Introduction
The commercial blueberry production in Korea began in the early 2000s and since then the producing area has been rapidly expanding, e.g., 112 ha in 2007 to 1,516 ha in 2013 (Kim, 2013). This rapid expansion of the producing area should require the establishment of proper propagation methods for Korean growers and nurserymen. The main type of blueberry growing in Korea is the northern highbush blueberry (Vaccinium corymbosum). Practically highbush blueberries can be propagated by both hardwood and softwood cuttings, with preference usually given to the hardwood cuttings (Schulte and Hancock, 1983). The hard wood cuttings are known to be the easiest to handle and are less perishable, since cuttings are taken from the previous year’s growth when the stems are dormant.
It is known that rooting of the blueberry cuttings is not difficult (Strik et al., 1993) but their rooting capacity and root growth may be influenced according to cultivars, growing media, cutting take time and use of PGRs. Hormonal rooting agents containing auxin helps adventitious rooting of cuttings. Indolebutyric acid (IBA) is known to be most effective; other auxins often used are indoleacetic acid (IAA) and naphthalene acetic acid (NAA); IBA is more effective in rooting than IAA or NAA (Fogaca and Fet-Neto, 2005). For highbush blueberries, IBA increases rooting at least for softwood cuttings (Eck and Childers, 1989; Celik and Odabas, 2009) whereas its effect is inconsistent for hardwood cuttings (Eck and Childers, 1989). Lee and Lee (2009) reported that IBA soaking treatments at 50 and 100 mg · L−1 for 20 h had no effect on the rooting of ‘Rancocas’ highbush blueberry cuttings and rather seemed to retard their shoot growth of rooted cuttings. Some studies evaluated the IBA effect on the establishment of the cutting by dipping the cutting bases for just a few seconds into a more concentrated solution. Sun and Bassuk (1991) reported that IBA at a range of 0~1000 mg · L−1 logarithmically increased the rooting capability of MM.106 apple rootstocks, but higher levels of IBA proved inhibitive. It is generally known that too high concentrations of auxin could contribute to basal rotting especially in softwood cuttings, thus should be avoided (Hartmann et al., 2002). To our knowledge, naphthalene acetic acid (NAA) has rarely been tested as a rooting agent for hardwood cut- tings of blueberry, but this hormone readily promotes rooting of the tissue cultured plantlets of apple (Lane and McDougald, 1982) and Eucalyptus species (Fogaca and Fet-Neto, 2005).
The light levels given to the cuttings during the rooting period may affect rooting capacity of cuttings. During the initial rooting period, especially before the substantial amount of roots has developed, shading over the cutting bed may be required to control the surface temperature of the cuttings, since increased surface temperature can lead to increased water loss out of the cutting tissues thus decreasing the establishment success of the cutting. Little studies with regards to this matter have been conducted with blueberry.
Thus the objectives of this study were to determine the effects of rooting agents on rooting and growth of the hardwood cuttings of highbush blueberries, and to investigate how the shading treatment imposed over the cutting bed influences their establishment success. Three highbush blueberry cultivars ‘Bluecrop’, ‘Duke’ and ‘Sunrise’ were used.
Methods and Materials
1. Plant materials and hardwood cuttings
Three highbush blueberry varieties were used in this study, including ‘Bluecrop’, ‘Duke’ and ‘Sunrise’. Hardwood cuttings were taken from strong, healthy shoots of the past season’s growth that were about 30 cm long and 5 mm or lower in diameter. The cuttings were stored at 3°C in plastic bags lined with moist paper until the cutting. In early April, cuttings were made 12 cm long having about five buds when cutting bases were cut on a slant, with the terminal portion of the shoot with flower buds being removed. Treated cuttings were pushed on a slant into rooting medium (peat moss : sand = 7 : 3 in volume; pH 4.5) in the propagation box of 50 cm L × 35 cm W × 10 cm H, until only their top two buds were exposed. Before planting, rooting medium of 9 cm in depth had been soaked for 4 h to insure uniform moisture distribution. Fifty cuttings per propagation box were planted with three replicates per treatment. The propagation boxes were placed in a plastic rain shelter where there was no mist system and neither light nor temperature was controlled. A single shoot per cutting was allowed to grow.
Ninety days after the cutting, rooted cuttings were transplanted into individual plastic pots (12 cm in diameter)
containing only peat moss for 90 days of further growth. The plants were immediately fertigated with 2000-fold diluted solution of Hyponex (5N-10P-5K) fertilizer at 300 ml per pot, three times at 20-day intervals.
2. Expt. 1: Response of highbush blueberry cuttings to rooting agent treatments
This experiment was conducted in early April, 2008. Before planting, one cm-long, freshly cut bases of the cuttings were quickly dipped into solutions of IBA and NAA for 5 s, both at 0, 500 and 1000 mg · L−1 in 50% ethanol. They were also treated with Rootone® powder (0.4% 1- naphthylacetamide (NAD)) and the excess amount of the powder on the cuttings was removed by light tapping. After planting, the cutting bed was covered with a single layer of black shade cloth of 70% light penetration, placed one meter over the bed, for 90 days of the rooting period until early July.
Thirty days after the cutting, the percent bud break of the cuttings was measured, and 60 days later, the time when rooted cuttings were ready to be transplanted into individual pots, the percent rooting, root growth, shoot length and leaf number were measured. Also the proportion of the cuttings with no roots was recorded when divided into two groups, only callus formed and dead. The establishment success of the rooted cuttings transplanted into individual pots was evaluated after 90 days of further culture with Hyponex fertilizer, when the total number of shoot branches and leaves, and mean branch length and thickness were measured.
3. Expt. 2: Effects of shading treatments on hardwood cuttings of highbush blueberries
This experiment was conducted in early April, 2007 and used the same varieties as in Expt. 1. Immediately after the cutting, a single layer of black shade cloth of different shading levels (30, 50, 70 and 90%) was placed one meter over the cutting bed. This coverage remained for 90 days of the rooting period from early April to early July. During that period, temperatures of the rooting medium were daily monitored using Campbell 107B soil temperature sensors connected to a CR-10X datalogger (Campbell Scientific Co., USA), and averaged for the applicable periods of respective months. The relative difference in leaf chlorophyll content was indirectly determined using SPAD-502 meter (Minolta, Japan). Other measurements were the same as in Expt. 1.
4. Experimental design and data analysis
For both experiments, the propagation boxes (a box with 50 cuttings per treatment) in a plastic rain shelter were arranged in a completely randomized design for each variety, with three replicates. For comparing treatment means, Fisher’s least significant difference (LSD) was computed at a significance level of 5% by the SAS GLM procedure.
Results and Discussion
1. Expt. 1: Response of highbush blueberry cuttings to rooting agent treatments
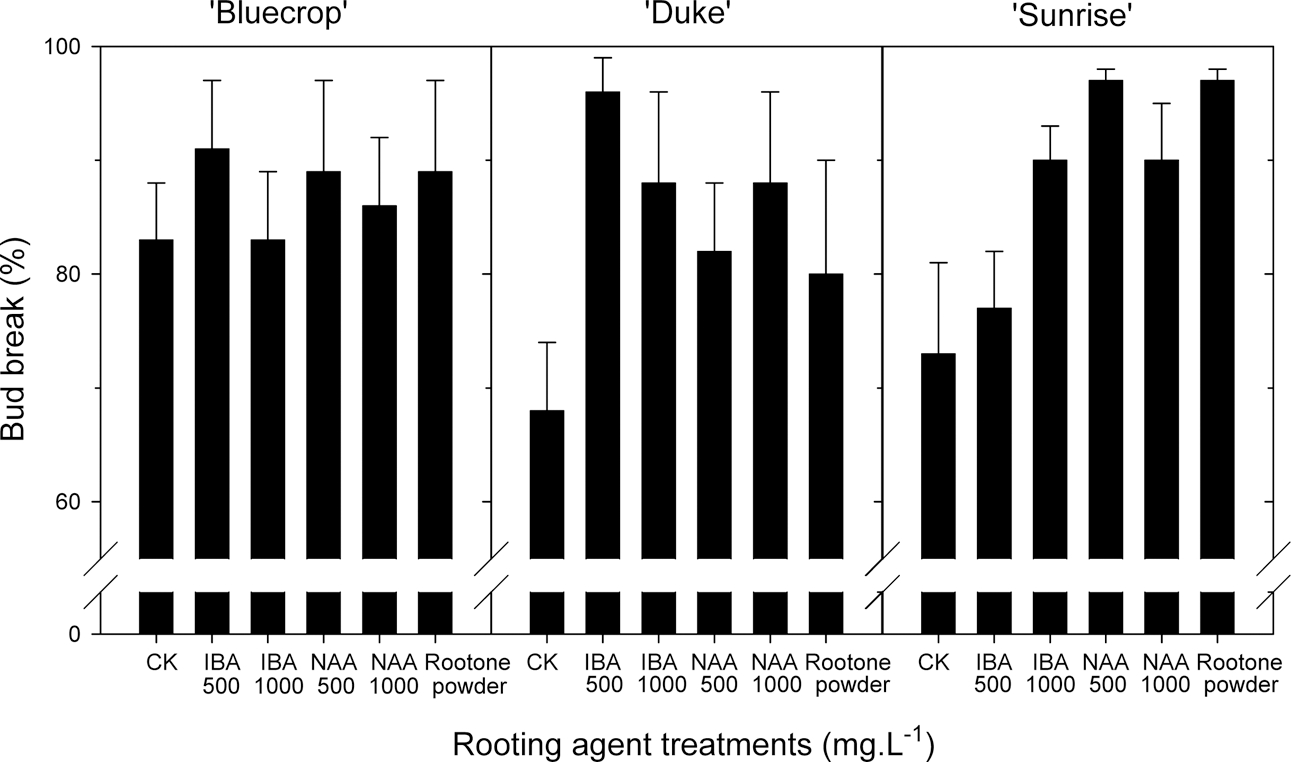
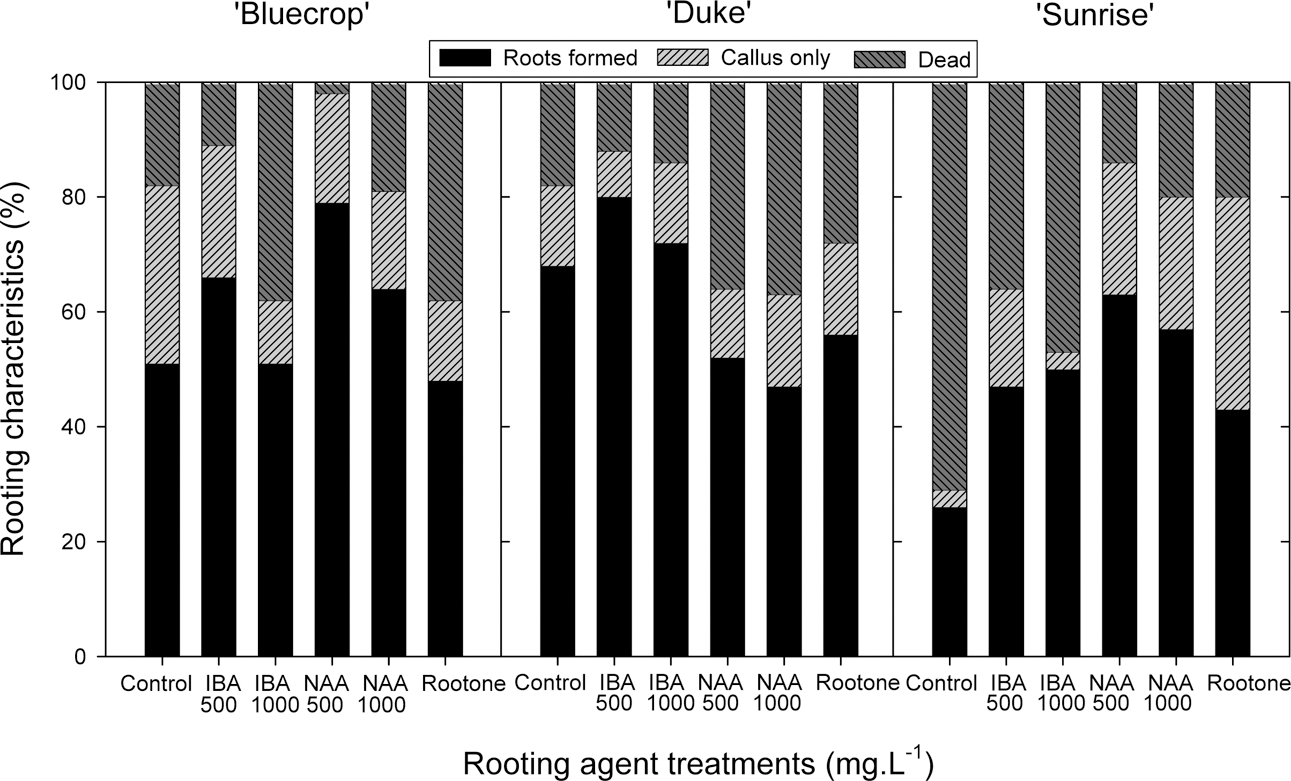
Thirty days after the cutting, the percent bud break was generally increased 6 to 14% by a quick-dip treatment into rooting agent solutions, depending on the type of agents (Fig. 1). For example, with ‘Duke’ variety, IBA at 500 mg · L−1 was most effective among treatments, increasing the bud break by about 25%. The rooting percentage, which was determined 90 days after the cutting, was significantly increased by 28 and 37% with 500 mg · L−1 NAA for ‘Bluecrop’ and ‘Sunrise’ varieties, respectively (Fig. 2). In ‘Duke’, IBA at 500 mg · L−1 increased the rooting rate by 12% compared to untreated control, but NAA at 500 and 1000 mg · L−1 appeared to retard the adventitious root formation. Meanwhile, a commercial rooting agent Rootone (0.4% NAD, USA) did not promote rooting in all varieties except ‘Sunrise’, as compared to untreated control. It was noticeable that lower rates of IBA and NAA were more effective in inducing root formation than higher rates (Fig. 2). In untreated control, the percent rooting failure of the cuttings was found to be much higher in ‘Sunrise’ (76%) than ‘Bluecrop’ (18%) and ‘Duke’ (22%). This unexpected value could be an outlier rather than being caused by a cultivar factor, since in Expt. 2 the percent rooting failure in ‘Sunrise’ was only 21% (Fig. 4) and it is known that blueberry cuttings are known to be relatively easy-to-root species. Strik et al. (1993) stated that the percent rooting of the blueberry cuttings ranges from 50 to 98% and the factors affecting rooting include varieties, cutting’s nutritional status, growing media, time of taking cuttings and PGR applications. This rooting failure appeared to be increased with increasing application rate, as seen in ‘Bluecrop’ and ‘Sunrise’, and interestingly in ‘Duke’ NAA treatments induced higher failure than did IBA, suggesting NAA may be toxic to the cuttings in that variety (Fig. 2).
The root and shoot growth of the cuttings was determined 90 days after the cutting, and the results showed that in ‘Duke’ IBA at 500 mg · L−1 significantly increased root weight, shoot length and leaf number (Table 1). In ‘Bluecrop’, root growth was not influenced by any of rooting agent treatments but a significant increase in shoot growth (shoot length and leaf number) was found only with NAA at 500 mg · L−1. In ‘Sunrise’, however, no rooting agent treatments influenced root and shoot growth of cuttings except 1000 mg · L−1 IBA causing a significant reduction in root weight (Table 1). In all varieties, the higher application rate appeared to reduce root development than did the lower rate, as more obviously seen in the variable of root weight. Copes and Mandel (2000) reported with Douglasfir that both NAA and IBA had broad ranges of rootenhancing activity, with the most effective rate occurring at 24.6 mM (4927 mg · L−1) for IBA and between 2.5 (465 mg · L−1) and 7.4 mM (1376 mg · L−1) for NAA, and also that rooting capacity decreased with both too little and too much auxin and there was no additional effect with any combination of IBA and NAA.
Table 1.
Effect of rooting agents on the root and shoot growth of hardwood cuttings of three highbush blueberry varieties, determined 90 days after the cutting.
We evaluated overall shoot growth of the transplanted cuttings into individual containers after 90 days of culture with Hyponex fertilizer. In ‘Bluecrop’ and ‘Sunrise’, rooting agent treatments generally increased shoot growth in terms of shoot branch number, branch length and diameter and leaf number (Table 2). Among them, NAA at 500 mg · L−1 was most effective in both varieties. In ‘Duke’, however, shoot growth except for branch number appeared to be slightly reduced by rooting agent treatments, as seen especially in NAA at 1000 mg · L−1 and Rootone.
Table 2.
Effect of rooting agents on the overall shoot growth of hardwood cuttings of three highbush blueberry varieties, determined after 90 days of further growth in individual pots fed Hyponex.
Overall shoot growth of the cuttings seemed to be positively related to the initial size of root development that was determined before transplanting rooted cuttings into individual containers (data not shown). Using the data pooled across all varieties and treatments, we correlated shoot growth parameters with initial root weight and found that there were significant relationships between root weight and shoot length (R2 = 0.48, P = 0.0015) and between root weight and leaf number (R2 = 0.35, P = 0.0098). However, parameters such as shoot branch number and branch diameter were little correlated with root weight. These results indicate that the establishment success of hardwood cuttings of highbush blueberry could depend on how the cuttings are taken care of during the initial rooting period.
The cutting success could be influenced by the treatment methods of rooting agents. There are two methods in large, the dilute solution soaking method and the quick-dip in concentrated solution (Hartmann et al., 2002). The dilute solution soaking method showed some success with hardto- root cuttings of Prunus, conifers, and deciduous shrubs when their basal portions were soaked for a maximum of 4 h at 50 to 150mg · L−1 (Macdonald, 1986). Eck and Childers (1989) reported that rooting efficacy on the softwood cuttings of blueberry was greatly improved by IBA at 50 mg · L−1 for 20 h, but its effect with hardwood cuttings was inconsistent. With hardwood cuttings of highbush blueberries, however, IBA soaking treatment for 24 h at 50 mg · L−1 reduced root and shoot development (Lee and Lee, 2009). They insisted that the use of rooting agents for hardwood cutting of blueberry may not be necessary. The second method ‘quick-dip into concentrated solution’ has been known to be effective in several woody species. Yoo and Kim (1996) reported that quick-dip treatments into auxin solutions promoted root formation on the softwood cuttings of white forsythia (Abeliophyllum distichum Nakai). With softwood cuttings of snowbell (Styrax japonica), rooting efficacy was increased up to 90% when the bases of cuttings were dipped in IBA solution at 7000 mg · L−1 (Kwon et al., 1997). Our study also showed a positive effect of the quick-dip treatment on the initial root development and subsequent shoot growth of hardwood cuttings of highbush blueberries when 1 cm-long, freshly cut bases of the cuttings were dipped for 5 s in IBA and NAA solutions both at 500 mg · L−1 (Tables 2 & 3).
Table 3.
Effect of shading treatments during root development on the root and shoot growth of hardwood cuttings of three highbush blueberry varieties, determined 90 days after the cutting, just before transplanting. The cutting bed was covered with black shade cloth of different shading levels.
| Shading (%) | Root length (cm, longest) | Root weight (g FW) | Shoot length (cm) | No. of leaves per cutting | SPAD values |
|---|---|---|---|---|---|
| ‘Bluecrop’ | |||||
| 0 | 6.2 | 0.20 | 6.0 | 6.2 | 35.8 |
| 30 | 6.5 | 0.21 | 6.8 | 6.5 | 36.5 |
| 50 | 6.3 | 0.18 | 5.9 | 6.3 | 32.4 |
| 70 | 6.0 | 0.14 | 6.2 | 5.8 | 33.6 |
| 90 | 4.5 | 0.09 | 4.5 | 4.2 | 33.2 |
| LSD (p =0.05) | 1.1 | 0.03 | 1.1 | 1.1 | 3.4 |
| ‘Duke’ | |||||
| 0 | 6.2 | 0.14 | 4.2 | 9.0 | 33.8 |
| 30 | 6.5 | 0.17 | 4.1 | 8.9 | 35.1 |
| 50 | 6.0 | 0.15 | 4.0 | 8.3 | 34.9 |
| 70 | 4.0 | 0.09 | 3.7 | 4.5 | 32.2 |
| 90 | 3.8 | 0.03 | 4.0 | 5.4 | 30.3 |
| LSD (p =0.05) | 1.0 | 0.02 | 0.8 | 1.4 | 3.3 |
| ‘Sunrise’ | |||||
| 0 | 5.3 | 0.23 | 4.1 | 6.5 | 35.7 |
| 30 | 5.7 | 0.24 | 5.9 | 6.7 | 36.2 |
| 50 | 3.0 | 0.19 | 3.4 | 6.5 | 37.3 |
| 70 | 4.8 | 0.21 | 4.0 | 6.4 | 32.4 |
| 90 | -z | - | - | - | - |
| LSD (p =0.05) | 1.1 | 0.05 | 1.0 | 1.6 | 4.5 |
2. Expt. 2: Effects of shading treatments on hardwood cuttings of highbush blueberries
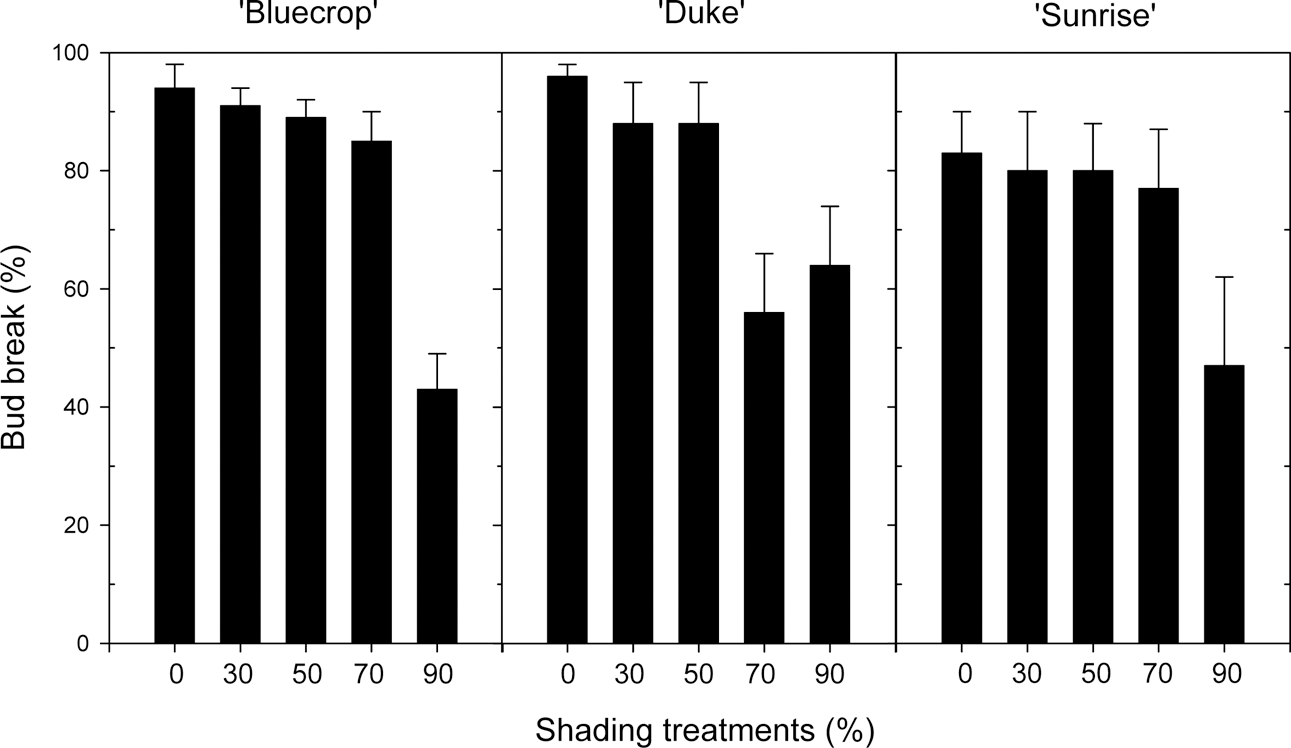
Shading treatments over the cutting bed reduced the temperature of rooting media, with its effect in proportion to the shading level (data not shown). Compared to non-shading control, shading treatments during June reduced the media temperature by 1.1 to 2.2°C around 9 a.m., by 3.3 to 4.8°C around 1 p.m., and by 2.6 to 3.6°C around 5 p.m., depending on the shading level. Thirty days after cutting, the percent bud break was decreased by shading treatments proportionally to shading levels (Fig. 3). For example, 70% shading reduced the bud break rate by more than 35%, and among 3 varieties the natural bud break rate was lowest with ‘Sunrise’.
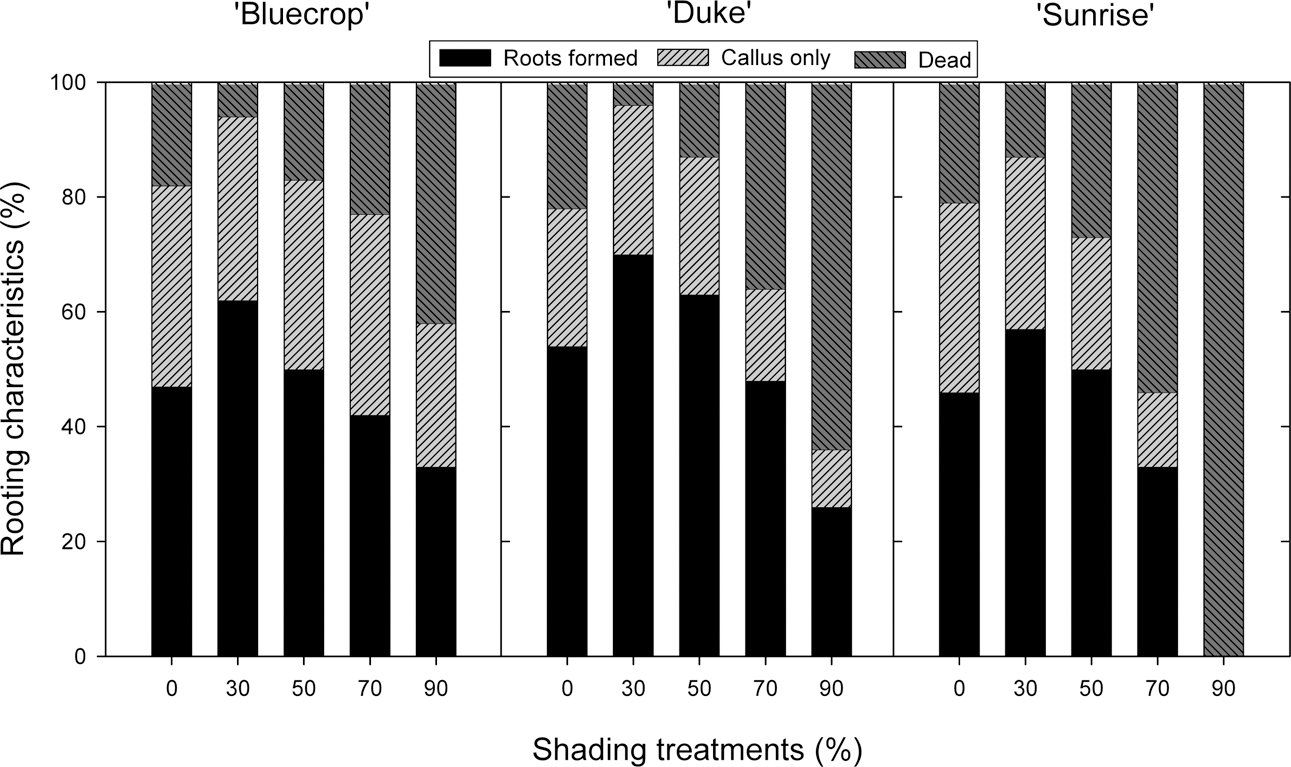
The rooting percentage, 30 days after the cutting, was slightly increased 11 to 16% depending on variety, when the cutting bed was covered with black shade cloth of 30% shading level (Fig. 4). At shading levels higher than 50% shading, however, rooting was substantially reduced: in ‘Sunrise’ 90% shading was too extreme for survival, killing all the cuttings.
Ninety days after cutting, root and shoot growth seemed to be increased by 30% shading and its effect was similar to that of 50% shading (Table 3). Shading levels higher than 50% would reduce the quality of cuttings when their root and shoot sizes were substantially decreased, as shown in ‘Duke’. Leaf chlorophyll content represented by the SPAD values tended to be decreased by shading treatments, with their effect seemingly being proportional to the shading level (Table 3).
The results of shoot growth of the cuttings after 90 days of further culture in individual pots showed that shoot growth was generally increased by 30% shading but a significant increase occurred only in ‘Sunrise’ for the variables of shoot branch number and length (Table 4). There was no significant difference between 30% and 50% shading treatments in their shoot growth performance. Shading treatments higher than 50% seemed to have an inhibitive effect on shoot growth, as clearly observed in ‘Duke. In ‘Sunrise’ 90% shading caused complete failure of the cutting.
Table 4.
Effect of shading treatments during root development on the overall shoot growth of hardwood cuttings of three highbush blueberry varieties, determined after 90 days of further growth in individual pots with Hyponex fertilizer. The cutting bed was covered with black shade cloth of different shading levels.
These results indicated that hardwood cuttings of blueberry may require some shading over the cutting bed to protect cuttings from desiccation stress that might occur during the initial rooting period. Actually we practiced the hardwood cutting on the cutting bed without a mist or fog system, so it was expected that the 30% shading gave some help in reducing water loss out of tissues of the cuttings during rooting, thus favoring the establishment success of the hardwood cuttings.
The rooting capacity of cuttings can vary according to the species as well as environmental conditions (Hartmann et al., 2002). Rooting of the cuttings of box trees (Buxus microphylla var. koreana Nakai) was improved when they were exposed to long days, but decreased with decreasing light levels (Kim et al., 1977). For blueberry plants, we observed that the establishment success of the hardwood cuttings was substantially decreased by shading of the cutting bed at the levels higher than 50% shading (Tables 3 & 4). Considering blueberry plants like full sun (Trehane, 2004), it is expected that light should not be restricted during the cutting period. However, hardwood cuttings on the cutting bed with no mist system as in this study would need the minimum amount of shading to minimize water stress from the cuttings especially during the initial period of cutting while avoiding light limitation.
In conclusion, the percent rooting and root growth of hardwood cuttings of highbush blueberries were improved by application of rooting agents. For ‘Bluecrop’ and ‘Sunrise’ varieties, NAA at 500 mg · L−1 was most effective while in ‘Duke’ it was with IBA at 500 mg · L−1. These treatments were found to work better than a commercial product ‘Rootone’. The 30% shading treatment cast over the cutting bed increased the percent rooting, and root and shoot growth, compared to no shading control, which was determined 90 days after the cutting. However, shading levels higher than 50% shading were found to be inhibitory for the establishment success of hardwood cuttings of highbush blueberries, especially under the environmental conditions with no mist system.