Introduction
Materials and Methods
1. Plant materials and germination conditions
2. Cultivation environments
3. Light treatments
4. Nutrient solution management
5. Plant growth measurements
6. Photosynthesis measurement
7. Statistical analysis
Results and Discussion
Conclusion
Introduction
By 2050, the global population is projected to reach 9.8 billion, with approximately 66% residing in urban areas. This rapid urbanization, coupled with the adverse effects of climate change, water scarcity, and the decline of arable land, presents significant challenges to global agricultural productivity (Hang et al., 2019). In response, plant factories have emerged as a promising solution in modern agriculture, offering the potential to enhance food security, ensure stable production, and improve economic sustainability (Kozai et al., 2019).
Plant factories are highly controlled cultivation systems in which environmental parameters such as light intensity and spectrum, temperature, humidity, CO2 concentration, and nutrient supply are precisely regulated to optimize plant growth. These systems enable consistent, year-round crop production regardless of external climatic or geographical conditions (Cha et al., 2012). Furthermore, plant factories promote efficient resource use and are particularly well-suited for high-density cultivation of high-value crops, especially leafy vegetables and herbs (Park, 2016).
Among the various environmental factors, light plays a pivotal role in plant growth and photosynthesis, serving as the primary energy source driving these physiological processes (Wang et al., 2016). It influences multiple plant functions, including morphology, biomass accumulation, and secondary metabolite production (Samy, 2023). The three principal light parameters affecting plant development are light quality (spectrum), light intensity, and photoperiod, each exerting distinct effects on plant physiology and metabolic activity (Shafiq et al., 2021). Among these, light intensity has a particularly strong impact on the growth rate and productivity of leafy vegetables and herbs (Mohamed et al., 2021). In plant factories, optimizing light intensity is essential for achieving efficient and sustainable crop production, with a photosynthetic photon flux density (PPFD) of 200-300 μmol·m-2·s-1 generally considered optimal for leafy vegetables (Cha et al., 2012).
Due to their compact morphology and short growth cycles, leafy vegetables and herbs are commonly cultivated in vertically stacked, high-density systems in plant factories (Kozai, 2012). Among these crops, lettuce (Lactuca sativa L.) is widely grown owing to its rapid growth, low energy demand, high nutritional value, and short production period (Yudina et al., 2023). Basil (Ocimum basilicum L.), a high-value aromatic herb, is recognized for its fast growth and rich content of bioactive compounds with antimicrobial and antioxidant properties, making it commercially important across culinary, pharmaceutical, and cosmetic industries (Saha et al., 2016). Both crops are commonly used in commercial plant factories and serve as model species in studies exploring plant responses to light environments (He et al., 2021). Previous studies have shown that yield, morphology, and secondary metabolite production in these species are significantly affected by light conditions (Anum et al., 2024; Larsen et al., 2020).
In most plant factories, light intensity is maintained at a constant level throughout the photoperiod and across consecutive days. To date, most studies have focused on the effects of constant light intensity or the combined influence of light intensity and photoperiod, which together determine the daily light integral (DLI). However, in natural environments, light intensity fluctuates both diurnally and from day to day. While some research has addressed plant responses to diurnal changes, limited information is available on crop responses to day-to-day fluctuations in light intensity under conditions of equal cumulative light. Therefore, this study aimed to investigate the growth responses of lettuce and basil to various daily fluctuating light intensity regimes applied during a defined cultivation period (the final seven days before harvest), with the light integral kept constant across all treatments.
Materials and Methods
1. Plant materials and germination conditions
Lettuce (L. sativa L. ‘Frillice’) and basil (O. basilicum L. ‘Sweet basil’) were used as plant materials. Seeds were sown in pre-soaked urethane sponges and germinated under dark conditions for two days for lettuce and three days for basil.
2. Cultivation environments
Lettuce and basil were cultivated in a plant factory located at the University of Seoul. The air temperature was controlled at 24/22°C (day/night), and the relative humidity was maintained at 65 ± 5%. White LED lighting provided a PPFD of 150 μmol·m-2·s-1 with a 16-hour photoperiod (16 h light / 8 h dark). The planting density was 0.022 plants·cm-2 for both species. A deep flow technique (DFT) hydroponic system was used for cultivation.
3. Light treatments
At 25 days after sowing (DAS), lettuce and basil were subjected to eight different light treatments. The cumulative light integral for seven days was uniformly fixed at 100.8 mol·m-2 across all treatments. The control treatment maintained a constant light intensity of 250 μmol·m-2·s-1, while treatments L1-L7 included daily fluctuations in light intensity as described in Table 1.
Table 1.
Daily light intensity patterns of the eight light treatments applied in this study.
4. Nutrient solution management
A Yamazaki nutrient solution, suitable for leafy vegetables, was used. The pH was maintained at 6.0-6.5. Electrical conductivity (EC) was set at 1.0 dS·m-1 light treatment before and increased to 1.6 dS·m-1 for lettuce and 2.0 dS·m-1 for basil from 25 DAS onward.
5. Plant growth measurements
Measured growth parameters included leaf length, number of leaves, SPAD value, leaf area, and shoot and root fresh and dry weights for lettuce. For basil, plant height was also recorded. SPAD values were obtained using a chlorophyll meter (SPAD-502PLUS, Konica Minolta, Tokyo, Japan), and leaf area was measured with a leaf area meter (LI-3100, LI-COR, Lincoln, NE, USA). Fresh weights were determined using an electronic balance (KERN EWJ 300-3, Kern & Sohn, Balingen, Germany), and dry weights were obtained after drying samples at 70°C for 7 days.
6. Photosynthesis measurement
Photosynthetic rates were measured daily from one to seven days after the start of light treatments using a portable photosynthesis system (CIRAS-4, PP Systems, Amesbury, MA, USA). Measurements were conducted on the third fully expanded leaf of each plant.
7. Statistical analysis
All data were analyzed using SAS software (version 9.4; SAS Institute Inc., Cary, NC, USA). Two-way analysis of variance (ANOVA) was conducted to evaluate the main and interaction effects of light fluctuation treatment and crop species. Differences among treatment means were further examined using Duncan’s multiple range test at a significance level of p < 0.05.
Results and Discussion
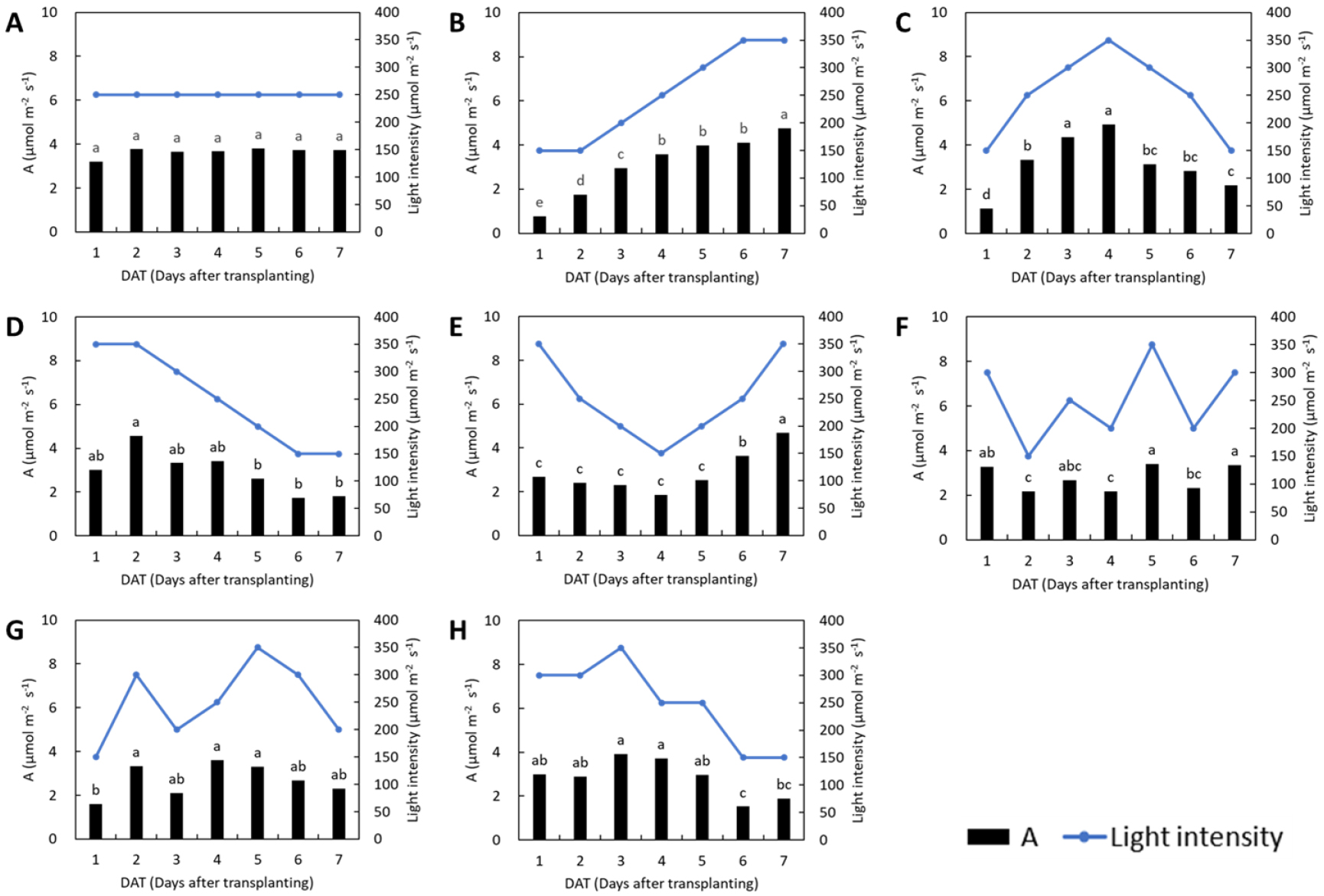
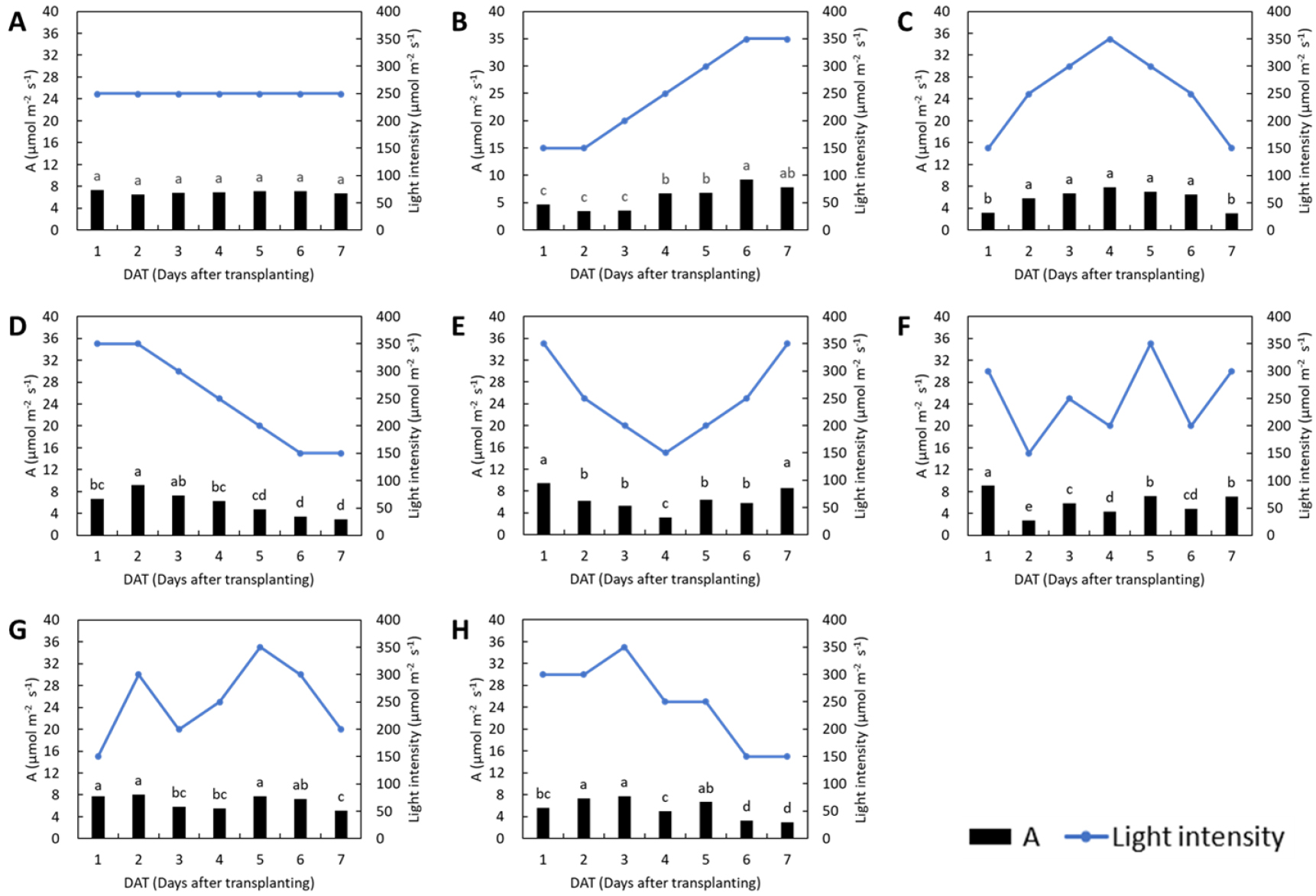
Lettuce in all treatments exhibited similar trends in photosynthetic responses to changes in light intensity (Fig. 1). In the control, where the light intensity remained constant, the photosynthetic rate remained stable throughout the treatment period. On days with a consistent high light intensity (350 µmol·m-2·s-1), the photosynthetic rate on day 7 in the L1 treatment and day 2 in the L3 treatment increased significantly compared to the previous day. This indicates delayed acclimation to high light, as photosynthetic performance improved following initial exposure. In the L2 treatment, the photosynthetic rate varied in accordance with the increase–decrease pattern of light intensity. In the L4 treatment, no significant change in the photosynthetic rate was observed during the initial reduction in light intensity, but a significant increase occurred following the subsequent rise in light intensity. Treatments with irregular light patterns (L5, L6, and L7) also showed changes in photosynthesis generally corresponding to light intensity fluctuations. In the L5 and L7 treatments, which began under high light intensity, the photosynthetic rate on day 1 did not increase markedly, suggesting a limited initial response to elevated light levels. For basil, the photosynthetic rate remained stable in the control treatment with constant light intensity (Fig. 2). In the L1 treatment, the photosynthetic rate tended to increase with rising light intensity; however, unlike in lettuce, the rate on day 7 under the 350 µmol·m-2·s-1 condition did not increase compared to day 6. In the L2 treatment, the photosynthetic rate was significantly lower only on days 1 and 7 under the 150 µmol·m-2·s-1 condition, with no significant differences observed during days 2-6 despite fluctuations in light intensity. In the L3 treatment, the photosynthetic rate significantly decreased in response to reduced light intensity, while under the 350 µmol·m-2·s-1 condition, an increase not observed on day 1 became apparent on day 2. The L4, L5, L6, and L7 treatments also exhibited response patterns that generally corresponded to changes in light intensity. In the L6 treatment, despite an increase in light intensity from 150 to 300 µmol·m-2·s-1 between days 1 and 2, the photosynthetic rate did not differ significantly. Photosynthesis is driven by the absorption of light energy and its efficiency generally increases with increasing light intensity (Zhou et al., 2019). However, it is also essential to understand how photosynthesis responds to temporal fluctuations in light intensity, rather than considering only its response to steady-state levels. This study was therefore conducted to investigate the effects of varying light fluctuation patterns on photosynthetic responses. Yin and Johnson (2000) investigated the potential of several species to acclimate to fluctuating light environments and found that plants were capable of photosynthetic acclimation by increasing their light-saturated photosynthetic rate (Pmax), although the degree of acclimation varies by species.
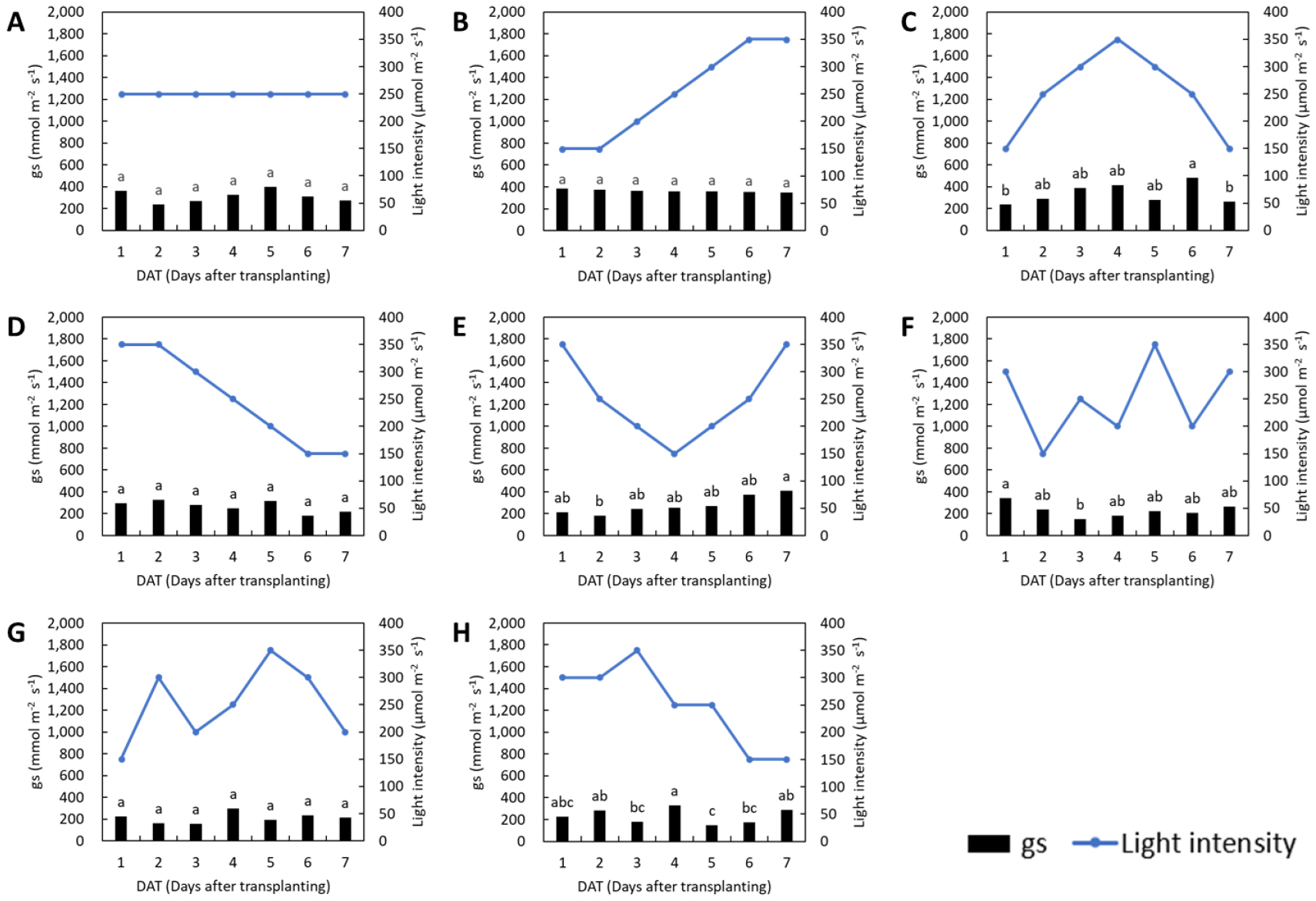
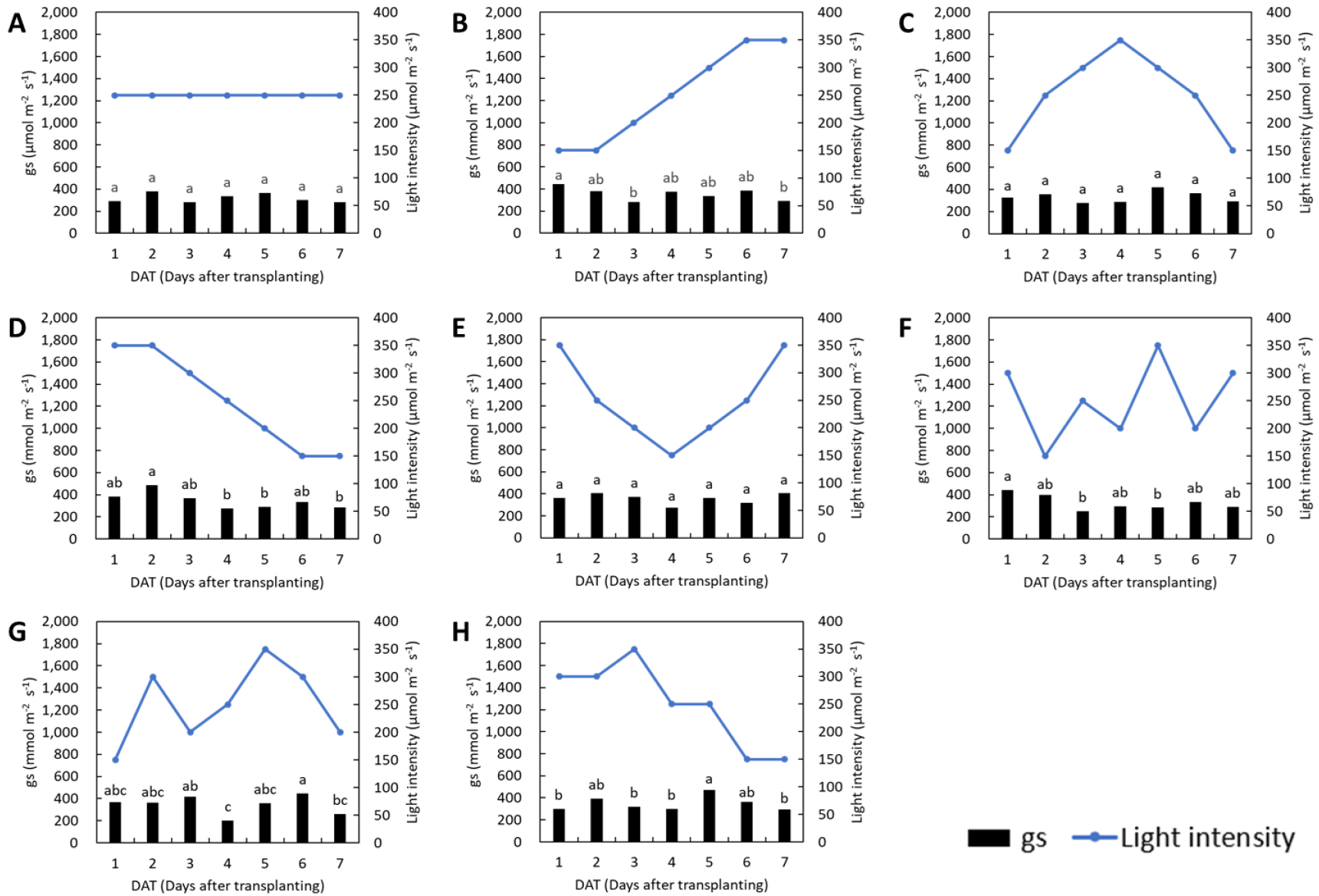
The changes in stomatal conductance in response to fluctuations in light intensity were less pronounced than those observed in the photosynthetic rate for both lettuce and basil (Figs. 3 and 4). In the control groups of both species, stomatal conductance remained stable throughout the treatment period. In lettuce, stomatal conductance showed no significant changes under the L1, L3, and L6 treatments, while in basil, no significant differences were observed under the L2 and L4 treatments. Previous research has shown that exposure to low light prior to high light conditions delays photosynthetic induction by extending the time required to reach a steady-state photosynthetic rate, and induces stomatal limitation by restricting CO2 diffusion into the leaf (Yamori et al., 2020). In this study, a gradual increase in light intensity did not lead to a reduction in stomatal conductance in either species, whereas a rapid increase in light intensity resulted in a significant decrease.
The initial growth of lettuce and basil prior to the light fluctuation treatments is shown in Table 2. Although all treatments received the same cumulative light integral, lettuce and basil exhibited distinct growth responses after seven days of light fluctuation (Tables 3 and 4). In lettuce, the highest growth was observed in both the control (constant light intensity at 250 µmol·m-2·s-1) and the L5 treatment, which involved daily fluctuations with varying amplitudes. It has been reported that, under constant light conditions, the regulation of photosynthetic electron transport and CO2 fixation is optimized to enhance photosynthetic efficiency and reduce the generation of reactive oxygen species (ROS) (Wang et al., 2024). Consequently, it is hypothesized that lettuce demonstrated enhanced growth in the control treatment, attributable to these conducive conditions. In contrast, the lowest growth was observed in the L6 treatment, where both fresh and dry weights were approximately half of those in the highest-performing treatments. For basil, the highest growth was recorded in the L2 treatment, which followed a gradual increase and subsequent decrease in light intensity. In contrast, lower growth was observed in the L4 and L5 treatments. Notably, the L5 treatment, which involved the most extreme fluctuations in daily light intensity, including three instances of abrupt changes to 150 µmol·m-2·s-1, promoted the highest growth in lettuce. However, it resulted in the lowest growth in basil, suggesting species-specific responses to fluctuating light environments. In the L2 treatment, where basil exhibited the highest growth, lettuce showed a sharp decline in photosynthetic rate when the light condition shifted from an increasing to a decreasing phase. In contrast, basil maintained a relatively stable photosynthetic rate without noticeable changes during the transition from 350 to 200 µmol·m-2·s-1. This physiological difference may explain the disparity in growth responses between basil and lettuce under fluctuating light conditions. Furthermore, fluctuations in light intensity caused more pronounced changes in leaf area, fresh weight, and dry weight in lettuce than in basil. According to the two-way ANOVA results (Table 5), significant interaction effects (A×B) were observed for leaf area and dry weights, suggesting species-specific responses to light fluctuation patterns. The ‘Frillice’ lettuce used in this study is a cultivar developed specifically for cultivation under controlled environment conditions, whereas ‘Sweet basil’ is a genotype selected from varieties typically grown in open-field conditions. Therefore, the greater variability in growth observed in ‘Frillice’ lettuce across different light fluctuation treatments may be attributed to its sensitivity to changes in light environments.
Table 2.
Initial growth of lettuce and basil before the light fluctuation treatment.
Table 3.
Growth parameters of lettuce after 7 days of fluctuating light intensity.
| Treatment |
Leaf length (cm) |
No. of leaves (/plant) | SPAD value |
Leaf area (cm2/plant) | Fresh weight (g) | Dry weight (g) | ||
| Shoot | Root | Shoot | Root | |||||
| Control | 13.1 az | 11.0 a | 30.4 a | 253 a | 21.45 a | 4.90 a | 1.05 a | 0.21 ab |
| L1 | 12.1 a | 10.7 a | 35.6 a | 211 ab | 16.08 ab | 4.32 a | 0.73 ab | 0.13 cd |
| L2 | 12.1 a | 09.3 ab | 31.9 a | 218 a | 17.68 ab | 4.46 a | 0.83 ab | 0.17 bc |
| L3 | 12.5 a | 10.0 ab | 35.8 a | 240 a | 18.93 a | 3.91 a | 0.87 ab | 0.13 cd |
| L4 | 12.5 a | 11.3 a | 33.5 a | 266 a | 19.91 a | 3.50 a | 1.01 a | 0.17 bc |
| L5 | 12.3 a | 11.0 a | 33.6 a | 250 a | 20.31 a | 4.84 a | 1.03 a | 0.23 a |
| L6 | 13.1 a | 08.0 b | 32.2 a | 136 b | 10.10 b | 2.96 a | 0.51 b | 0.11 d |
| L7 | 12.7 a | 09.3 ab | 33.6 a | 201 ab | 15.69 ab | 4.16 a | 0.74 ab | 0.19 ab |
Table 4.
Growth parameters of basil after 7 days of fluctuating light intensity.
| Treatment |
Plant height (cm) |
Leaf length (cm) |
No. of leaves (/plant) | SPAD value |
Leaf area (cm2/plant) | Fresh weight (g) | Dry weight (g) | ||
| Shoot | Root | Shoot | Root | ||||||
| Control | 6.3 az | 8.2 c | 12.7 a | 33.4 bc | 164 b | 6.72 b | 4.77 bcd | 0.59 b | 0.19 a |
| L1 | 5.9 ab | 8.9 abc | 10.3 a | 33.1 c | 155 bc | 6.42 bc | 3.79 e | 0.58 b | 0.17 a |
| L2 | 5.5 abc | 9.9 a | 15.3 a | 36.0 ab | 177 a | 7.76 a | 5.79 a | 0.71 a | 0.21 a |
| L3 | 6.0 a | 8.8 bc | 14.3 a | 34.5 abc | 160 b | 6.77 b | 5.06 abc | 0.50 c | 0.19 a |
| L4 | 5.8 abc | 9.0 abc | 11.3 a | 33.9 bc | 142 de | 5.89 c | 4.03 de | 0.44 c | 0.17 a |
| L5 | 5.1 bc | 8.4 bc | 13.3 a | 34.3 abc | 130 e | 5.16 d | 3.94 de | 0.46 c | 0.19 a |
| L6 | 5.0 c | 9.3 ab | 12.3 a | 34.0 bc | 146 cd | 6.13 bc | 4.42 cde | 0.57 b | 0.21 a |
| L7 | 5.0 c | 9.2 abc | 14.3 a | 36.9 a | 157 bc | 6.80 b | 5.38 ab | 0.60 b | 0.22 a |
Table 5.
Effects of light fluctuation treatment (A), crop species (B), and their interaction (A × B) on plant growth parameters.
The light environment plays a key role in shaping morphological traits such as shoot and root biomass, plant height, and leaf area, as well as physiological processes including floral initiation, rooting, shoot formation, and leaf unfolding (Chen et al., 2004; Kim, 1999). As light intensity directly influences photosynthesis, it also affects overall plant growth and productivity. Numerous studies have shown that higher light intensity enhances growth rate, leaf expansion, photosynthetic efficiency, and chlorophyll content (Anum et al., 2024; Suh, 2017; Yang et al., 2018). According to Kelly et al. (2020), lettuce growth increases with higher light integrals, and under equivalent light integral conditions, growth is further promoted under lower light intensity with extended photoperiods. Chang and Chang (2014) also reported that lettuce growth and nitrogen accumulation vary depending on light quality during different growth stages. For basil, Larsen et al. (2022) found that fresh weight, dry matter content, and plant height increased with higher cumulative light integrals. Moreover, short-term application of high PPFD before harvest improved both fresh and dry biomass. In contrast to previous studies, our results revealed that basil growth decreased in the L4 and L5 treatments, where high PPFD was applied only on the final day. Unlike Larsen et al. (2022), who applied high PPFD for five days, the sudden one-day increase may have imposed stress on the plants, limiting their growth.
Under diurnally dynamic lighting conditions, lettuce readily adjusted its photosynthetic processes in response to prevailing light intensities, enabling it to maintain consistent growth and productivity. Based on these findings, Mäkinen et al. (2025) suggested that DLI can be flexibly redistributed across the photoperiod without compromising overall plant growth. In basil, Kaiser et al. (2024) reported that the marketable fresh weight was not significantly affected by hourly alterations in light intensity compared to plants grown under constant light conditions. However, in our study involving daily fluctuations in light intensity, we found that even under the same cumulative light integral during the treatment period, growth responses varied depending on the light fluctuation pattern and crop species, when compared to constant light conditions. While the treatment was applied only during the final seven days before harvest, this finding supports the view that adjustment to changing light intensities requires coordinated regulation of photosynthesis, photoprotection, and overall plant physiology to maintain stable growth under fluctuating light conditions (Niu et al., 2024).
Conclusion
This study investigated the photosynthetic and growth responses of lettuce and basil under varying patterns of daily light intensity fluctuation, applied under identical light integral conditions. Despite receiving the same light integral, plant growth varied depending on the fluctuation pattern, with responses being species-specific. These findings highlight the practical value of applying crop-specific light fluctuation strategies during selected periods of the cultivation cycle to enhance growth performance. In commercial plant factories, implementing diurnally dynamic lighting often requires additional system costs and may induce unintended changes in microclimate conditions due to fluctuating light environments. Therefore, an alternative approach to applying daily light fluctuation could involve spatially dividing cultivation zones with different fixed light intensities and moving the cultivation trays, according to a programmed daily light fluctuation strategy. To optimize the effects of daily light fluctuation strategies on crop yield and quality, further studies are needed to explore optimal photoperiod settings in combination with daily light variation schemes that provide different cumulative light levels at each growth stage throughout the entire cultivation period.