Introduction
In recent years, the production area of tomato using grafted seedlings has increased in many countries in order to improve resistance to biotic and abiotic stresses (Lee and Oda, 2003; Rivero et al., 2003). The successful production of grafted transplants requires highly technical grafting skill and environmental control pre- and post-grafting, especially during healing and acclimatization periods (Vu et al., 2013, 2014). In conventional cultures, the grafting is done in tunnels made from plastic film and shaded by cloth to avoid heat buildup and maintain high relative humidity and low light intensity until the union is formed (Davis et al., 2008; Oda, 1999). Therefore, it is difficult to control the optimal environment for healing and acclimatization of grafted seedlings under natural conditions by changing environmental conditions such as day, night, and seasons. To achieve optimal environment for healing, several types of acclimatization chambers have been developed and are widely used by commercial plug seedling growers in Korea and Japan. Higher survival ratios, faster growth, and higher quality of grafted plants were reported in the healing chambers under highly controlled conditions (Kim et al., 2001; Nobuoka et al., 2005; Vu et al., 2013).
The key environmental factors that influence the healing and acclimatization of grafted seedlings are temperature, humidity, and light. In the healing chambers, they can be easily controlled to achieve optimal environment. The moisture in the substrate of grafted seedlings is also thought to be an important factor that affects the survival rate and seedling quality, but information about this effect is limited. Oda, (1999) suggested that in order to improve survival rate of grafted plants, the seedlings should be exposed to sunshine and the water should be withheld from the plants to avoid spindly growth before grafting. Further, Lee et al. (2010) suggested that maintenance of proper moisture content before and after grafting is critical for the production of uniform grafted seedlings. Although moisture in the substrate of grafted seedlings is an important factor, it is difficult to control during the healing and acclimatization process. Therefore, this study was conducted in order to elucidate the effect of the water contents in the substrate on the survival rate and tomato seedling quality during pre- and post-grafting period.
Materials and Methods
1. Plant material and growing of scions and rootstocks
Tomato seeds were sown in the 128-cell plug trays (Bumnong. Co., Ltd., Jeongeup, Korea) containing commercial nursing substrate (BM2, Berger Group Ltd., Quebec, Canada). ‘Unicorn’ cultivar was used as scion and rootstock and was ‘self-grafted’ for this study. While in the plastic house, the seedlings were watered daily or as required. One week after sowing, the seedlings were fertilized by overhead irrigation twice a week with a commercial fertilizer (Wonder Grow Fertilizers, Chobi Co., Ltd., Seoul, Korea). Twenty-two days after sowing, when two true leaves had unfolded, the seedlings were used for grafting.
2. Grafting method and healing and acclimatization process
Grafting was done by splice grafting method. After placing the scion on the rootstock, ordinary grafting clips were used to fix the grafted position tightly together (Lee and Oda, 2003). After grafting, the plants were placed in healing chambers for 10 days. Healing chambers were also equipped with an auto-control air conditioning system for healing the grafts. Light intensity was approximately 30μmol·m−2·s−1 provided by fluorescent lamps. Relative humidity was maintained 85-90%, temperature was set above 23°C.
3. Water content in substrate treatments
The effect of water content in the substrate during preand post-grafting period was studied by treating tomato seedlings with three levels of water content (high, medium, and low) in the substrate of both the scion and rootstock for 6 days (3 days before and 3 days after grafting). Three levels of water content (high, medium, and low) were applied overhead irrigation 3 times, twice, and once a day about 30, 15, 10mL of water/cell/day, respectively. Treatments with medium water were applied twice a day with sufficient water (similar irrigation schedule of Seedling Company). After grafting, grafted seedlings were placed in the healing room for 10 days. After 10 days in the healing room, the seedlings were transferred to plastic house under natural conditions for 5 days. The grafting clips were removed 2 days after seedlings were out of the healing room.
4. Data collection and analysis
All grafted seedlings were evaluated for signs of grafting failure on the 10th day after grafting. Grafted seedlings on the 15th day after grafting were harvested for analysis of growth characteristics. Plant height (cm), number of leaves, fresh and dry weights of the shoot and root of the seedlings were measured. Leaf area was measured with a leaf area meter (Delta-T Device Ltd., Burwell, Cambridge, UK); leaf chlorophyll value was measured using a chlorophyll meter (SPAD-502 Plus, Konica Minolta Sensing Inc., Osaka, Japan). The fresh shoot and root were dried in an oven (MOV-212F, Sanyo Electric Co., Ltd., Osaka, Japan) at 80°C for 72 hrs before measuring the dry mater. T/R ratio (shoot dry weight/root dry weight ratio) and compactness (shoot dry weight/plant height) were calculated according to Kim et al. (2008). For the root system morphology, the WinRHIZO Pro 2009c (Regent Instruments, Inc, Quebec, Canada) images analysis system was used, coupled with professional scanner Epson 10000XL (Seiko Epson Corporation, Nagano, Japan) according to Arsenault et al. (1995). The roots were detached from their shoots, placed in a tray (15 × 30 × 2cm) with water and positioned on the scanner. Scanned images were analyzed by the WinRHIZO program for total root surface area, total root length, and number of root tips.
The experimental design was a split-plot. Water contents in the substrate of rootstock were the main plot and water contents in the substrate of scion were the sub plot in this study. Nine treatments were designed by combination of three levels (high, medium, and low) of water content in the substrate of rootstock and three levels (high, medium, and low) of water content in the substrate of scion. In each replication, one 128-cell plug tray with 64 plants was measured. For the statistical analysis, ten seedlings per treatment from each replicate were randomly selected. Data were analyzed using the analysis of variance of the Statistical Analysis System (SAS Institute Inc., Cary, NC, USA). Mean separations were calculated using Duncan’s multiple range test.
Results and Discussion
1. The effect of water content in the substrate during pre- and post-grafting period on survival rate of tomato seedlings
The effect of water content in the substrate on survival rate during pre- and post-grafting period is summarized in Table 1. The water content in the rootstock substrate had a statistically significant effect on the seedling’s survival rate, while the water content in the scion substrate did not affect the survival rate significantly. The maximum graft-take ratios (100%) were observed in all treatments with high water content in the rootstock substrate and the survival rate decreased with decreasing water content in the rootstock substrate. The lowest survival rate (75.0%) was observed in the combination of scion low and rootstock high water content in the substrate treatment.
Table 1.
Effect of water content in the substrate during pre- and post-grafting period on survival rate of tomato seedlings.
| Water content in the substrate of scion | Water content in the substrate of rootstock | Survival rate (%) |
|---|---|---|
| High | High | 100 az |
| Mid | 96.8 ab | |
| Low | 78.1 c | |
| Mid | High | 100 a |
| Mid | 96.9 ab | |
| Low | 81.3 c | |
| Low | High | 100 a |
| Mid | 93.8 b | |
| Low | 75.0 c | |
| Significance | ||
| Scion (S) | NSy | |
| Rootstock (R) | ** | |
| S ×R | NS | |
This result agrees with suggestion of Lee et al. (2010) that maintenance of proper moisture content before and after grafting is critical for the production of uniform grafted seedlings. Based on previously reported histological analysis, the transport activities of the vasculature in the grafts were recovered after 3 days and auxin modulated the vascular reconnection 2 days after grafting (Yin et al., 2012). Thus, high water content in the rootstock substrate for 6 days (3 days before and 3 days after grafting) is a very important factor for the contact formation between the scion and the rootstock. Therefore, the maintenance of proper moisture content in the rootstock is critical for improved survival rate of grafted tomato seedlings.
2. The effect of water content in the substrate during pre- and post-grafting period on growth characteristics of tomato seedlings
The different water content in the rootstock substrate had a statistically significant effect on plant height, leaf chlorophyll value, and stem diameter, while the water content in the scion substrate had no significant effect on any growth characteristic. Plant height and stem diameter decreased with deceasing water content in the rootstock substrate; however, no significant difference was detected between high and medium water levels. The highest plant height was obtained in the treatment with scion low and rootstock high water content in the substrate. The water content in the rootstock substrate also significantly affected the leaf chlorophyll value, which increased with decreasing water content in the substrate. Stem diameter decreased with decreasing water contents in the rootstock substrate (Table 2).
Table 2.
Effect of water content in the substrate during pre- and post-grafting period on growth characteristics of tomato seedlings.
| Scion | Rootstock | Plant height (cm) | Number of leaf | Leaf length (cm) | Leaf width (cm) | Leaf chlorophyll value (SPAD) | Stem diameter (mm) |
|---|---|---|---|---|---|---|---|
| High | High | 7.87 abz | 3.3 a | 9.08 a | 5.48 ab | 34.60 b | 2.96 a |
| Mid | 7.61 ab | 3.3 a | 8.02 ab | 5.68 a | 36.90 ab | 3.02 a | |
| Low | 7.28 b | 3.2 a | 7.75 ab | 5.24 ab | 38.88 a | 2.60 b | |
| Mid | High | 7.96 ab | 3.2 a | 9.96 a | 5.64 a | 35.47 ab | 3.05 a |
| Mid | 7.89 ab | 3.5 a | 8.99 a | 5.74 a | 35.67 ab | 2.97 a | |
| Low | 7.16 b | 3.2 a | 7.87 ab | 4.83 b | 37.58 a | 2.77 b | |
| Low | High | 8.21 a | 3.8 a | 9.97 a | 5.34 ab | 30.41 c | 2.86 a |
| Mid | 7.73 ab | 3.4 a | 8.45 ab | 5.47 ab | 36.33 ab | 2.69 b | |
| Low | 7.12 b | 3.2 a | 7.01 b | 5.09 b | 37.96 a | 2.50 c | |
| Significance | |||||||
| Scion (S) | NSy | NS | NS | NS | NS | NS | |
| Rootstock (R) | * | NS | NS | NS | * | * | |
| S R | NS | NS | NS | NS | NS | NS | |
Leaf area, dry weight of shoot and root, T/R, and compactness were significantly different between the various water contents of the rootstock substrate, but not the substrate of the scion. The leaf area and dry weight of shoot and root decreased with decreasing water content in the rootstock substrate; however, no significant difference was detected between high and medium water levels. The T/R increased while the compactness of the grafted seedlings decreased with decreasing water content in the rootstock substrate. The highest value of compactness was observed in seedlings with scion medium and rootstock high water levels (Table 3). These results are in accordance with those of Mahmoud et al. (2011) who found that all growth traits of tomato plants were greatly reduced by successive decreases in irrigation.
Table 3.
Effect of water content in the substrate during pre- and post-grafting period on leaf area, dry weight of shoot and root, T/R ratio, and compactness of tomato seedlings.
| Scion | Rootstock | Leaf area (cm2) | Dry weight (g) | T/Rz ratio | Compactnessy (mg.cm–1) | |
|---|---|---|---|---|---|---|
| Shoot | Root | |||||
| High | High | 12.8 ax | 0.114 a | 0.025 a | 4.251 b | 13.42 ab |
| Mid | 11.7 ab | 0.109 ab | 0.023 ab | 4.915 a | 13.39 ab | |
| Low | 11.0 b | 0.094 b | 0.022 b | 5.004 a | 12.02 b | |
| Mid | High | 13.8 a | 0.129 a | 0.026 a | 4.236 b | 15.04 a |
| Mid | 12.2 ab | 0.108 ab | 0.025 a | 4.387 b | 13.51 ab | |
| Low | 10.7 b | 0.098 b | 0.020 b | 4.894 a | 12.69 b | |
| Low | High | 13.7 a | 0.119 a | 0.119 a | 4.080 b | 14.98 a |
| Mid | 12.3 a | 0.106 ab | 0.024 a | 4.922 a | 12.50 b | |
| Low | 10.0 b | 0.090 b | 0.018 c | 5.525 a | 11.04 c | |
| Significance | ||||||
| Scion (S) | NSw | NS | NS | NS | NS | |
| Rootstock (R) | * | * | * | * | * | |
| S R | NS | NS | NS | NS | NS | |
3. The effect of water content in the substrate during pre- and post-grafting period on root morphology of tomato seedlings
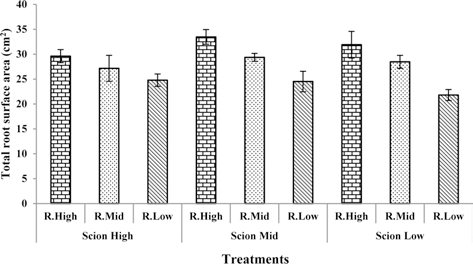
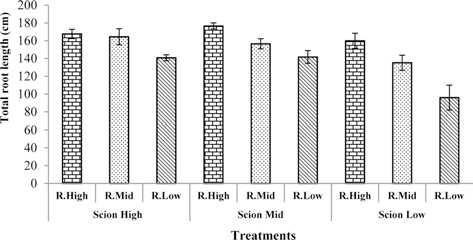
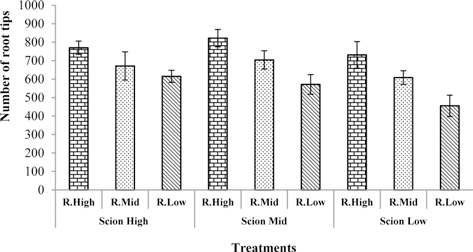
Many earlier studies show that water deficit inhibits root growth (Sharp and Davies, 1979, 1985; Kuzmanoff and Evans, 1981). Accordingly, the total root surface area, total root length, and number of root tips decreased with deceasing water content in the rootstock substrate in this study. The highest values of total root surface area, total root length, and number of root tips were observed in seedlings grafted with rootstock high and scion medium water content in the substrate. The lowest values of total root surface area, total root length, and number of root tips was observed in rootstock low and scion low water content in the substrate (Fig. 1, 2, 3).
Conclusion
The maintenance of proper moisture content in the rootstock during pre- and post-grafting period is critical for improved survival rate and quality of grafted tomato seedlings. Based on the results of this study, it is not advisable to withhold water from the grafts; in contrast, the best results were achieved at the highest levels of water content in the substrate of the rootstock. The maximum survival rate (100%) and the highest value of compactness were observed in the treatment with high water content in the rootstock substrate and medium water content in the scion substrate.