Introduction
Materials and Methods
1. Plant materials and growth conditions
2. Light treatment
3. Photosynthetic rate determination
4. Fruit yield measurement
5. Fruit quality analysis
6. Fruit antioxidant capacity analysis
7. Statistical analyses
Results and Discussion
1. Photosynthetic rate
2. Fruit yield
3. Fruit quality characteristic
Conclusions
Introduction
Greenhouse cultivation is a key strategy in climate-smart agriculture that facilitates highly efficient crop production under increasingly volatile environmental conditions. By shielding crops from external stressors and using controlled environmental systems, greenhouses achieve yields equal to or greater than those in open-field systems, while requiring up to 90% less land area (Gruda, 2005). This spatial efficiency significantly reduces the agricultural environmental footprint. Additionally, greenhouses serve as buffers against extreme weather conditions and climate-induced production instability, supporting cultivation via precise temperature, humidity, and photoperiod control (Soni et al., 2017).
Tomato (Solanum lycopersicum L.), a perennial plant commonly grown in greenhouses, has a year-round harvesting season. This provides diverse health benefits, such as reducing the risk of chronic diseases, improving skin conditions, supporting muscle function, and enhancing immunity (Collins et al., 2022). However, a significant decline in the daily light integral during winter shortens the harvest period and suppresses productivity at northern latitudes (Kubota et al., 2018; Palmitessa et al., 2020a).
Supplemental lighting (SL) is an essential technique for year-round tomato production as it compensates for insufficient solar radiation. Maintaining adequate photosynthetic photon flux density (PPFD) via SL is critical for sustaining photosynthetic activity and supporting fruit development under low ambient light conditions (Palmitessa et al., 2020b). Additionally, SL can effectively control the fruit yield based on the installation strategy. Verheul et al. (2022) reported consistently superior efficiency of top lighting (lamps positioned above the plant canopy) in improving overall plant growth and fruit yield compared with midlighting, which transmits light into the canopy layer. Furthermore, the spectral quality of SL strongly influences the physiological characteristics of crops (Li and Kubota, 2009). For example, blue light accelerates flowering and ethylene production in tomatoes, thereby promoting fruit ripening and the activity of antioxidants such as lycopene, phenolics, flavonoids, and vitamin C (He et al., 2022). In contrast, red light effectively promotes flowering and fruit enlargement (Li and Kubota, 2009). Commercial LEDs combine phosphors with 450 nm wavelength blue LEDs based on indium gallium nitride to produce a high-efficiency white LEDs spectrum with a constant wavelength peak (Pattison et al., 2018). However, reports analyzing plant responses to various peak wavelengths are limited (Johkan et al., 2012; Lee and Kim, 2014). Specifically, shifting the peak wavelength modulates photosystem II efficiency, thereby affecting growth and fruit yield (Nguyen et al., 2022). Therefore, the optimization of the SL spectral composition for specific tomato cultivars can significantly increase their productivity.
SL is indispensable for the enhanced and sustainable production of greenhouse tomatoes. However, there are limited reports on fruit yield and quality characteristics based on the quality of SL in short-day greenhouses. In our previous studies, specific full-spectrum light treatment improved growth and fruit yield in cherry tomatoes (Nguyen et al., 2022). In this study, we evaluated the effects of specific SL spectra on the photosynthetic rate, fruit yield, and quality of tomatoes grown in a greenhouse under top-lighting with a constant PPFD. This approach can contribute to sustainable horticultural practices by reducing climatic uncertainty.
Materials and Methods
1. Plant materials and growth conditions
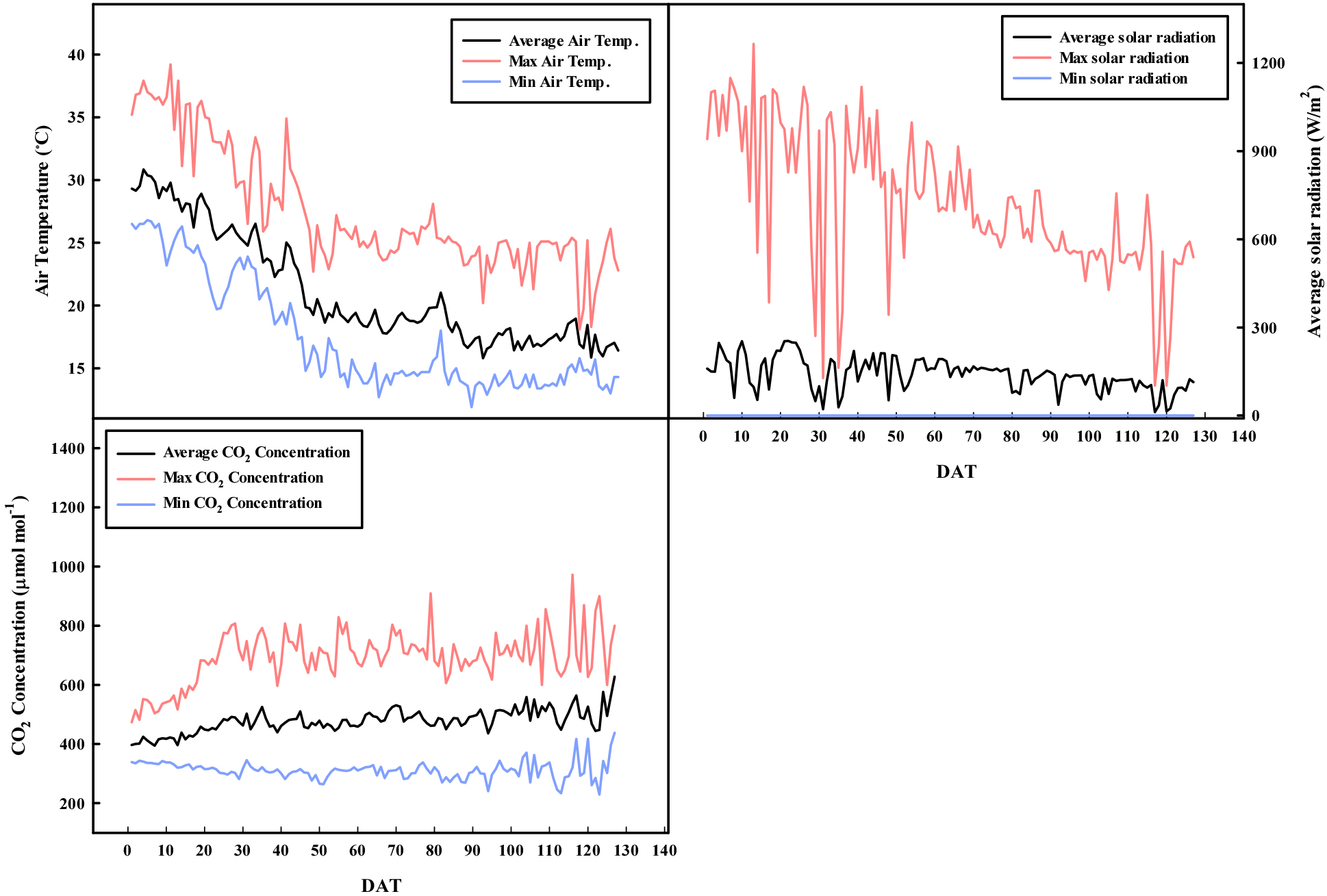
Tomato plants (cv. Pellinore (Enza Zaden, Enkhuizen, the Netherlands) were cultivated in this study. The seedlings were grown for 40 d, transplanted in a commercial greenhouse in Sacheon-si, Korea (35° 4’ 8.02’’N 127° 55’ 52.54’’E) on August 16, 2023, and harvested until December 21, 2023. The plants were cultivated in a coir medium (Coco peat; Duck Yang Coco, Anseong, Korea) and uniformly irrigated using a nutrient supply system (MAGMA-1000 V2.0; Green Control System, Damyang, Korea) with Yamazaki nutrient solution (pH 5.5; electrical conductivity: 3.2 ± 0.1 dS·m-1). Environmental parameters such as air temperature, CO2 concentration, and solar radiation were monitored using a greenhouse environmental control system (MAGMA-1000; Green Control System). The internal environment of the greenhouse during the growth period is shown in Fig. 1. Plants were cultivated using a one-stem system and managed twice weekly via picking, thinning, and defoliation. Plant height remained constant throughout the experimental period. Bumblebees were used to support fruit fertilization.
2. Light treatment
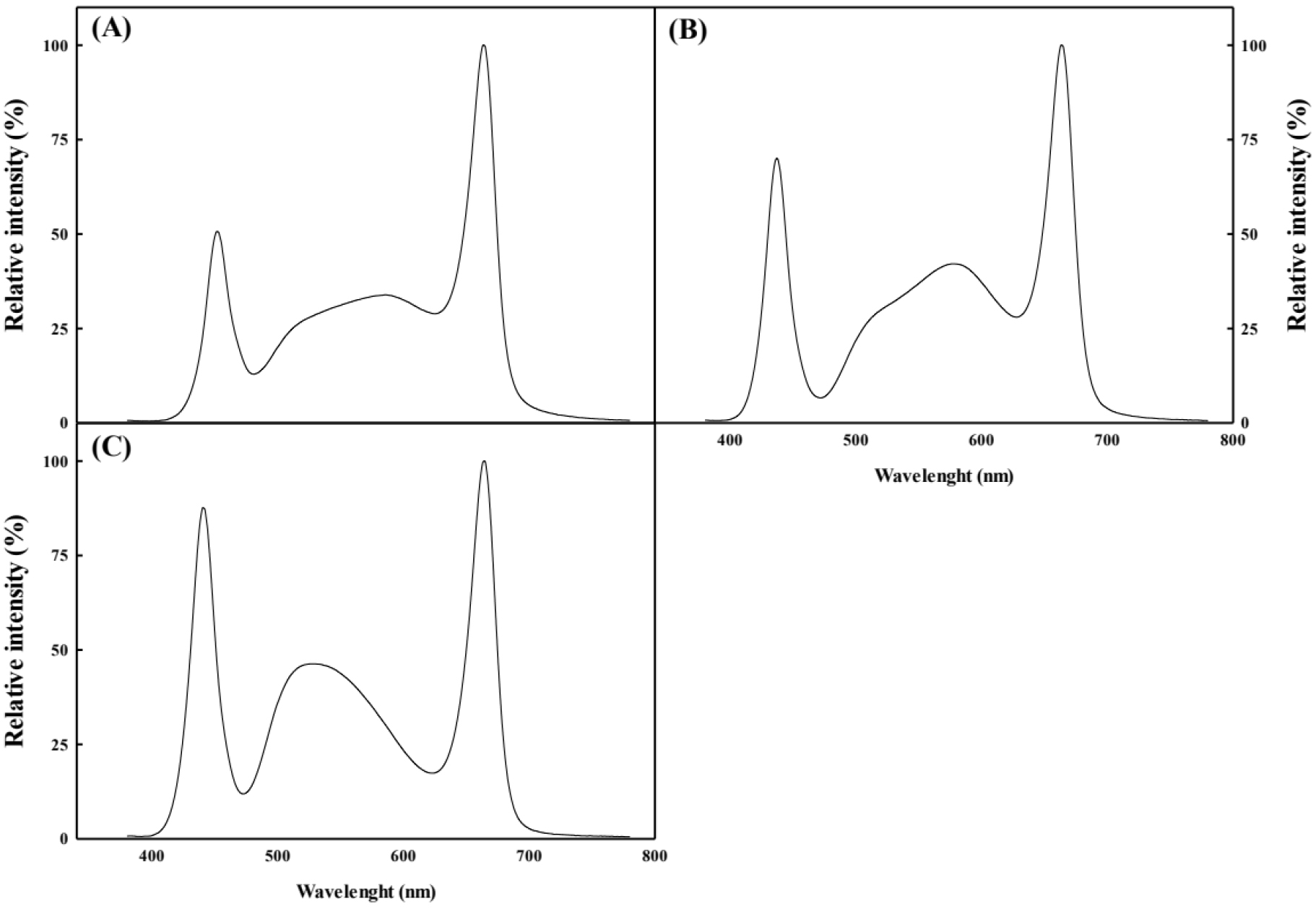
SL treatment was performed from October 13, 2023 (58 days after transplanting [DAT]) to December 21, 2023 (126 DAT). The control group was not treated with the SL. The treatment groups were as follows: full-spectrum LEDs (FSL), blue-specific full-spectrum LEDs (BSFL), and green-specific full-spectrum LEDs (GSFL) (Table 1; Fig. 2). Light intensity was measured at the apex of the plant body using a spectrum analyzer (LI-180 Spectrometer; LI-COR, Lincoln, NE, USA) and was 400 ± 25 µmol·m-2·s-1 under a 10/14 h photoperiod. The SL treatment was performed with the top lighting applied 2 m above the plants. To minimize uneven light distribution across groups, the plants were repositioned every three days using a “high wire” system to maintain consistent plant height and spacing.
Table 1.
Different supplemental lighting (SL) sources with distinct peak wavelengths and photosynthetic photon flux density (PPFD) ratios used in this study
| Type of LEDs light source | Peak (nm) | PPFD-ratio (%) | ||||
| Red | Green | Blue | Red | Green | Blue | |
| FSL | 663 | 585 | 450 | 47 | 34 | 19 |
| BSFL | 663 | 578 | 436 | 42 | 36 | 22 |
| GSFL | 663 | 526 | 436 | 34 | 38 | 28 |
3. Photosynthetic rate determination
Photosynthetic rate was measured at 111 DAT using a portable IR photosynthesis analyzer (LI-6400XT; LI-COR, NE, USA). The photosynthetic rate of fully expanded leaves from the first lower stem of the fifth cluster was measured on a sunny day between 9:00 and 11:30 am. Measurements were performed under the following conditions: CO2 at 500 ± 1.0 ppm, 2,000 ± 1.8 µmol photons·m-2·s-1, chamber temperature at 25.0 ± 0.5°C, relative humidity of 70 ± 1%, and a flow rate of 500 µmol·s-1.
4. Fruit yield measurement
Fruits harvested from October 26, 2023 (70 DAT), to December 21, 2023, were divided into clusters 1-6. Fruits were harvested at four-day intervals from each experimental group, and the fresh weight was immediately measured. Tomatoes were harvested from plants in the “breaker” stage (when yellow or pink color first appears, but not more than 10%) (Gude et al., 2020).
5. Fruit quality analysis
For fruit quality analysis, mean colorimeter (CR-20; Konica Minolta Inc., Tokyo, Japan) measurement values were used as follows: R = 40.8 ± 1.6, G = 23.2 ± 2.2, and B = 23.0 ± 1.6. Brix was measured using a Brix meter (PR-101a; Atago, Tokyo, Japan) by diluting 1 g of sample powder with 10 mL of distilled water. The lycopene content was determined using a lyophilizer (PVTFD20R; Ilshin, Dongducheon, Korea) by mixing 0.5 g of sample powder lyophilized at -70°C with 5 mL of ethyl alcohol and heating at 75°C. After adding 1 mL of 80% potassium hydroxide, it was heated again at 75°C. Then, hexane (2.5 mL) and high-performance liquid chromatography (HPLC) water were added, and the mixture was centrifuged (Centrifuge 5430; Eppendorf, Hamburg, Germany) at 3,000 rpm for 3 min. The supernatant was extracted and concentrated, and the resulting extract was dissolved in 2 mL of a mixture of dichloromethane and HPLC methanol (1:1, v/v) and filtered through a syringe filter (0.45 μL; Whatman Inc., Maidstone, UK). The filtrate was then used for HPLC measurements. A C-18 chromatographic column was used (Luna 5μ C18(2); 100 Å; 250 × 4.6 nm), the mobile phase was V (acetonitrile):V (methanol):V (tetrahydrofuran) = 70:25:5, the column temperature was 30 °C, flow rate was 1 mL·min-1, and injection volume was 20 μL. pH was measured using a calibrated pH meter (MP220, METTLER TOLEDO, London, UK). To determine the titratable acidity, 1 g of sample powder diluted with 50 mL of distilled water was titrated with 0.1 N NaOH until reaching a pH of 8.2 ± 0.1.
Acidity (%, lactic acid) = ([0.09·0.1·0.1 N NaOH vol {mL}·1.002]·[sample volume {mL}]-1)·100
where 1.002 is the coefficient of NaOH (0.1 N), 0.1 is the molarity of NaOH, and 0.09 is the lactic acid correction factor for the sample.
6. Fruit antioxidant capacity analysis
Total antioxidant activity was measured using the 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assay. ABTS⁺ radical solution was prepared by mixing 7 mM ABTS solution with 2.45 mM K2S2O8 and reacting in the dark for 14 ± 2 h. The solution was diluted with methanol to adjust the absorbance to 0.8-0.9, followed by the addition of 0.1 mL of sample extract to 0.9 mL of ABTS⁺ solution. The mixture was vortexed and incubated in the dark at room temperature for 3 min, and absorbance was measured at 732 nm. Finally, the ABTS radical scavenging activity was calculated using an ascorbic acid standard curve.
7. Statistical analyses
All data were analyzed using Duncan’s multiple-range test to separate the mean values (p < 0.05). Statistical analyses were conducted using SAS software (version 9.4; SAS Institute Inc., Cary, NC, USA), and all graphs were created using SigmaPlot 8.0 (Systat Software Inc., San Jose, CA, USA).
Results and Discussion
1. Photosynthetic rate
The photosynthetic rate of tomato leaves at 111 DAT was lowest in the control group without SL and highest in the BSFL group. Moreover, the photosynthetic rate of the BSFL group was 31% higher than that of the control group. The FSL and GSFL groups showed different results, although their wavelength peaks were different from those of the BSFL group. The photosynthetic rate of the GSFL group was not significantly different from that of the BSFL group. However, the photosynthetic rate in the FSL group was considerably lower than that in the BSFL group, but not significantly different from that in the control group (Fig. 3). Chlorophyll a, which is the primary photoactive pigment in photosystems I and II, most strongly absorbs light at 436 nm (Larkum, 2020). In this study, the blue peak wavelength of 436 nm significantly increased photosynthetic efficiency compared with FSL. This may have been due to the efficient excitation of the photosystem by providing an appropriate wavelength matching the absorption spectral peak of chlorophyll a, as well as the increased energy transfer efficiency resulting from the energy gradient (van Amerongen et al., 2000). In this study, SL did not significantly increase the photosynthetic rate in the FSL group. Irrigation was equivalent to insolation outside of the greenhouse. Liang et al. (2020) reported that decreasing soil moisture linearly decreased the photosynthetic rate of tomato plants. Therefore, SL may have increased the water requirement without providing additional irrigation to the coco peat blocks, thereby decreasing the photosynthetic efficiency of the tomatoes.
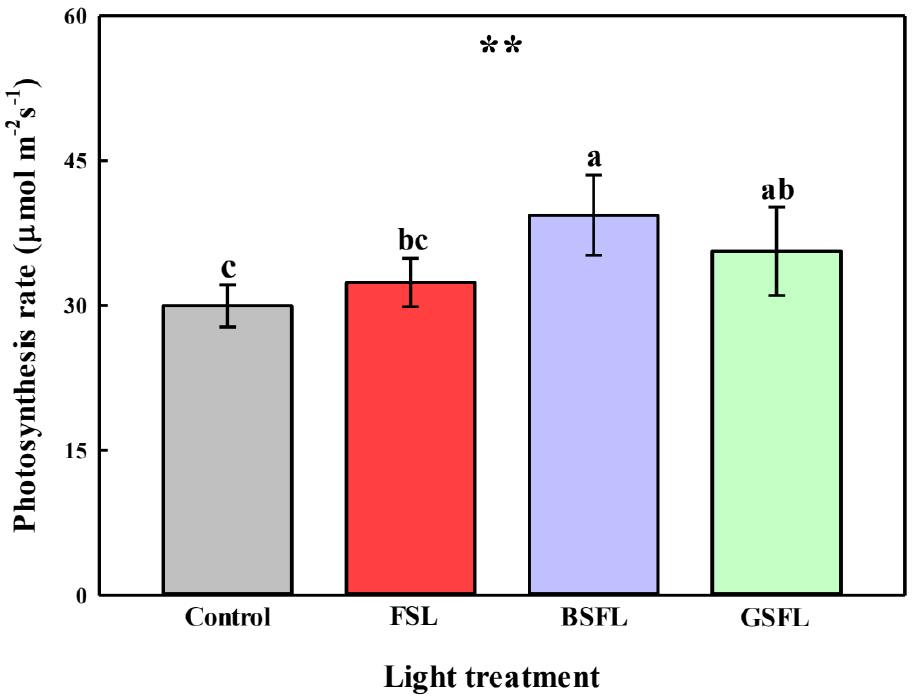
Fig. 3
Photosynthetic rates of tomato plants 111 days after transplanting (DAT) under different SL spectra. Abbreviations: FSL; Full-spectrum LEDs, BSFL; blue specific full-spectrum LEDs; and GSFL; green specific full-spectrum LEDs. Values are represented as the mean ± standard error (n = 4). **, p ≤ 0.01
2. Fruit yield
The total cumulative yields of tomato fruits harvested over 57 d were 12.1, 12.8, 14.7, and 14.5 kg in the control, FSL, BSFL, and GSFL groups, respectively. The control group exhibited the lowest tomato fruit yield, whereas the FSL, BSFL, and GSFL groups achieved 6, 22, and 20% higher tomato fruit yields, respectively, than the control group (Fig. 4). On the last day of the experiment, day 126 (12/21), BSFL and GSFL, with a peak wavelength of 436 nm, exhibited the highest fruit yields. The fruit yield results in this study were similar to the previously reported photosynthesis results (Fig. 3). For example, Nguyen et al. (2022) found that blue and green light with wavelengths of < 450 nm significantly increased the yield of tomatoes.
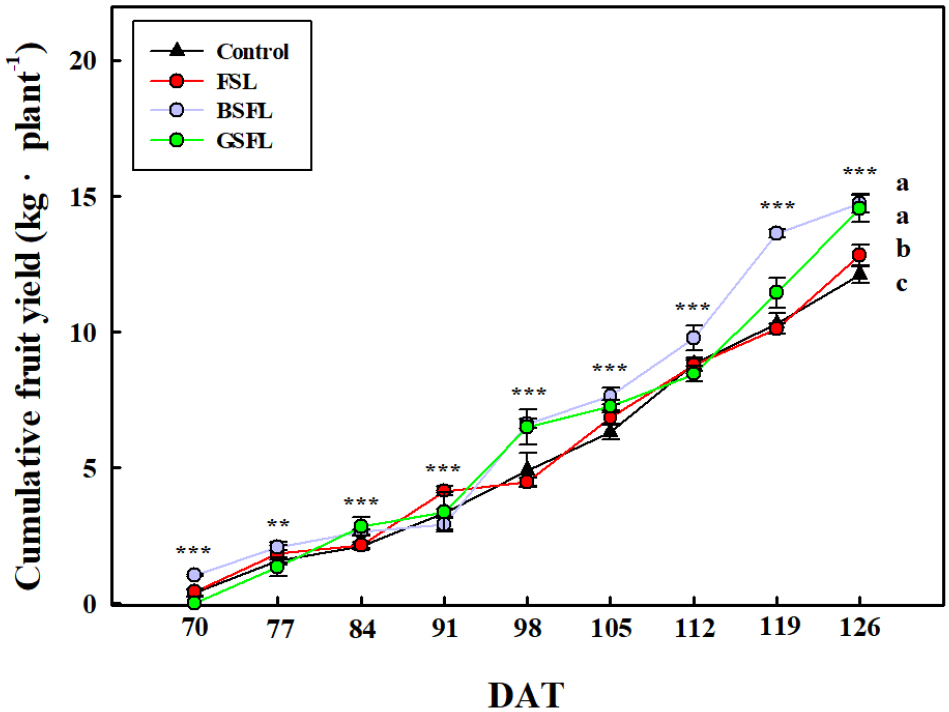
Fig. 4
Time course of cumulative fruit yield in tomato under different supplemental lighting (SL) treatments. Asterisks indicate significant differences compared with the control at each harvest time point (***, p ≤ 0.001; **, p ≤ 0.01). Letters (a, b, c) represent results of Duncan’s multiple range test (p < 0.05) among all treatments at each time point. Harvest dates correspond to 70-126 days after transplanting (DAT). FSL, full-spectrum LEDs (450 nm blue peak); BSFL, blue-specific LEDs (436 nm); GSFL, green-specific LEDs (526 nm); Control, no supplemental lighting. Values are mean ± SE (n = 4)
3. Fruit quality characteristic
Significant differences were observed in the fruit characteristics of tomatoes grown under different SL wavelengths (Fig. 5). The Brix content, which reflects the flavor of the agricultural product, increased by 3, 6, and 3% in the FSL, BSFL, and GSFL groups, respectively, compared with that in the control group, with the highest content observed in the BSFL group. The lycopene content was the highest in the control group, but decreased significantly under SL conditions. Specifically, the lycopene content was significantly higher in the BSFL and GSFL groups than in the FSL group. Moreover, the pH and acidity of the fruits exhibited opposite trends. The pH was significantly higher under SL conditions, with the GSFL group showing the highest pH. In contrast, acidity significantly decreased under SL conditions, being the lowest in the FSL and GSFL groups. The proper development of fruits and Brix content strongly depends on the photosynthetic products in the leaves (Baldet et al., 2006). In a study by Kläring and Krumbein. (2013), Brix composition was controlled in a manner similar to the photosynthetic rate. Additionally, in a study by Choi (2021), the soluble sugar content was positively correlated with photosynthesis. This increase in Brix was attributed to the improved availability of plant assimilates (Dorais et al., 2002), which is consistent with the results of this study (Fig. 5A). Lycopene is the main antioxidant in tomatoes. The control group exhibited the highest lycopene content, which was higher than that of the SL treatment groups (Fig. 5B). Lycopene accumulated more strongly as pH decreased (Bartkiene et al., 2013), and antioxidant capacity increased under low light intensity (Verkerke et al., 2015). The low lycopene levels in the control were possibly due to the accumulation of antioxidants under low light intensity during winter. Furthermore, at a high level of daily light integration, lycopene levels were the highest under blue and green light among the different light spectra (Fan et al., 2023), possibly because of the peak shift in wavelength (Fig. 5B). pH and acidity are important quality attributes in the secondary processing of tomato fruits, which contribute to food safety and flavor. The pH of tomatoes increases as they mature, and acidity decreases with an increase in pH (Anthon et al., 2011). In the present study, pH and acidity showed opposite trends, indicating that tomatoes grown under SL conditions exhibited sweet and mild flavors (Fig. 5 C and D). Tomato fruits contain antioxidant metabolites, such as vitamins, carotenoids, phenolic compounds, and phenolic acids, which provide adequate protection by neutralizing unstable free radicals (Raiola et al., 2014). A significant difference in tomato ABTS radical scavenging activity was observed, depending on the presence or absence of SL and light quality under a short-day photoperiod. The highest observed was in the control group, whereas the lowest was observed in the FSL group (Fig. 6). ABTS radical scavenging activity is a relative indicator of antioxidant compositions that can react with ABTS, as the steric accessibility rather than the chemical properties of the antioxidant controls the reaction (Schaich et al., 2015). Therefore, to obtain a more comprehensive evaluation of antioxidant activity, multiple complementary assays (ABTS, FRAP, ORAC, lipid peroxidation inhibition) should be employed (Sadeer et al., 2020). Blue light regulates the overall antioxidant levels in tomatoes by increasing the phenolic and flavonoid contents and stimulating the expression of regulators involved in the biosynthesis of lycopene, an important carotenoid, thereby increasing the lycopene content (He et al., 2022). In this study, the ABTS radical scavenging activity of tomatoes was similar to that of lycopene (Fig. 5B). This suggests that properties related to the antioxidant capacity of fruits can be modulated by shifting the wavelength toward the blue and green peaks.
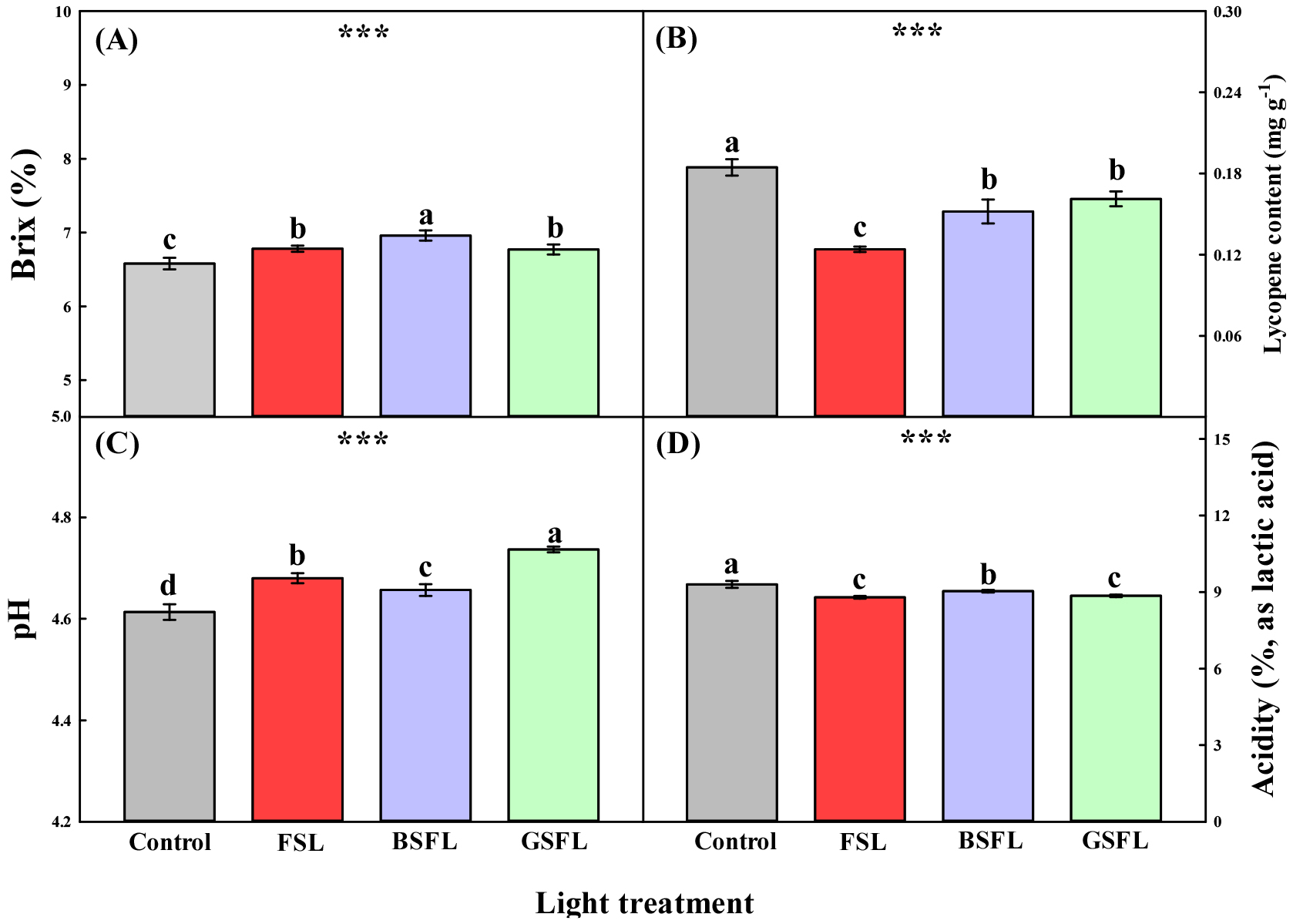
Fig. 5
Fruit quality parameters. Brix (A), Lycopene content (B), pH (C), and acidity (D) in the 5th harvested tomato cluster. Abbreviations: FSL; Full-spectrum LEDs, BSFL; blue specific full-spectrum LEDs; and GSFL; green specific full-spectrum LEDs. Values are represented as the mean ± standard error (n = 4). ***, p ≤ 0.001; NS, not significant
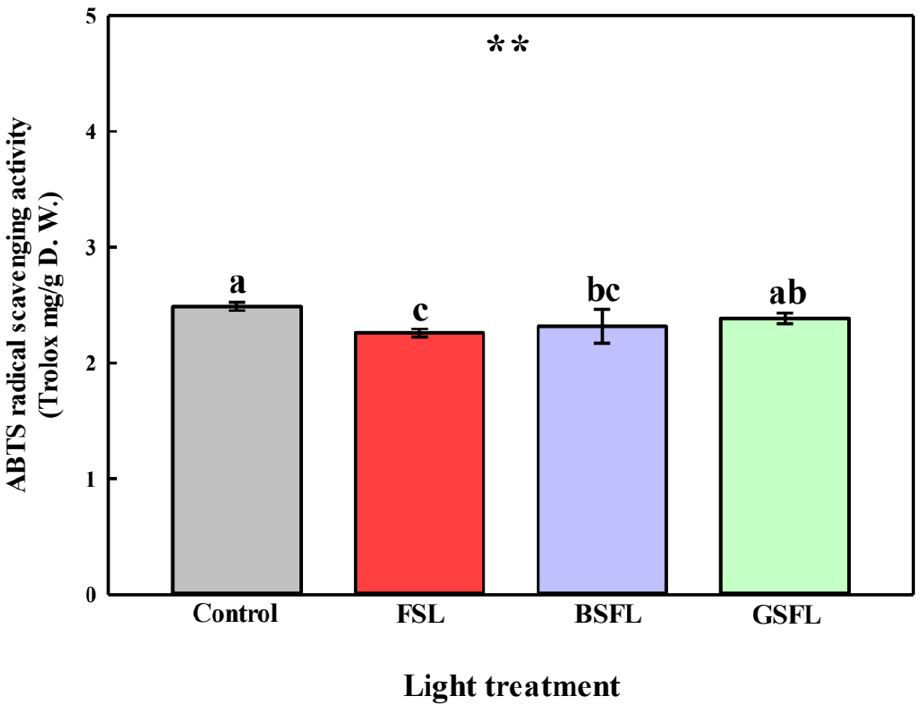
Fig. 6
2,2’-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical scavenging activity of the 5th harvested tomato cluster. Abbreviations: FSL; Full-spectrum LEDs, BSFL; blue specific full-spectrum LEDs; and GSFL; green specific full-spectrum LEDs. Values are represented as the mean ± standard error (n = 4). **, p ≤ 0.01
A limitation of this experiment was that the red photon fraction could not be maintained completely constant across treatment groups due to changes in the PPFD ratio that occurred when adjusting the spectrum of the phosphor-converted full-spectrum LEDs (Table 1). However, higher photosynthetic rates and yields were achieved with BSFLs and GSFLs, which had lower red ratios. Therefore, the present results should be interpreted as a combined effect of a shift in peak wavelength and a moderate shift in the color ratio in a realistic commercial spectrum, rather than an effect of a perfectly fixed red intensity peak shift.
Conclusions
In summary, this study demonstrates that spectral peak optimization of supplemental lighting (SL) significantly enhances tomato production under short-day greenhouse conditions. SL treatments increased photosynthetic rate, fruit yield, and Brix content compared with non-supplemented controls. Notably, BSFL and GSFL (featuring a blue light peak at 436 nm aligned with chlorophyll a absorption) achieved 22% and 20% yield increases, respectively, substantially outperforming standard full-spectrum LEDs (FSL, 450 nm) with only 6% increase. This demonstrates that small wavelength shifts can substantially impact crop performance. However, lycopene and ABTS radical scavenging activity were lower in the SL treatment and higher in the control, likely due to stress-induced phytochemical accumulation under low-light conditions. This trade-off underscores the need for tailored SL strategies aligned with specific production objectives. Overall, our findings highlight that spectral peak difference of SL, particularly aligning the blue peak with photosynthetic pigment absorption, is critical for optimizing greenhouse production. Future research should explore dynamic spectral management approaches that balance yield, quality, and energy efficiency for diverse production goals.