Introduction
Materials and Methods
1. Planting material and Experimental design
2. Crop management conditions
3. Measured growth parameters
4. Nutrient solution composition
5. Inorganic nutrient analysis
6. Statistical analysis
Results and Discussion
1. Growth characteristics affected by nitrate availability
2. Biomass accumulation
3. Chlorophyll content and leaf area reduction
4. Mineral nutrient accumulation affected by nitrate deficiency
Introduction
Perilla frutescens, commonly referred to as perilla, is a traditional leafy vegetable widely cultivated across East Asia. In recent years, it has attracted growing interest for its distinctive aroma and richness in bioactive compounds, enhancing its value as a functional food (Lee et al., 2020; Lim et al., 2022). Perilla leaves are notably abundant in polyphenols, including flavonoids, rosmarinic acid, and anthocyanins—phytochemicals recognized for their antioxidant, anti-inflammatory, and anticancer properties (Kim et al., 2019; Seo et al., 2021). As a result, the demand for premium-quality perilla leaves has expanded in both domestic and international markets, prompting the need for advanced cultivation techniques that support year-round production.
Traditional soil-based cultivation is frequently limited by environmental variability, pest outbreaks, and soil-borne diseases, which negatively affect both yield and product quality. In contrast, hydroponic systems enable precise control over environmental and nutritional parameters, thereby improving crop uniformity, reducing disease incidence, and enhancing production stability (Bugbee, 2004; Kim et al., 2003). Additionally, advancements in smart farming technologies have further strengthened hydroponic practices by allowing real-time monitoring and automated control of growing conditions to optimize the accumulation of health-beneficial phytochemicals (Kozai et al., 2019; Son et al., 2020).
In hydroponic cultivation, the composition and concentration of mineral nutrients in the nutrient solution are key determinants of plant development, yield, and secondary metabolite biosynthesis (Taiz et al., 2015; Yamazaki, 1982). Nutrient deficiencies or imbalances can disrupt physiological processes, impair growth, and ultimately reduce productivity (Bennett, 1993; Bould et al., 1983; Choi and Park, 2007a,b; Kim et al., 2025; Nelson, 2003). Among the macronutrients, nitrogen (N) is particularly essential for vegetative growth, as it is a core constituent of chlorophyll, amino acids, and nucleic acids (Kant et al., 2011). In soilless systems, nitrogen is mainly provided in the forms of nitrate (NO3-N) and ammonium (NH4-N), with nitrate being the more favorable source due to its greater stability and efficient assimilation under aerobic conditions, whereas excessive ammonium can lead to toxicity and disrupt ionic balance.
While the influence of nitrate availability has been well documented in leafy vegetables such as lettuce, spinach, and kale, limited information exists regarding its effects on perilla, a species with distinct growth patterns and nutritional needs. Relying on generalized hydroponic nutrient formulas may lead to suboptimal growth or unintended physiological stress in perilla (Park et al., 2019). Therefore, a targeted evaluation of perilla’s response to nitrate deficiency is necessary to develop nutrient management strategies that are specifically adapted to its cultivation.
This study was designed to investigate the effects of nitrate-nitrogen deficiency on the growth performance and physiological responses of hydroponically grown perilla. In addition to confirming the essential role of nitrate-nitrogen, this study helps define the threshold concentrations at which perilla growth becomes deficient, adequate, or critically limited. Identifying these levels is important for both maximizing yield and minimizing excessive nitrate accumulation in edible tissues, which has implications for human health.
Materials and Methods
1. Planting material and Experimental design
The experiment was conducted to assess the physiological responses of leaf perilla (Perilla frutescens cv. ‘Namcheon’) to different nitrate-nitrogen levels under hydroponic cultivation. Seeds were sown on February 24, 2023, into 200-cell plug trays filled with a commercial nursery substrate (Baroker; Nongwoo Bio, Korea). Following germination, uniform seedlings of two weeks age exhibiting at least two fully expanded true leaves were selected. The roots of the selected seedlings were gently washed to remove any residual substrate before transplanting. Each seedling was inserted into a cubical hydroponic sponge with a central hole and transferred to a simplified deep flow technique (DFT) hydroponic system (Higreen Hydroponic Machine B; Gawhatech, Korea). The experiment was conducted in a 40 m2 Venlo-type glass greenhouse located at Pusan National University, Miryang, Korea (35.233°N, 128.881°E), where environmental conditions were consistently maintained throughout the cultivation period. A completely randomized design (CRD) was used, with four plants per treatment and three replications (n = 12 per treatment). The cultivation period lasted for 80 days after transplanting (DAT).
2. Crop management conditions
Plants were cultivated in a simplified deep flow technique (DFT) hydroponic system, with each unit containing 10 L of nutrient solution. The nutrient solution was replaced every 14 days during the initial 40 days of cultivation and subsequently replenished on a weekly basis. Greenhouse air temperature was regulated at 20 ± 1°C using an automated climate control system to ensure stable environmental conditions. To suppress flowering and maintain vegetative growth, 200 W metal halide lamps (Osram HCI-T; Germany) were installed 2 m above the plant canopy at 2 m spacing intervals and operated continuously from sunset to sunrise. Lateral branches were pruned once or twice per week to encourage the dominance of the main stem.
3. Measured growth parameters
Lateral branches were pruned once or twice per week to encourage the dominance of the main stem. Plant growth parameters, including plant height, number of leaves, number of nodes, and stem diameter, were measured at 20, 30, 50, 60, and 80 days after transplanting (DAT). Chlorophyll content measurements began at 30 DAT. For biomass evaluation, three plants per treatment were randomly selected at both 60 and 80 DAT to determine fresh and dry weights. At 80 DAT, accumulated leaf area per plant was also assessed. The number of leaves was recorded by counting those exceeding 3 cm in length, and total leaf area was measured using a leaf area meter (LI-3000; LI-COR, USA). Stem diameter was measured 1 cm above the shoot base with a digital vernier caliper (DC150P; Bluetec, China). Fresh weight was measured immediately after harvesting, while dry weight was determined after drying plant samples in a forced-air oven at 70°C for 72 hours. Chlorophyll content was measured using a SPAD meter (SPAD-502; Minolta Co., Ltd., Japan), with three readings per leaf averaged for analysis. The number of nodes was determined by counting the leaf-bearing nodes with leaves longer than 3 cm, starting from the shoot base.
4. Nutrient solution composition
A modified Yamazaki nutrient solution, formulated for leafy vegetables, was used as the standard (control) solution in this experiment. The macronutrient concentrations in the control solution were (me·L-1): NO3-N, 6.0; NH4-N, 0.5; PO4-P, 1.5; K, 6.0; Ca, 2.0; Mg, 1.0; and SO4-S, 4.0. Micronutrient concentrations were maintained at consistent levels across all treatments (mg·L-1): Fe, 3.0; BO3, 0.5; Mn, 0.5; Zn, 0.05; Cu, 0.05; and MoO4, 0.001.
To investigate the effects of nitrate-nitrogen deficiency, five treatments were established by adjusting the NO3-N concentration in the standard solution: control (0% deficiency, 6.0 me·L-1), 30% deficiency (4.2 me·L-1), 50% (3.0 me·L-1), 70% (1.8 me·L-1), and 100% (0.0 me·L-1). All other macronutrient and micronutrient concentrations were kept constant among treatments. The electrical conductivity (EC) of each nutrient solution was adjusted to 1.0 dS·m-1, and the pH was maintained at approximately 6.0 throughout the cultivation period. Both EC and pH were measured at the time of solution preparation, which was conducted weekly.
5. Inorganic nutrient analysis
To evaluate the effects of nitrate-nitrogen deficiency on nutrient uptake, the aerial parts of the plants were analyzed for total nitrogen (T-N), phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg) content. Dried plant tissues were initially ground using a Wiley mill (Model 4, Thomas Scientific, Swedesboro, NJ, USA) to pass through a 1 mm mesh screen, followed by fine grinding with a cyclone impact mill (Model 3010-014, UDY Corporation, Fort Collins, CO, USA) to ensure sample homogeneity. For T-N analysis, 0.3 g of ground tissue was digested with concentrated sulfuric acid (H2SO, ≥95%, Sigma-Aldrich, St. Louis, MO, USA), and nitrogen content was determined using a semi- automatic Kjeldahl nitrogen analyzer (Pro-Nitro S, Model 4002851, J.P. Selecta, Barcelona, Spain). Phosphorus concentration was measured using the molybdenum blue colorimetric method following acid digestion. Potassium levels were determined via atomic absorption spectrophotometry (AA-7000, Shimadzu Corporation, Kyoto, Japan), while calcium and magnesium concentrations were analyzed using EDTA complexometric titration. Each nutrient analysis was performed in triplicate to ensure data reliability and precision.
6. Statistical analysis
Statistical analysis was conducted using SAS software (version 9.4; SAS Institute Inc., Cary, NC, USA). Treatment effects were evaluated using Duncan’s Multiple Range Test (DMRT) at a significance level of p < 0.05.
Results and Discussion
1. Growth characteristics affected by nitrate availability
Nitrogen (N) is a vital macronutrient involved in the synthesis of amino acids, nucleic acids, chlorophyll, and various enzymes, thereby playing a central role in plant growth and development (Marschner, 1995). Among the different forms of nitrogen available to plants, nitrate- nitrogen (NO3-N) is the primary form absorbed and assimilated by most higher plants. It directly affects cellular division and tissue development, contributing significantly to overall plant vigor (Epstein and Bloom, 2005). In this study, the impact of nitrate-nitrogen deficiency on the growth and physiological responses of hydroponically grown leaf perilla (Perilla frutescens) was assessed using a modified Yamazaki nutrient solution as the base formulation.
After 80 days of cultivation under varying levels of NO3-N deficiency, shoot growth exhibited a marked decline in response to reduced nitrate availability (Table 1). Plant height in the control group (0% deficiency) reached 73.5 cm, whereas it decreased to 37.3 cm under complete nitrate deprivation (100% deficiency), corresponding to a 49.3% reduction. Comparable reductions were observed in other shoot-related parameters: the number of nodes decreased by 41.7% (from 10.8 to 6.3), stem diameter by 42.2% (from 10.9 mm to 6.3 mm), and number of leaves also showed a significant decline. These growth reductions can be attributed to inhibited amino acid and protein synthesis, decreased chlorophyll content, and impaired cell division under nitrogen-deficient conditions (Wang et al., 2023).
Table 1.
Effects of nitrate-nitrogen deficiency on plant height, number of leaves, number of nodes and stem diameter in hydroponically grown of ‘Namcheon’ perilla.
| NO3-N deficiencyz (%) |
Plant height (cm) | No. of leaves | No. of nodes |
Stem diameter (mm) |
| 20 days after treatment | ||||
| 0y | 7.9 ax | 5.2 ab | 2.6 ab | 3.5 a |
| 30 | 9.2 a | 5.8 a | 2.9 a | 3.5 a |
| 50 | 7.7 a | 4.8 b | 2.4 b | 3.5 a |
| 70 | 9.3 a | 5.7 ab | 2.8 ab | 3.5 a |
| 100 | 8.2 a | 5.3 ab | 2.7 ab | 3.6 a |
| 30 days after treatment | ||||
| 0 | 23.4 a | 7.7 a | 4.8 a | 7.0 a |
| 30 | 24.6 a | 7.8 a | 4.9 a | 7.6 a |
| 50 | 24.0 a | 7.5 a | 4.8 a | 6.8 a |
| 70 | 22.1 ab | 7.2 a | 4.6 a | 7.0 a |
| 100 | 16.3 b | 5.5 b | 3.8 b | 4.7 b |
| 50 days after treatment | ||||
| 0 | 36.6 a | 6.5 ab | 7.3 a | 8.5 a |
| 30 | 36.1 a | 7.0 a | 6.5 b | 8.7 a |
| 50 | 33.8 a | 6.7 a | 6.3 b | 7.9 b |
| 70 | 35.3 a | 6.7 a | 6.3 b | 7.7 b |
| 100 | 8.9 b | 4.0 b | 2.3 c | 5.7 c |
| 60 days after treatment | ||||
| 0 | 56.2 a | 10.8 a | 8.2 a | 9.6 a |
| 30 | 54.0 a | 9.9 b | 8.0 a | 9.9 a |
| 50 | 55.3 a | 9.0 c | 7.3 ab | 8.9 b |
| 70 | 48.9 a | 8.5 c | 6.8 b | 8.7 b |
| 100 | 30.9 b | 6.7 d | 5.2 c | 5.8 c |
| 80 days after treatment | ||||
| 0 | 73.5 a | 12.8 a | 10.8 a | 10.9 a |
| 30 | 66.1 a | 11.7 ab | 10.8 a | 9.8 ab |
| 50 | 59.2 ab | 10.2 ab | 9.5 a | 9.1 b |
| 70 | 58.5 ab | 9.3 b | 9.4 a | 8.9 b |
| 100 | 37.3 c | 5.2 c | 6.3 b | 6.3 c |
These results are in agreement with previous findings by Choi and Park (2007b), who reported that nitrogen deficiency in perilla led to suppressed shoot elongation and accelerated chlorosis in older leaves. Similar inhibitory effects on shoot growth due to nitrogen deficiency have also been documented in other crops, including eggplant (Teixeira et al., 2009) and oriental melon (Won et al., 2023).
2. Biomass accumulation
Nitrate deficiency resulted in a significant reduction in biomass production in leaf perilla (Table 2, Fig. 5). Shoot fresh weight of control group averaged 93.4 g, whereas a sharp decline to 15.5 g under complete nitrate deprivation (100% NO3-N deficiency) was recorded, indicating an 83.4% decrease. Similarly, leaf fresh weight dropped from 62.4 g in the control to 8.0 g, corresponding to an 87.2% reduction. Dry weight accumulation exhibited a comparable pattern, with all nitrate-deficient treatments showing statistically significant declines (p < 0.05). These reductions in biomass are likely associated not only with impaired photosynthetic performance but also with suppressed cell division and expansion, reduced protein and enzyme synthesis, and altered assimilate partitioning under nitrogen-limited conditions (Lawlor, 2002). Based on the observed dry weight data, the critical nitrate-nitrogen concentration for perilla can be identified as the lowest level at which plant dry mass remains within 90% of the maximum (control) value. In this study, 3.0 me·L-1 NO₃-N (50% deficiency) resulted in approximately 90% of the maximum dry weight, suggesting this is the critical threshold. Levels below this are considered deficient, while 4.2-6.0 me·L-1 are adequate for optimal growth.
Table 2.
Effects of nitrate-nitrogen deficiency on fresh weight and dry weight in hydroponically grown of ‘Namcheon’ perilla.
| NO3-N deficiencyz (%) | Fresh weight (g/plant) | Dry weight (g/plant) | ||||
| Leaves | Stem | Total | Leaves | Stem | Total | |
| 60 days after treatment | ||||||
| 0y | 60.3 ax | 25.8 a | 86.1 a | 5.0 a | 3.7 a | 8.7 a |
| 30 | 51.5 ab | 25.3 a | 76.8 a | 4.6 a | 3.8 a | 8.4 a |
| 50 | 39.7 bc | 16.2 ab | 55.8 b | 4.5 a | 3.8 a | 8.3 a |
| 70 | 36.1 c | 13.5 ab | 49.6 b | 3.1 b | 3.3 a | 6.4 b |
| 100 | 13.3 d | 6.2 b | 19.4 c | 1.1 c | 1.8 b | 2.9 c |
| 80 days after treatment | ||||||
| 0 | 62.4 a | 31.1 a | 93.4 a | 7.0 a | 4.8 a | 11.8 a |
| 30 | 56.4 b | 31.5 a | 87.9 a | 5.5 a | 4.7 a | 10.2 ab |
| 50 | 44.5 c | 19.8 ab | 64.3 b | 5.8 a | 4.6 a | 10.3 ab |
| 70 | 39.0 c | 14.2 bc | 53.2 b | 3.4 ab | 4.5 a | 7.9 c |
| 100 | 8.0 d | 7.4 c | 15.5 c | 0.7 b | 2.2 b | 3.0 d |
3. Chlorophyll content and leaf area reduction
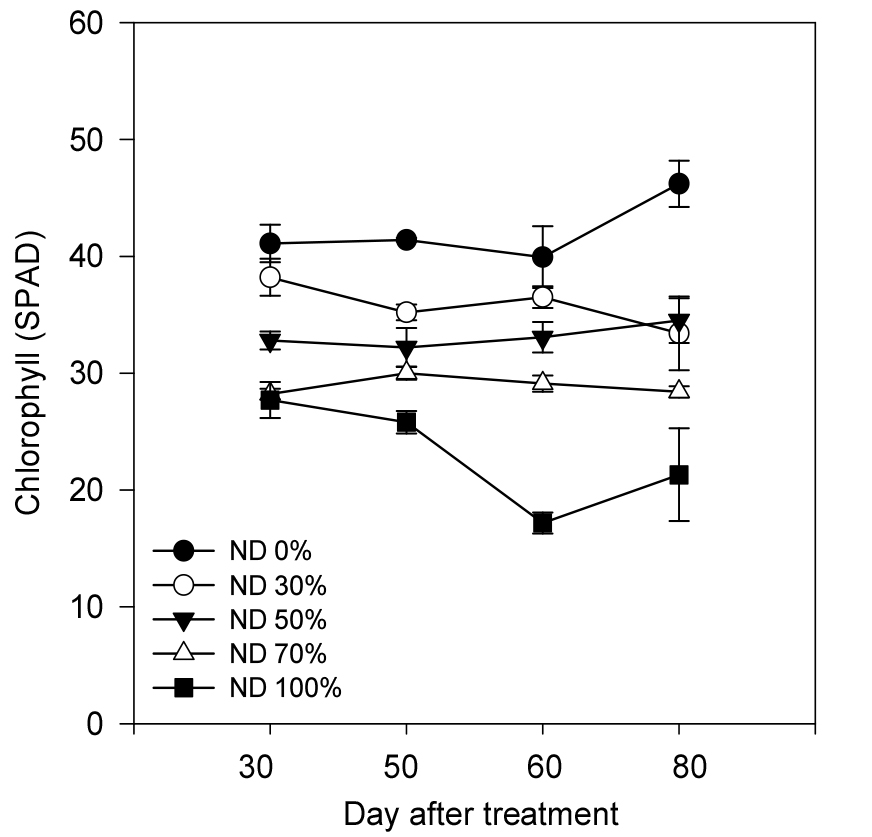
Nitrate-nitrogen deficiency significantly affected chlorophyll content and leaf area in leaf perilla. SPAD measurements revealed a marked decline in chlorophyll content with increasing nitrate deficiency (Fig. 1). Control plants exhibited an average SPAD value of 46.2, which decreased to 21.3 under complete nitrate deprivation (100% NO3-N deficiency), representing a 53.9% reduction. This decline was evident from early growth stages and became more pronounced with higher deficiency levels. Given that nitrogen is a core component of the chlorophyll molecule particularly in stabilizing the Mg2+ ion and is essential for chlorophyll biosynthesis, nitrate deficiency limits photosynthetic efficiency (RDA, 2021). In addition to reduced chlorophyll content, nitrogen deficiency impairs the synthesis of key photosynthetic proteins such as Rubisco and components of the electron transport chain, thereby contributing to reduced SPAD values and diminished net photosynthetic rates. Similar reductions in SPAD values under nitrogen stress have been observed in lettuce (Christin et al., 2015).
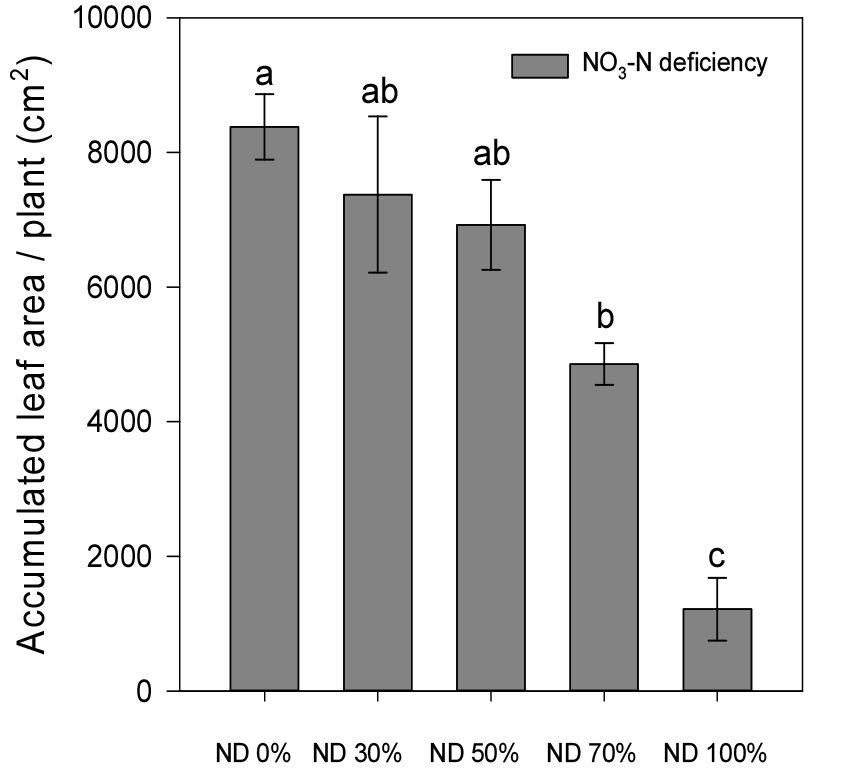
Leaf area also decreased substantially in response to nitrate deficiency (Fig. 2). While control plants produced a total leaf area of 8,986.4 cm2, this was reduced to 1,477.6 cm2 under 100% nitrate deficiency which is 83.6% decline. This loss of photosynthetically active surface area, resulting from fewer and smaller leaves, likely contributed to diminished carbon assimilation and overall biomass accumulation (Peng et al., 2020).
Overall, nitrate-nitrogen availability proved critical for maintaining growth and physiological functioning in hydroponically grown perilla. Under complete nitrate deprivation, plants achieved only 10-20% of the growth observed in control treatments (Fig. 3), underscoring the importance of sufficient nitrate supply from early developmental stages to ensure optimal growth and productivity in soilless systems.
4. Mineral nutrient accumulation affected by nitrate deficiency
In hydroponic systems, nitrate-nitrogen serves not only as a key macronutrient but also plays an important regulatory role in the uptake and distribution of other essential mineral elements. Optimal nitrogen fertilization enhances nutrient assimilation and supports physiological processes related to ion regulation in pepper plants (Baghour et al., 2000).
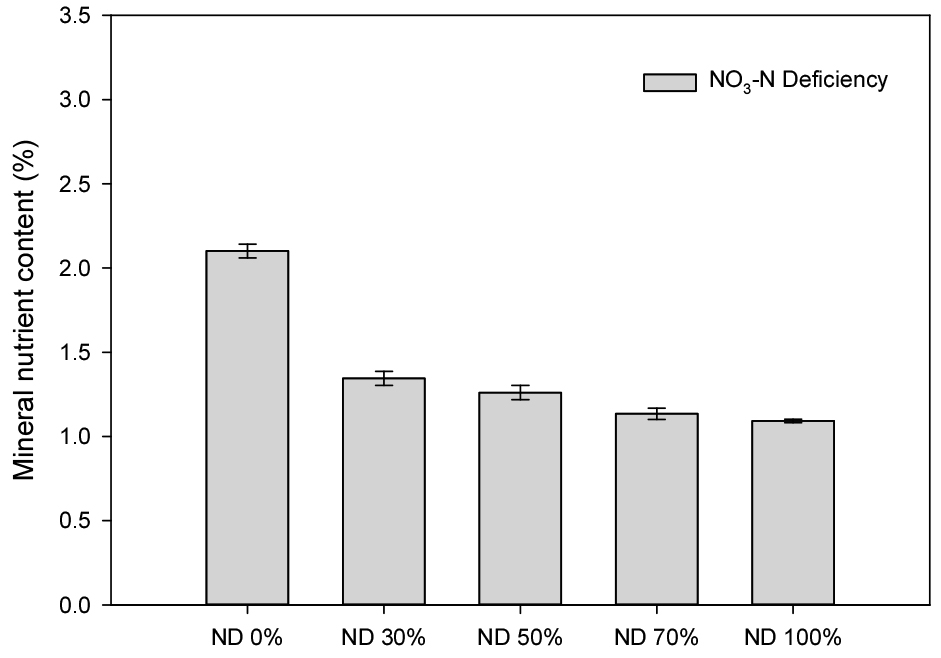
The concentrations of major mineral nutrients in perilla shoots after 80 days of cultivation under varying nitrate levels are presented (Table 3, Fig. 4). In the control treatment, shoot concentrations were as follows: total nitrogen (T-N), 2.82%; P2O5, 1.78%; K2O, 2.64%; CaO, 2.08%; and MgO, 0.63%. As nitrate deficiency increased, total nitrogen content declined proportionally, reaching 1.4% in the 100% deficiency group, indicating a 50.4% reduction compared to the control. Similar decreasing trends were observed for potassium, calcium, and magnesium. Notably, calcium content exhibited a sharp decline, likely due to its poor phloem mobility and limited translocation capacity under nitrate-limited conditions (Barker and Pilbeam, 2015).
Table 3.
Mineral element composition of ‘Namcheon’ perilla after 80 days of hydroponic cultivation with Yamazaki nutrient solution based on three replicates.
| T-N | P2O5 | K2O | CaO | MgO |
| (%) | (%) | (%) | (%) | (%) |
| 2.82 ± 0.03 | 1.78 ± 0.03 | 2.64 ± 0.22 | 2.08 ± 0.11 | 0.63 ± 0.01 |
These findings are consistent with prior studies in lettuce (Lee et al., 2000), which also reported significant reductions in tissue nitrogen concentrations under limited nitrogen supply. Nitrate has been shown to act synergistically with other nutrients, facilitating the uptake and assimilation of phosphorus and magnesium and, along with potassium, contributing to osmotic regulation and stomatal function (Epstein and Bloom, 2005).
These findings provide practical threshold values for nitrate-nitrogen management in hydroponic perilla, supporting both high yield and food safety by avoiding excessive nitrate accumulation. This disruption compromises overall nutrient balance and physiological health, underscoring the critical role of adequate nitrate supply in sustaining optimal mineral nutrition in hydroponically grown perilla.