Introduction
Materials and Methods
1. Pod materials by artificial self-pollination
2. Seed media preparation by aseptic condition
3. Callus induction from protocorm-derived pseudobulbs explants
4. Effect of different natural extracts on callus proliferation and PLBs induction from callus culture
5. Effect of different plant growth regulators and natural extracts on shoot regeneration and multiplication of B. auricomum Lindl. from calli-derived PLBs culture
6. Statistical analysis
Results and Discussion
1. Morphology of flower and pod materials by artificial self-pollination
2. Effect of germination on culture medium and callus induction from subcultured pseudobulb explants
3. Effect of different natural extracts on callus proliferation and PLBs induction from callus culture
4. Effects of different plant growth regulators and natural extracts on shoot regeneration and multiplication of B. auricomum Lindl. from calli-derived PLBs culture
Conclusion
Abbreviations
Introduction
Orchidaceae is the second-largest angiosperms family with roughly around 28,484 species (Govaerts et al., 2017). The family makes up nearly 10% of the most beautiful flowering species of plants Tsai et al. (2013). Historically, ‘Orchid’ is derived from the Greek word, it means testicles, because some species of the Orchid genus are shaped in the form of subterranean tubers (Patil, 2007). Among the various orchid species, Bulbophyllum Thouars. is one of the largest genera in the Orchidaceae family with more than 2,200 species spread throughout the tropics, and the subtropics. Most of these are found in Asia (Vermeulen et al., 2014). The name Bulbophyllum is taken from the Greek word “bulbos” meaning bulb, and “phyllon” meaning leaf. This name refers to the plant’s pseudobulbs (Wood, 2005). The pseudobulb or thickened stem is of central importance to the growth of these orchids due to storage organs for food and water and functions such as a bulb (Ng and Hew, 2000).
Bulbophyllum auricomum Lindl. commonly known as the royal flower of Myanmar or Thazin-Pan in the native to Myanmar and is also found in Thailand, and the Indonesian islands Sumatra and Java. It is found in Bago, Tanintharyi, Yangon of Region, Mon and Rakhine States of Myanmar (Hundley et al., 1987). This orchid is the most attractive flower for Myanmar’s people. Because of B. auricomum Lindl. has an admirable fragrance, it is used as the national symbol of Myanmar and has long adorned women’s hair (Hinsley et al., 2017). It is an epiphytic, sympodial orchid and grows naturally on tree trunks covered with humus and mosses. The pseudobulb is about 8-10 cm long, ovoid- oblong, carrying one or two leaves at the top. This flower blooms once a year in the late fall and early winter. Its asexual reproduction through vegetative division is inefficient, as only one or two plants are produced per bulb per year and thus it is difficult to collect the required number of pseudobulbs in nature. Also, the germination rate of its seeds is very low (0.2-0.3%) because orchid seeds are small and devoid of the endosperm. Hence, seedlings take a long time to germinate in their native habitats and grow in the absence of suitable hosts (Than, 2013). B. auricomum Lindl. is an expensive and rare plant that blossoms once a year. Its flower has high commercial value in local markets because it is mostly used to decorate Burmese women on festival days. So, this orchid has become endangered due to over-collection and exploitation by traders.
The tissue culture technique can use for B. auricomum Lindl. and can produce many micropropagules in a short period of time that have almost the same properties as the parent and can save these endangered species from extinction in their natural habitat. Furthermore, epiphytic orchids have been developed and are being successfully cultured in MS (Murashige and Skoog) media (Paul et al., 2019). Activated charcoal (AC), plant growth regulators (PGRs), and organic growth supplements have also been shown to influence in vitro shoot regeneration, multiplication, and growth of orchid seedlings (Kim et al., 2019). Activated charcoal (AC) can adsorb excessive hormones, vitamins (Fridborg and Eriksson, 1975), and phenolic and carboxylic compounds (Fridborg et al., 1978) and is also used to stimulate the production of PLBs (Chen et al., 2002). Plant growth regulators (PGRs) such as auxins and cytokinins can stimulate the regulation of tissue proliferation and further development into plantlets (Arditti and Krikorian, 1996).
Various types of natural supplements including tomato juice, coconut water, banana extract, and potato extract are commonly added to orchid plant tissue culture media because they have natural vitamins, phenols, fiber, hormones, and proteins (Gnasekaran et al., 2009). Among them, three natural extracts (coconut water, banana, and potato extract) were used in the present investigation. Coconut water is the liquid endosperm of coconut and contains soluble sugars as a natural source of carbon, amino acids, phenols, fiber, and vitamins, and functions as a cytokinin that increases explant growth and regeneration by inducing cell division (Winarto et al., 2015). Banana has high nutritive value and high content of pyridoxine, vitamin B6, potassium, carbohydrates, and fiber (Dagnew et al., 2012). Potatoes are a proven source of quality proteins, carbohydrates, and minerals such as calcium, potassium, phosphorus, etc. (Casanas et al., 2002). This natural additive is known to promote the growth of orchids (Islam et al., 2000).
Previous researchers reported on in vitro propagation of B. auricomum Lindl. by using MS medium supplemented with various plant growth regulators collected from explants parts or seeds under natural self-pollination (Than, 2013; Thet and Aye, 2018). Another study conducted by (Khin et al., 2008) indicated a successful shoot multiplication of B. auricomum L. in VW (Vacin and Went, 1949) medium supplemented with natural extracts and plant growth regulators. However, there is no report on the effect of different combinations of natural extracts and plant growth regulators from seed germination under artificial self- pollination. Therefore, the objective of this study was to evaluate capsule or pod viability by artificial self-pollination; to prepare an MS basal culture medium for successful germination; to establish protocorms that continue developing and forming shoots from seed germination; to test callus induction by plant growth regulators (PGRs); and to determine the effects of different concentrations and combinations of natural extracts (coconut water, potato extract, and banana extract) on callus induction and PLBs formation in vitro and the effect of comparative studies under natural extracts with or without the plant growth regulators (PGRs) for plantlet shoot regeneration of B. auricomum Lindl.
Materials and Methods
1. Pod materials by artificial self-pollination
Mature flowers (5 days after flowering) of B. auricomum Lindl. were pollinated from mother plants with shade net greenhouse conditions. In artificial self-pollination, two pollinia were hand-pollinated by sterilized tooth sticks sprayed with 70% ethanol solution, and placed inside the stigma of the same flowers. The fertilized flower petals turn from white to yellowish after one week and the petals gradually become dry. After growing for four months, the mature pods after artificial self-pollination were collected from the Useful Plant Resource Center, Dagon University, Myanmar. For in vitro seed culture, the four-month-old mature seed of B. auricomum L. was carried out in the tissue culture laboratory, Department of Lifestyle Medicine, Jeonbuk National University, Republic of Korea.
2. Seed media preparation by aseptic condition
The collected pods were firstly washed under running water followed by surface sterilization through soaking in a 25% cocorex solution for 15 minutes and were transferred to a clean bench. The sterilized pods were then immersed in 70% ethanol solution for 10 minutes and then rinsed three times using autoclaved distilled water. The sterilized pods were dissected longitudinally with a sterile surgical blade to obtain the seeds. The sterilized seeds were cultured on MS (Murashige and Skoog, 1962) basal medium without plant growth regulators to study seed germination and seedling growth. All MS basal media used 30 g·L-1 of sucrose and 8 g·L-1 of agar. The pH value of the media was adjusted to 5.8 by adding a drop of 1N NaOH or 1N HCL before autoclaving at 121°C for 15 minutes. Culture media were incubated in a culture room at 25 ± 2°C under 16/8 (light /dark light condition) with the cool-fluorescent white light intensity of 3,000 lux.
3. Callus induction from protocorm-derived pseudobulbs explants
After seed cultured of three months, the protocorms from in vitro seedlings were successfully induced and sub-cultured on MS medium supplemented with different combinations of plant growth regulators (PGRs), such as 2.0 mg·L-1of 1-napathylacetic acid (NAA), 1.0 mg·L-1 of 6-benzyl aminopurine (BAP) and the addition of 1g·L-1 of activated charcoal (AC) for roots and shoots with pseudobulb explants.
4. Effect of different natural extracts on callus proliferation and PLBs induction from callus culture
After culturing of nine weeks, calli were obtained from the pseudobulb size (1.2 cm) explant of B. auricomum Lindl. in the above medium. To gather a large amount of callus, (0.23 g) of calli were sub-cultured by 11 different concentrations and combination treatments of natural extracts such as coconut water (CW), banana extract (BE), and potato extract (PE) were added to the MS basal medium with 1 g·L-1 of activated charcoal as shown in Table 1. The coconut (young coconut milk was finely mixed with coconut water using a blender), banana, and potato were weighed and blended using an electric blender and then added directly to the culture media before adjusting pH.
Table 1.
Different natural extracts tested for the callus subculture.
| Culture medium composition |
| 1. MS+ (100 mL·L-1) CW† |
| 2. MS+ (150 mL·L-1) CW |
| 3. MS+ (20 g·L-1) BE†† |
| 4. MS+ (30 g·L-1) BE |
| 5. MS+ (20 g·L-1) PE††† |
| 6. MS+ (30 g·L-1) PE |
| 7. MS + (20 g·L-1) BE + (20 g·L-1) PE |
| 8. MS + (20 g·L-1) BE + (30 g·L-1) PE |
| 9. MS + (30 g·L-1) BE + (20 g·L-1) PE |
| 10. MS + (30 g·L-1) BE + (30 g·L-1) PE |
| 11. MS†††† (control) |
5. Effect of different plant growth regulators and natural extracts on shoot regeneration and multiplication of B. auricomum Lindl. from calli-derived PLBs culture
In each subculture, callus and PLBs mass (0.23 g) were sub-cultured in MS medium supplemented with natural extract alone and a combination of natural extracts with plant growth regulators for plant regeneration. Natural extracts were used in the best medium MS with (150 mL·L-1) coconut water and (30 g·L-1) banana extract along with (20 g·L-1) potato extract from the best result data. In other natural extracts with plant growth regulators medium was MS supplemented with (150 mL·L-1) coconut water along with (2.0 mg·L-1) BAP and (1.0 mg·L-1) NAA; (30 g·L-1) banana extract and (20 g·L-1) potato extract in combination with (2.0 mg·L-1) BAP and (1.0 mg·L-1) NAA.
6. Statistical analysis
For in vitro propagation of callus, PLBs, and shoot culture, each experiment was repeated at least three times. Results were represented as means ± standard deviation (SD). The data of mean values were analyzed by two-way analysis of variance (ANOVA) using Sidark’s multiple comparison tests was performed to obtain the value of significance between the groups, where values with *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001 were considered statistically significance and ‘ns’ depicted non-significance set at the 0.05. All the statistical analyses were carried out through GraphPad Prism (version 9.3.0).
Results and Discussion
1. Morphology of flower and pod materials by artificial self-pollination
Flowers generally occur from December to February each year. The raceme flower with peduncle is slightly arched from the base of young pseudobulbs. Flowers are small, thinly textured, white color and life span. Three months after artificial self-pollination, the flowers successfully turned into mature pods as shown in Fig. 1. The mature pods are ovate in shape, dark green color, and ridges appeared. The pods of B. auricomum L. contained thousands of tiny white seeds. These results indicated that artificial self-pollination is necessary to attain better mature capsules.
2. Effect of germination on culture medium and callus induction from subcultured pseudobulb explants
Orchid seeds are devoid of functional endosperm and are unable to grow on their own (Rasmussen et al., 2015). Hence, seeds from the mature pod of B. auricomum Lindl. were cultured in MS basal medium without plant growth regulators (PGRs) for seed germination. After one month of seed culture, the formation of well-developed light green protocorm and eventually plantlets with the shoot and root differentiation occurred within two to three months. Data on seed germination were recorded for up to three months. Fig. 2 (a, b and c) shows that seeds germinated to a high degree in the MS medium. These results are in agreement with those using MS medium in the seed germination of several Coelogyne spp. such as C. flaccida Lindl. (Kaur and Bhutani, 2014). Subsequently, three months old developed plantlets of B. auricomum Lindl. were transferred to the MS medium supplemented with plant growth regulators, such as 2.0 mg·L-1 of NAA, 1.0 mg·L-1 of BAP, and 1 g·L-1 of AC were also added. After 2 months of growth, these PLBs developed into pseudobulb explants (data not shown). The growth of callus appeared friable in texture and yellowish to green in color at the base of pseudobulb explants (Fig. 2d). Several systems have been reported to be useful in callus induction and PLB formation through cytokinin and auxin combined effects including Dioscorea zingiberensis (Vidya et al., 2005) and Clerodendrum serratum (Naika and Krishna, 2008). Callus induction was initiated using the MS medium supplemented with NAA and BAP, which showed that high concentrations of cytokinin induced greenish and greater callus (Ashrafuzzaman et al., 2009). The callus induction in the MS supplemented with NAA and BAP culture medium in this study agrees with the references.

Fig. 2.
Seed germination and protocorm to plantlets development in B. auricomum Lindl.; a) swelling and enlargement of seed after one week of culture; b) seed germination followed the formation of protocorm after 1 month of culturing; c) shoots started differentiation from protocorm after 3 months of culturing; d) callus induction from subcultured pseudobulb plantlets under light conditions in 2 months of culturing.
3. Effect of different natural extracts on callus proliferation and PLBs induction from callus culture
Pseudobulb-derived callus masses (about 0.23 g) were sub-cultured in basal MS medium supplemented with 11 different treatments of natural extracts. Among them, callus and PLBs were observed in several media, but MS medium augmented with coconut water (150 mL·L-1) and banana extract (30 g·L-1) along with potato extract (20 g·L-1) obtained a large amount of callus and PLBs induction. The results presented in MS medium fortified with coconut water (150 mL·L-1) produced the highest callus fresh weight of 1.75 ± 0.08 and 3.01 ± 0.20 at 1 and 2 months, respectively (Fig. 3: 2a, 2b). The second highest fresh weight at 1 month (0.67 ± 0.09) and 2 months (1.87 ± 0.05) of callus was obtained by MS supplemented with (30 g·L-1) of banana extract along with (20 g·L-1) of potato extract (Fig. 3: 9a, 9b). Tefera and Wannakrairoj (2004) who stated that coconut water in tissue culture media enhanced callus initiation and shoot development. The effects of the banana extract on new PLBs regeneration in Dendrobium orchid were reported by (Aktar et al., 2008). The addition of potato extract can selectively enhance PLB proliferation due to the production of essential amino acids (Obsuwan and Thepsithar, 2014). The optimal concentration of coconut water at 150 mL/L has been applied to stimulate PLB development (Rohmah and Taratima, 2022). However, our results showed that MS medium with coconut water (150 mL·L-1) had significant effects on callus induction and the development of protocorm-like bodies (PLBs). Therefore, the present research is in agreement with previous reference reports.
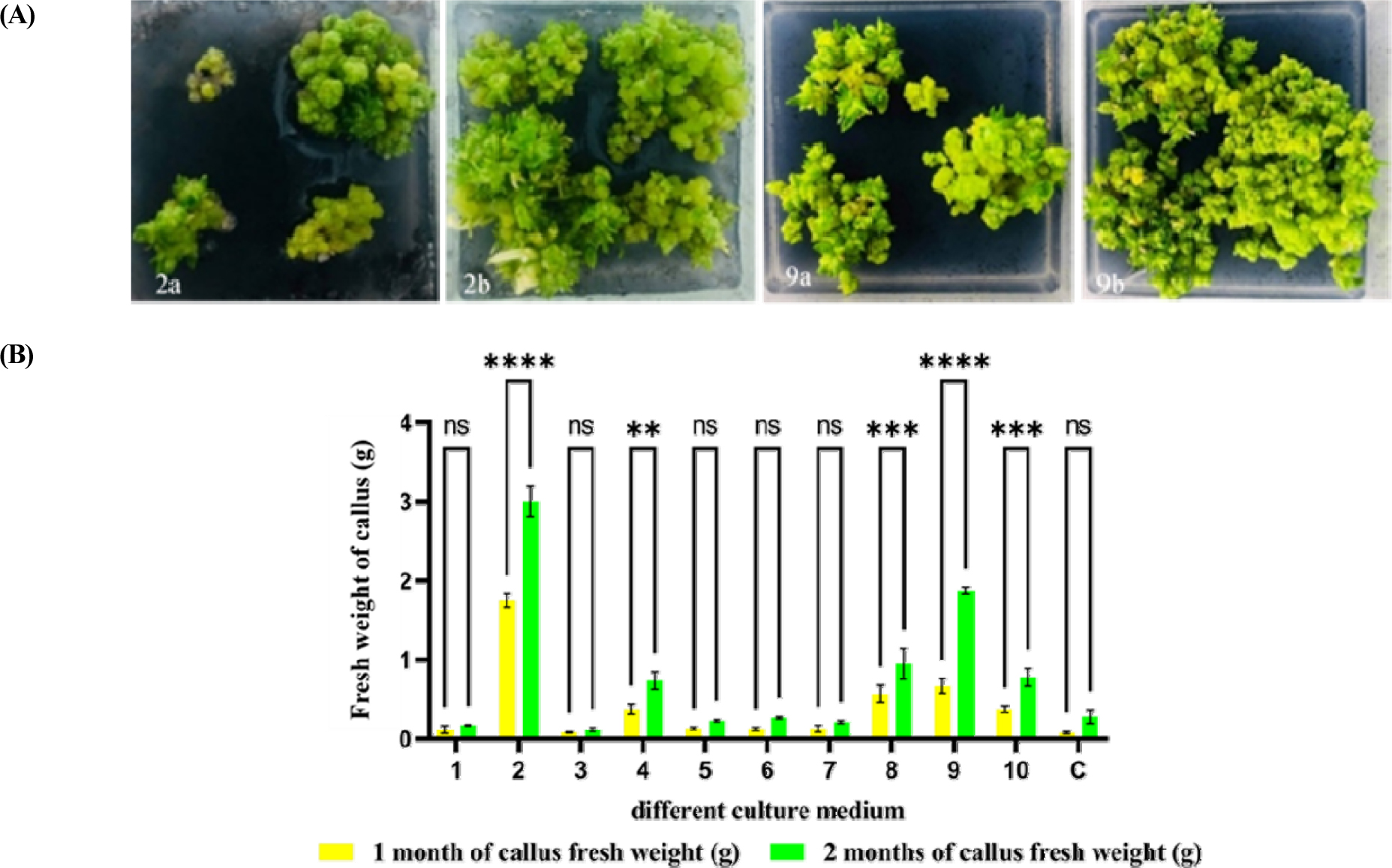
Fig. 3.
Effects of different combinations of natural extracts on callus proliferation and PLBs induction of fresh weight of B. auricomum Lindl. after 1 month and 2 months of culture. (A) Callus proliferation and PLBs induction effected a representative picture from 2a, 2b) MS +150 mL·L-1 of coconut water at 1 and 2 months of culture; 9a, 9b) MS + 30 g·L-1 of banana + 20 g·L-1 of potato extract, respectively. (B) Data of fresh weight are shown as mean ± SD of the value obtained from triplicate experiments. 1) MS + 100 mL·L-1 of CW†; 2) MS + 150 mL·L-1 of CW; 3) MS + 20 g·L-1 of BE††; 4) MS + 30 g·L-1 of BE ; 5) MS + 20 g·L-1 of PE†††; 6) MS + 30 g·L-1 of PE; 7) MS + 20 g·L-1 of BE + 20 g·L-1 of PE; 8) MS + 20 g·L-1 of BE + 30 g·L-1 of PE; 9) MS + 30 g·L-1 of BE + 20 g·L-1 of PE; 10) MS + 30 g·L-1 of BE + 30 g·L-1 of PE; C) MS†††† (control). The p-values of *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001 were considered significant.†CW: Coconut water; ††BE: Banana extract; †††PE: Potato extract; ††††MS: Murashige and Skoog.
4. Effects of different plant growth regulators and natural extracts on shoot regeneration and multiplication of B. auricomum Lindl. from calli-derived PLBs culture
Consequently, 2-month-old callus and PLBs were carried out to compare the effects of natural extracts and combinations of natural extracts with plant growth regulars for regeneration. Our results in Fig. 4 showed that the maximum plantlet regeneration was observed in MS medium supplemented with 150 mL·L-1 of coconut water and 2.0 mg·L-1 of BAP with 1.0 mg·L-1 of NAA by (3.37 ± 0.17) in 1 month and (6.41 ± 0.68) in 2 months, followed by MS medium fortified with 50 mL·L-1 of coconut water alone and so on. The MS basal medium (control) produced the minimum shoot regeneration. However, natural extract in combination with plant growth regulators (PGRs) was better than natural extract alone for producing a higher quantity and quality of plantlets of B. auricomum Lindl. Hormones such as auxins, cytokinins, and natural substances, are important factors that can be used to support and improve protocorm to orchid plantlets (Parthibhan et al., 2015). Furthermore, their types and concentrations of hormones and natural substances depend on the effects of shoot development (Prasertsirivatna and Koolpluksee, 2011). The synergistic effect of high levels of cytokinin along with low levels of auxins was reported to facilitate shoot proliferation (Faisal et al., 2018). Nayak et al., 1998 and Sheelavantmath et al., 2000 who found the enhancement of plantlet growth with the application of BAP and NAA. The present investigation found that the above characteristics agree with previous studies.
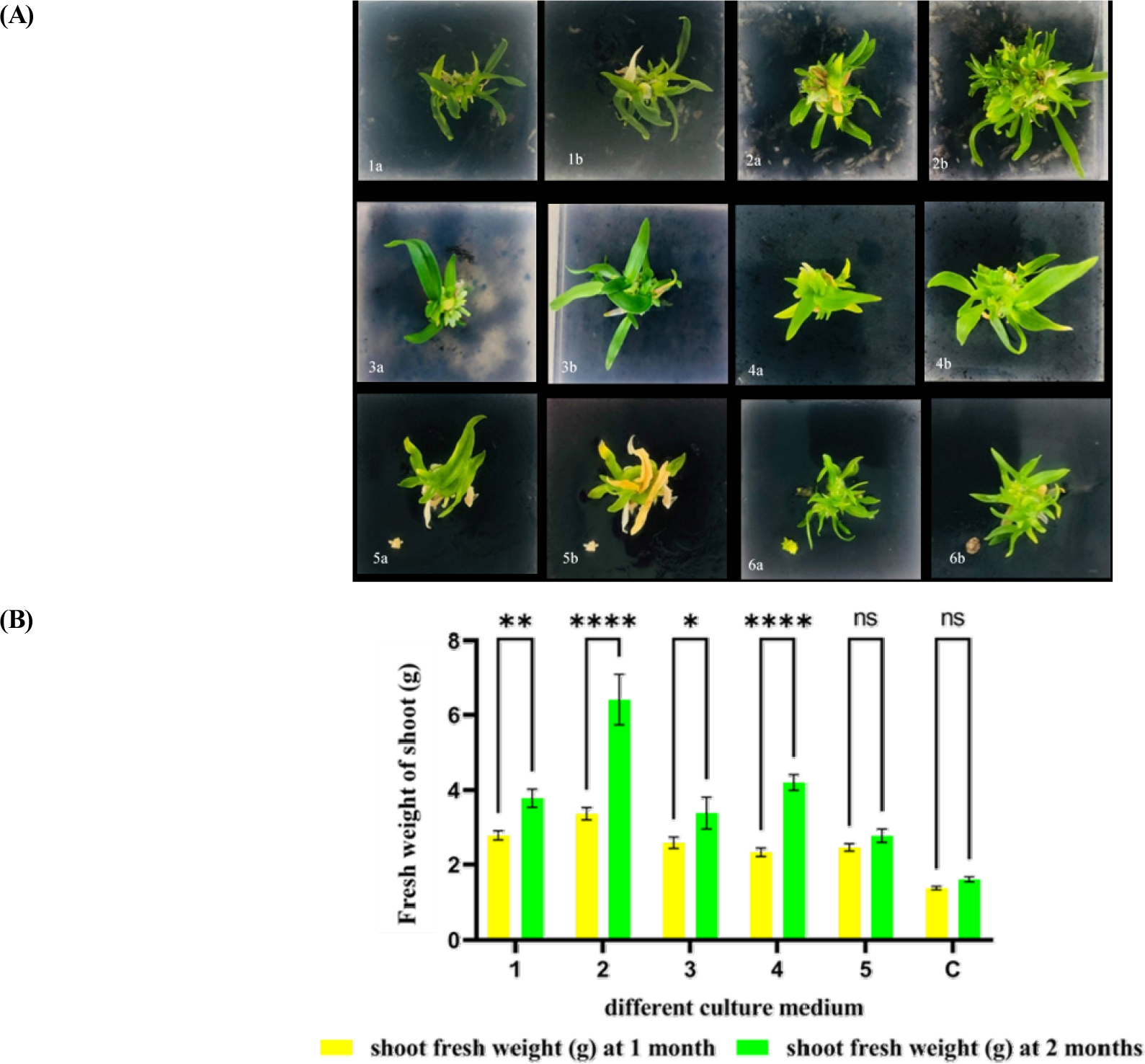
Fig. 4.
Effects of different combinations of plant growth regulators and natural extracts on shoot regeneration of fresh weight of Bulbophyllum auricomum Lindl. after 1 month and 2 months of culture. (A) The pictures exhibited a representative shoot regeneration of each medium. 1a, 1b) MS +150 mL·L-1 of CW†; 2a, 2b) MS +150 mL·L-1 of CW + 2.0 mg·L-1 of BAP†† + 1.0 mg·L-1 of NAA†††; 3a, 3b) MS + 30 g·L-1 of BE†††† + 20 g·L-1 of PE†††††; 4a, 4b) MS + 30 g·L-1 of BE + 20 g·L-1 of PE + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA; 5a, 5b) MS + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA; 6a, 6b) MS†††††† basal control after 1 and 2 months of culturing respectively. (B) Fresh weight of shoot regenerations was measured from callus-derived PLBs formation. 1) MS +150 mL·L-1 of CW; 2) MS +150 mL·L-1 of CW + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA; 3) MS + 30 g·L-1 of BE + 20 g·L-1 of PE; 4) MS + 30 g·L-1 of BE + 20 g·L-1 of PE + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA; 5) MS + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA; C) MS basal control. Data are shown as mean ± SD of the value obtained from triplicate experiments. The p-values of *p < 0.05, **p < 0.01, ***p < 0.001, and ****p <0.0001 were considered significant.†CW: Coconut water; ††††BE: Banana extract; †††††PE: Potato extract; ††BAP: 6-benzyl aminopurine; †††NAA: 1-napathyl acetic acid; ††††††MS: Murashige and Skoog.
Conclusion
In this study, it was found that the endangered B. auricomum Lindl. pods were regularly obtained through artificial self- pollination and successful seed germination was enhanced by MS basal medium. The combination of plant growth regulators (1.0 mg·L-1/L of BAP and 2.0 mg·L-1/L of NAA) on the MS basal media has been established callus induction from subcultured pseudobulb explants. Furthermore, this callus was sub-cultured by supplementation with 11 different natural extracts for callus-derived PLBs. The present study of combination of natural extracts and plant growth regulators showed that the most effective way to proliferate multiple shoot induction and plantlets growth were in (MS + 150 mL·L-1 of coconut water + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA), followed by (MS + 30 g·L-1 of banana extract + 20 g·L-1 of potato extract + 2.0 mg·L-1 of BAP + 1.0 mg·L-1 of NAA) from callus-derived PLBs. Therefore, combinations of natural extracts and plant growth regulators may be useful for micropropagation and endangered Bulbophyllum auricomum Lindl. This information can be applied to assist the propagation of endangered orchids.





