems, which enable automated, non-destructive, and monitoring of physiological and morphological traits.
Recent HTP technologies, such as automated CF imaging, hyperspectral reflectance, and thermal infrared, facilitate the simultaneous quantification of PSII efficiency, leaf temperature, and morphological characteristics under both controlled and field conditions. By applying these technologies, breeders can accurately trace the temporal dynamics of stress imposition and recovery, identifying genotypes characterized by early restoration of Fv/Fm and Fv/Fo or sustained photochemical stability under drought cycles. When combined with morphological and physiological indices, this technology can be used as a crucial indicator for breeding drought-tolerant kimchi cabbage cultivars. Moreover, the integration of CF-derived parameters within HTP workflows can accelerate breeding pipelines by enabling early-stage selection of genotypes with superior recovery phenotypes before maturity. Ultimately, replacing or complementing subjective visual assessments with automated, image-based quantification of PSII efficiency will enhance the accuracy, reproducibility, and throughput of drought-resilience screening in kimchi cabbage and other horticultural crops.
Conclusion
This study demonstrated that drought stress significantly reduced the growth, development, and photosynthetic performance of kimchi cabbage seedlings by decreasing plant leaf area, plant fresh weight, and PSII photochemical efficiency, as indicated by substantial decreases in Fv/Fm and Fv/Fo. Recovery after re-irrigation revealed cultivar-specific differences in the responses. ‘Bulam3ho’ showed fast recovery of leaf area and photosynthetic capacity, indicating a ‘rapid-recovery’ strategy driven by efficient water reabsorption and accelerated canopy re-expansion. In contrast, ‘Hwipalam’ maintained higher Fv/Fm stability and slower biomass accumulation, suggesting a more conservative, photoprotective response that minimizes PSII damage during stress. These contrasting responses suggest two complementary drought-resilience strategies, a rapid post-stress recovery type (‘Bulam3ho’) and a photochemically stable type (‘Hwipalam’). These strategies contribute differently to the overall drought adaptation potential of kimchi cabbage seedlings.
Correlation analyses confirmed that Fv/Fm and Fv/Fo, were closely associated with gas exchange and morphological traits, supporting their value as reliable, non-destructive indicators for assessing both drought-induced damage and recovery efficiency. Integrating CF data with morphological and physiological traits thus provides a comprehensive framework for quantifying drought resilience across genotypes and stress stages. Future studies should expand the assessment to additional CF parameters such as qL, qP, Rfd, and NPQ. Furthermore, the integration of CF-based phenotyping into HTP platforms can enhance breeding efficiency by enabling early and objective selection of genotypes exhibiting superior recovery dynamics and photochemical resilience under variable water regimes.