Introduction
Climate change–induced extreme high and low temperatures, as well as fluctuations in light intensity, reduce the stability of seedling production in protected horticulture. As a countermeasure, plant factory–based seedling production systems, which allow precise control of light, temperature, humidity, and nutrients, have gained increasing attention. By shielding seedlings from external environmental fluctuations, these systems enable the year-round production of uniform and high-quality seedlings. With the recent division of labor in seedling production and the expansion of the market, the demand for technologies capable of supplying standardized, high-quality seedlings on a stable, year-round basis has further increased. Watermelon (C. lanatus) is highly sensitive to environmental conditions during the early growth stage, and seedling quality is directly linked to transplant establishment, subsequent growth stability, and ultimately the final yield and fruit quality (Ertle and Kubota 2022; Korkmaz and Dufault 2001). To reduce disease incidence and enhance stress tolerance, grafted seedlings using rootstock–scion combinations are widely adopted in commercial watermelon production (Razi et al., 2024). Grafting has also been reported to overcome continuous cropping disorders and improve both productivity and fruit quality, establishing it as an essential technique in commercial cultivation (Devi et al., 2020).
While plant factory-based seedling production systems enable the uniform production of seedlings, research on optimal air temperature strategies that can simultaneously ensure graft union formation, stem elongation and thickening, and root development remains limited (Yang et al., 2016). Research on air temperature optimization using seedling quality indices in plant factory systems—where stable environmental control minimizes external environmental influences—has been very limited. Air temperature is a critical factor regulating seedling growth through fundamental physiological processes such as photosynthesis, respiration, and cell division and elongation. Excessive high temperatures may promote stem elongation but suppress thickening and root development, leading to etiolation and reduced seedling quality, whereas low temperatures can limit photosynthetic efficiency and cell division, thereby delaying overall growth (Li et al., 2024). Therefore, seedling quality cannot be sufficiently evaluated by simple growth parameters alone, and integrative indices such as the top-to-root (T/R) ratio, compactness, and crop growth rate (CGR) must be considered for a more comprehensive.
Therefore, this study aimed to identify the optimal daytime air temperature for grafted watermelon seedlings in a plant factory-based seedling production system, with the goal of contributing to the establishment of a stable year-round seedling production framework. In addition, the results of this study are expected to provide an important basis for advancing environmental control strategies in plant factory- based seedling systems.
Materials and Methods
In this study, grafted watermelon seedlings commonly used for low-temperature seedling production were selected, with ‘DongjangGun’ (‘DG’, Syngenta Korea Co., Ltd., Korea) serving as the rootstock and ‘SantaKkul’ (‘SK’, Nongwoobio Co., Ltd., Korea) as the scion. Rootstock seeds were sown in 32-cell trays (53 cm × 28 cm × 6.4 cm), and scion seeds were sown in 162-cell trays (54 cm × 28 cm × 4.6 cm), both filled with a commercially available artificial horticultural substrate (Eoullim, Shinsung Mineral Co., Korea). Grafting was performed at 12 days after sowing, when the first true leaves had expanded. Following grafting, seedlings were placed in a healing chamber for 6 days to promote graft union formation. Environmental conditions in the healing chamber were maintained at a light intensity of 10-30 µmol·m-2·s-1, a temperature of 28°C, and a relative humidity of 90 ± 5%. After healing, the grafted watermelon seedlings were cultivated for 21 days in a plant factory- based seedling production system installed at the experimental farm of Mokpo National University (34.94 N, 126.67 E).
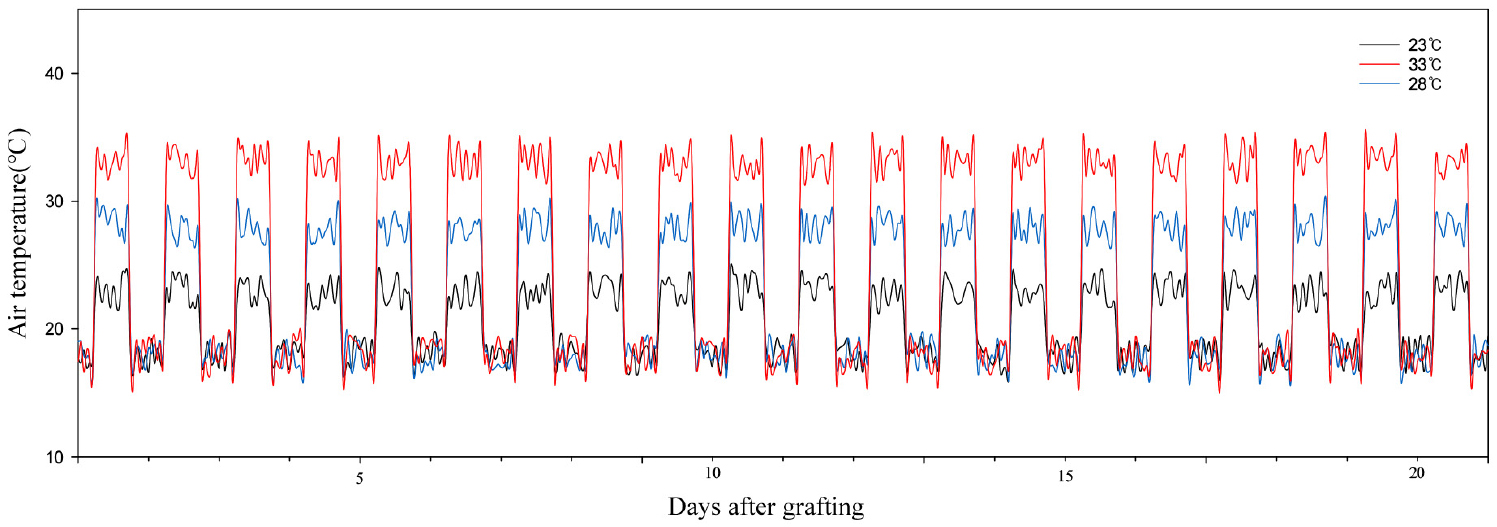
The environmental conditions in the seedling production system consisted of three different daytime air temperature treatments of 23 ± 2°C, 28 ± 2°C, and 33 ± 2°C, while the other conditions were kept constant, with a nighttime temperature of 18 ± 2°C, a relative humidity of 90 ± 5%, and a photoperiod of 12 h light and 12 h dark. Temperature and humidity were continuously monitored and recorded in real time using a data logger (WatchDog 1000 Series Micro Stations, Spectrum Technologies Inc., USA) (Figs. 1 and 2). Nutrient solution was supplied according to the PBG standard nutrient solution formula for protected fruit vegetables, adjusted to an EC of 1.84 dS·m-1 and pH 6.15 [KNO3 505g·t-1, Ca(NO3)2H2O 708g·t-1, NH4H2PO4 115g·t-1, MgSO4·7H2O 369g·t-1, H3BO3 3g·t-1, MnSO4·4H2O 2g·t-1, ZnSO4·5H2O 0.22g·t-1, CuSO4·5H2O 0.05g·t-1, Na2MoO4·2H2O 0.02g·t-1, EDTA-Fe 20g·t-1], and applied once daily via sub-irrigation. The light source was a plant growth LED (FSPG-1200OW, Shinhwa General Construction Co., Ltd., Korea) (Fig. 3). From the end of the healing period until 21 days after grafting, the photosynthetic photon flux density (PPFD) was maintained at 250 ± 25 µmol·m-2·s-1. Light intensity was measured at the shoot apex using a light meter (HD2102.1, Delta OHM, Italy). Growth measurements of grafted watermelon seedlings were conducted at 7, 14, and 21 days after grafting, with 15 replicates per treatment. Plant height was measured from the shoot base to the shoot apex, and root length was determined from the shoot base to the tip of the longest root. The number of stem nodes and total leaves was recorded. Leaf area was measured using a leaf area meter (LI-3100C, LI-COR, Inc., USA), and stem diameter was measured 1 cm above the shoot base using a digital caliper (CD-PS, Mitutoyo, Japan). Shoot and root fresh and dry weights were determined using an electronic balance. For dry weight measurement, samples were oven-dried at 70°C for more than 168 h in a drying oven (Upright Convection Oven, JSOF-250T, JSR, Korea). For seedling quality evaluation, the top-to-root ratio (T/R ratio), compactness, and crop growth rate (CGR) were calculated using the following formulas.
In the calculation of crop growth rate (CGR), W2 represents the total dry weight of an individual plant (including both shoot and root) on the measurement date, while W1 denotes the mean initial dry weight.
Statistical analyses were performed using SPSS software (Version 29.0.2.0, IBM SPSS Inc., USA). Mean comparisons among treatments were conducted using Duncan’s multiple range test at the p < 0.05 significance level. Graphs were generated using SigmaPlot software (Version 15.0, Systat Software Inc., USA).
Results and Discussion
This study was conducted to develop techniques for producing high-quality grafted watermelon seedlings in a plant factory system. Grafted seedlings were cultivated for 21 days under air temperature treatments of 23°C, 28°C, and 33°C, and their growth characteristics—including plant height, root length, number of leaves, leaf area, stem diameter, shoot and root fresh and dry weights, compactness, crop growth rate (CGR), and top-to-root (T/R) ratio—were evaluated.
Plant height increased significantly with rising air temperature. At 7, 14, and 21 days after grafting, grafted seedlings under the 33°C condition were tallest, showing significant differences among treatments (Table 1; Fig. 4). This indicates that higher temperatures promoted stem elongation. Similar findings were reported by Ohtaka et al.(2020), who observed that high temperature stimulated stem elongation accompanied by increased levels of gibberellins and indole-3-acetic acid. In the present study, gibberellin and indole-3-acetic acid concentrations also increased at 21 days under the 33°C condition, resulting in excessive elongation (31.87 cm) compared with 23°C (21.85 cm) and 28°C (25.71 cm), thereby leading to etiolation. Excessive elongation can weaken tissue structure and reduce both transplanting success and seedling quality (Jia et al., 2023). Thus, the increase in plant height under high temperature should be interpreted not only as growth promotion but also as a decline in seedling quality.
Table 1.
Growth characteristics of watermelon grafted seedlings grown under low-temperature conditions as affected by days after grafting and temperature treatments.
|
Days after grafting |
Temperature (°C) |
Plant height (cm) |
Root length (cm) |
No. of Nodes (/plant) |
No. of Leaves (/plant) |
Leaf area (cm2) |
Stem Diameter (mm) |
Fresh weight (g) |
Dry weight (g) | ||
| Shoot | Root | Shoot | Root | ||||||||
| 7 | 23 | 6.71 cz | 14.79 a | 1.27 a | 4.93 a | 46.06 b | 4.48 b | 4.82 c | 4.82 b | 0.42 c | 0.05 c |
| 28 | 7.29 b | 14.67 a | 1.33 a | 5.13 a | 61.59 a | 5.63 a | 6.01 a | 1.94 a | 0.57 a | 0.06 b | |
| 33 | 7.55 a | 14.41 a | 1.60 a | 5.07 a | 62.11 a | 5.57 a | 5.81 b | 1.99 a | 0.48 b | 0.07 a | |
| 14 | 23 | 10.43 c | 15.61 b | 1.87 b | 5.00 b | 48.39 b | 5.16 b | 4.82 c | 1.79 b | 0.46 c | 0.06 b |
| 28 | 14.81 b | 15.68 b | 2.47 a | 4.60 b | 65.94 a | 5.63 a | 6.37 a | 2.05a | 0.59 b | 0.08 a | |
| 33 | 18.90 a | 16.27 a | 2.93 a | 6.00 a | 65.41 a | 5.53 a | 6.16 b | 1.97 a | 0.68 a | 0.08 a | |
| 21 | 23 | 21.85 c | 17.94 b | 4.93 c | 6.13 b | 56.16 c | 5.49 b | 6.39 c | 1.46 c | 0.57 c | 0.05 b |
| 28 | 25.71 b | 19.47 a | 6.00 b | 6.60 b | 73.60 a | 5.81 a | 8.62 b | 2.22 a | 0.72 b | 0.08 a | |
| 33 | 31.80 a | 17.03 c | 7.07 a | 7.33 a | 67.64 b | 5.74 a | 8.92 a | 1.99 b | 0.84 a | 0.07 a | |
Root length was greatest under the 33°C condition at 14 days after grafting, but at 21 days it was longest under the 28°C condition at 19.47 cm, with significant differences among conditions. These results suggest that root cell elongation and division were promoted under the optimal temperature. Luo et al. (2020) also reported that high temperatures altered root structure, enhancing root elongation within an optimal range but suppressing growth under excessive heat. Similarly, Tokić et al. (2023) found that high temperatures beyond a certain threshold reduced primary and lateral root growth by inhibiting cell division. Consistent with these findings, the present study showed that while plant height was greatest under 33°C, root length was shortest under the same condition, indicating that extreme temperatures may restrict root elongation.
The number of nodes did not differ markedly among conditions at 7 and 14 days after grafting; however, by 21 days it was highest under the 33°C condition, with 7.07 nodes, compared with 4.93 and 6.00 nodes under the 23°C and 28°C conditions, respectively. This suggests that stem elongation under the 33°C condition contributed to increased plant height and consequently a greater number of nodes.
Leaf area did not differ significantly between the 28°C and 33°C conditions at 7 and 14 days after grafting, but by 21 days it was greatest under the 28°C condition. In contrast, the number of leaves was highest under the 33°C condition from 14 to 21 days. Generally, an increase in both leaf number and leaf area enhances photosynthetic capacity and contributes positively to biomass accumulation (Weraduwage et al., 2015), whereas reductions in these traits limit photosynthesis and restrict overall growth (Hu et al., 2020). Under unfavorable conditions such as water stress, decreased turgor pressure and inhibited cell expansion reduce leaf area, resulting in lower assimilate production and biomass (Khan et al., 2025). In this study, leaf area at 21 days was smaller under the 23°C and 33°C conditions compared with the 28°C condition, suggesting that these temperature regimes were less favorable for grafted seedling growth.
Stem diameter was greater under the 28°C and 33°C conditions than under the 23°C condition from 7 to 21 days after grafting, with no significant difference between 28°C and 33°C; however, it was consistently thicker under the 28°C condition than under 33°C. This suggests that excessive stem elongation under 33°C limited sufficient thickening. A thicker stem enhances the transport capacity of water and nutrients, improving shoot stability and contributing positively to fruit development (Peschiutta et al., 2013; Yang et al., 2022). Moreover, greater stem diameter facilitates the accumulation and distribution of photosynthates, supporting stable early growth and ensuring productivity at the fruiting stage (Feng et al., 2023; Samarah et al., 2021). Thus, the increase in stem diameter under the 28°C condition can be interpreted as a key physiological response ensuring grafted seedling stability.
Shoot fresh weight was highest under the 28°C condition at 7 and 14 days after grafting, but at 21 days it was greatest under the 33°C condition at 8.92 g. Root fresh weight was highest under the 28°C and 33°C conditions at 7 and 14 days, whereas at 21 days it was greatest under the 28°C condition at 2.22 g. Shoot dry weight was highest under the 28°C condition at 7 days, but at 14 and 21 days it was greatest under the 33°C condition. Root dry weight was highest under the 33°C condition at 7 days, while at 14 and 21 days both the 28°C and 33°C conditions were higher than the 23°C condition. These results indicate that shoot biomass accumulation was promoted under higher temperature conditions, whereas root biomass development was more favorable under the optimal temperature of 28°C. Excessive shoot growth under 33°C may have led to assimilate allocation favoring the aerial parts rather than root thickening. In contrast, the balanced increase in both shoot and root biomass at 28°C suggests that this temperature supports more stable grafted seedling growth. Previous studies have shown that biomass partitioning between shoots and roots is highly sensitive to temperature, with high temperatures often enhancing shoot growth at the expense of root development (Poorter et al., 2012; Wang et al., 2016).
In addition to morphological and biomass traits, seedling quality was further assessed using the T/R ratio, compactness, and crop growth rate (CGR), which are widely recognized as integrative indicators of seedling vigor. A higher T/R ratio, approaching 1, indicated relatively greater root growth compared with shoot growth, thereby reflecting superior root system development (Jeong et al., 2020). Compactness, calculated as the shoot dry weight divided by plant height, showed a positive association with sturdier and higher-quality seedlings, consistent with previous findings (Ju et al., 2023). Similarly, CGR, which represents the increase in dry matter per unit area over a given period, was higher in vigorous seedlings, indicating more active biomass accumulation (Sridhar et al., 2019).
The T/R ratio was closest to 1 under the 33°C condition at 7 days, under the 23°C and 28°C conditions at 14 days, and under the 28°C condition at 21 days (Fig. 5). Compactness was highest under the 28°C condition at 7 days, under the 23°C and 28°C conditions at 14 days, and again under the 28°C condition at 21 days. CGR was greatest under the 28°C condition at 7 days, under the 33°C condition at 14 days, and under both the 28°C and 33°C conditions at 21 days. Although slight differences were observed at 7 and 14 days, all three indices indicated that the 28°C condition was the most effective by 21 days after grafting. These indices complement the direct measurements of morphological and physiological traits: the higher compactness under 28°C reflected sturdier growth, consistent with greater stem diameter and balanced biomass allocation between shoots and roots. In contrast, excessive shoot elongation under 33°C resulted in lower compactness and a less favorable T/R ratio, despite higher shoot biomass. Such imbalances may compromise transplanting performance and reduce overall seedling quality, whereas 28°C can be considered the optimal air temperature for producing vigorous and well-balanced grafted watermelon seedlings.
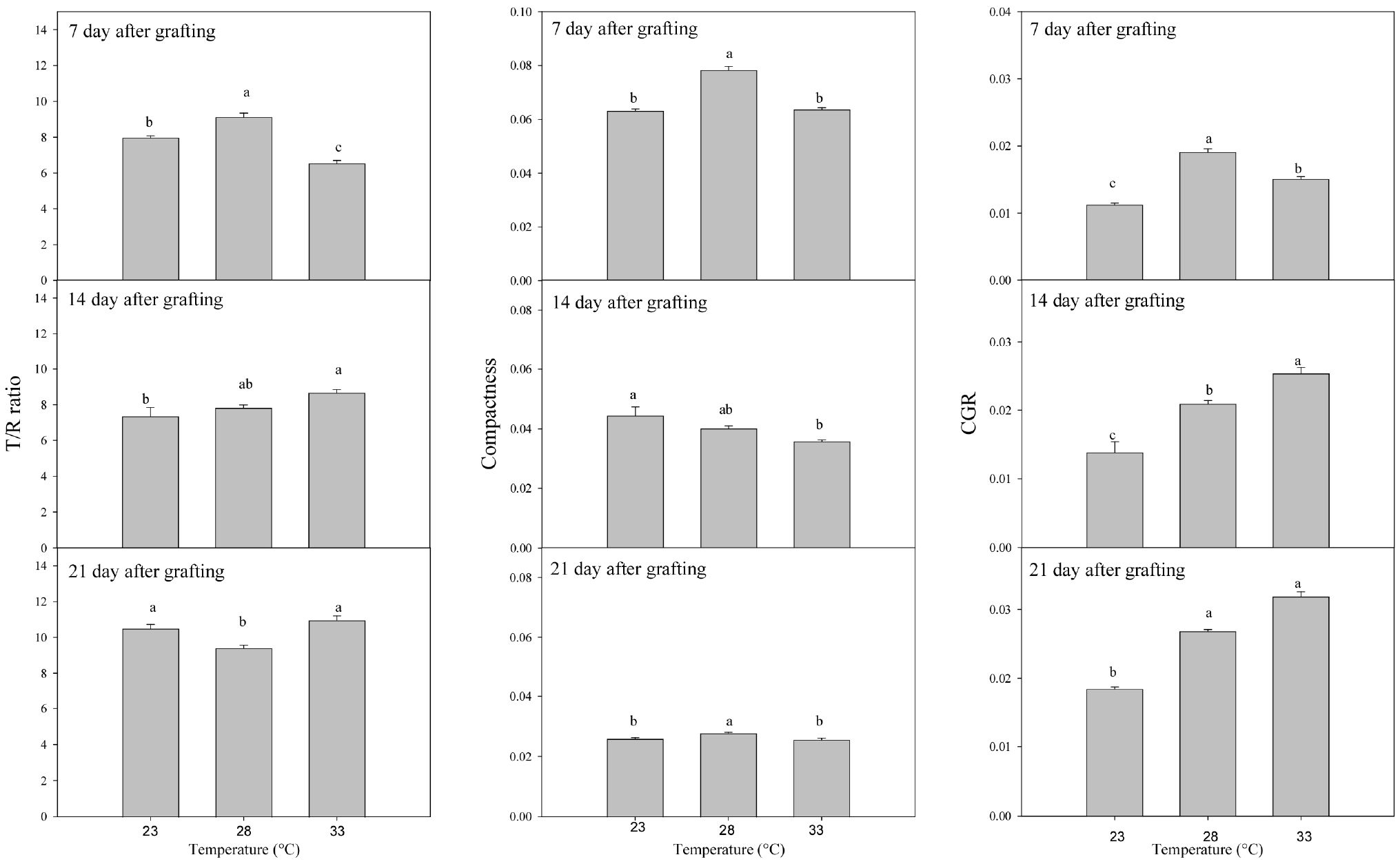
Fig. 5
Top-to-root ratio (T/R ratio), compactness, and crop growth rate (CGR) of watermelon seedlings grafted with ‘SantaKkul’ scion onto ‘DongjangGun’ rootstock at 7, 14, and 21 days after grafting under different air temperatures. Vertical bars represent the standard error of the mean. Different letters indicate significant differences based on Duncan’s multiple range test at p < 0.05.
Overall, analysis of growth responses up to 21 days after grafting showed that most growth traits were superior under the 28°C and 33°C conditions compared with 23°C. However, excessive shoot elongation and reduced stem thickening under 33°C indicated a decline in grafted seedling quality. In contrast, the 28°C condition consistently promoted balanced growth, sturdier morphology, and more favorable quality indices such as T/R ratio, compactness, and CGR. These findings suggest that 28°C represents the optimal air temperature for producing vigorous and well-balanced grafted watermelon seedlings in plant factory systems.