Introduction
Persimmon fruit are highly popular due to their outstanding taste and high nutritive value. In general, persimmon fruit are most likely classified into two major groups based on the astringency (Nam et al., 1998). Non-astringent persimmon is also referred to as sweet persimmon due to the lack of astringency (Song et al., 2005). Of sweet persimmon cultivars, ‘Fuyu’ persimmon is highly important for fresh fruit market due to flesh elasticity and other sensory factors (Taniwaki et al., 2009). For this reason, ‘Fuyu’ persimmon is widely planted and produced. Furthermore, fruit quality is consistent after a wide ranges of postharvest treatments, including modified atmosphere storage (Neuwald et al., 2009; Park et al., 1997), short-term high CO2 treatment (Besada et al., 2014), UV irradiation (Choi, 2011), hot water (Burmeister et al., 1997; Park and Kim, 2002), and controlled atmosphere storage (Burmeister et al., 1997; Wright and Kader, 1997). Astringent cultivars are also widely cultivated for the production of processed persimmon products, such as semi-dried or dried soft, cut and dried, or frozen soft persimmon commodities. To create these products, a steady supply of fresh fruit is essential after harvest. To achieve this goal, the main requirement is the maintenance of consistent fruit quality during cold storage. However, of all the commonly considered fruit quality attributes, flesh firmness changes relatively quickly in astringent fruit (Nakano et al., 2001), although there can be a wide range of values for flesh firmness after storage, regardless of astringency (Romaguera et al., 2009).
Similar to non-astringent persimmon fruit, numerous approaches are utilized for fruit quality maintenance during storage, including controlled atmosphere storage, modified atmosphere storage, and 1-methylcyclopropene (1-MCP) treatment (Arnal et al., 2008; Besada et al., 2008; Harima et al., 2001; Nakano et al., 2001; Salvador et al., 2004). Although these treatments all have utility, the management of storage temperature is the most critical for maintenance of fruit quality attributes of astringent persimmon cultivars during cold storage (Arnal and Del Rio, 2004; Salvador et al., 2005). During cold storage, several types of physiological disorders, such as peel blackening, and flesh browning, are common on non-astringent and astringent persimmon (Besada et al., 2010; Lee and Yang, 1997; Lee, 2001b; Park and Lee, 2008). A wide range of storage temperatures caused variable development of chilling injury in ‘Fuyu’ and ‘Suruga’ persimmon fruit (Collins and Tisdell, 1995). Symptoms of chilling injury include skin browning, flesh browning, skin blackening and fruit softening in cold stored persimmon fruit, irrespective of astringency (Besada et al., 2010; Harima et al., 2002; Lee, 2001a; Nakano et al., 2001; Park and Lee, 2008). Therefore, the objective of this study was to test the hypothesis that lower storage temperature would much better maintain persimmon fruit quality attributes than higher storage temperature for ‘Sangjudungsi’, an astringent persimmon fruit cultivar, stored in air for up to three months. At the same time, an additional objective was to evaluate the effect of storage temperature on the incidence of storage disorders, such as skin blackening, fruit softening, and rot during cold storage.
Materials and methods
1. Plant material
‘Sangjudungsi’ persimmon (Diospyros kaki Thunb.) fruit used in these experiments were collected from mature trees grown at the commercial persimmon orchard in Sangju, Gyeongsangbuk-do, Republic of Korea. Uniformly sized fruit were harvested randomly from the perimeter of each sample tree on October 22, 2015, and a portion of fruit were assessed for fruit quality and maturity attributes at harvest by slicing three replicates of 15 fruit and subjecting them to standard quality evaluation procedures. The remaining fruit were immediately transported to cold storage facilities and subsequent experimental procedures were carried out at the postharvest laboratory at Kyungpook National University campus in Daegu, Republic of Korea.
2. Storage temperature and fruit quality evaluation
Fruit were stored in air at -1, 0.5, or 3°C with 90% relative humidity. Persimmon fruit are generally stored at around 0-1°C. Thus, the selected range of experimental storage temperatures sought to maximize the effect of temperature on fruit quality during cold storage. Fruit were evaluated at 0, 1, 2, or 3 months of cold storage. As fruit quality attributes, ethylene production, respiration rate, flesh firmness, soluble solids concentration (SSC), top and side peel color variables (L*, a*, b*), and water loss were evaluated during cold storage with five fruit (n=5) per replicate per removal.
For the measurement of ethylene production, three fruit were incubated in 1.6 L containers for one hr. Then headspace gas was sampled for ethylene production and respiration rate by withdrawing a 1 mL gas sample from the containers. The sample was injected into a gas chromatograph (GC2010, Shimadzu, Tokyo, Japan), equipped with a flame ionization detector and fitted with a Porapak Q (80/100 2m, Young In Frontier, Seoul, Korea) column. Analyses were run isothermally with an oven temperature of 90°C and injector and detector temperatures of 100°C and 200°C, respectively. The flow rate of He was 20 mL min-1. Ethylene was quantified by peak area using an external standard for calibration. Carbon dioxide was measured using a Hewlett-Packard 6890 gas chromatograph (Hewlett-Packard, Wilmington, DE, USA), equipped with a stainless steel column packed with silica gel (0.92m × 2 mm, i.d.) and fitted with a thermal conductivity detector (TCD). The oven temperature was 100°C, and the TCD current was set to low. The flow rate of helium was 10mL min-1. Carbon dioxide was quantified by peak area with an external standard used for calibration.
Peel color on top and side regions of the fruit peel were measured with a chromameter (Minolta CR-200, Minolta CO., Osaka, Japan). The measurement of peel color variables was done with three readings per region using the same chromameter. Color measurements were expressed as lightness (L*), a* (green-red tonality) and b* (blue-yellow tonality). Flesh firmness was assessed on the same fruit by taking three readings at the equator of each persimmon with a penetrometer (Fruit Pressure Tester, FT 327, Alfonsine, Italy). Expressed juice from the penetrometer readings was used for soluble solids content (SSC) measurement with a refractometer (Model PR-201α, Atago Co., LTD., Tokyo, Japan). Incidence and severity rate of storage disorders, peel blackening, softening, and fruit rot were assessed and recorded. Disorder incidence was expressed as percent fruit affected (n=15). The severity of storage disorders was subjectively scored as 0=0%, 1=1% to 10%, 2=11% to 25%, 3=26% to 50%, 4=51% to 75%, and 5=76% to 100% area of the peel with the largest area with the corresponding symptoms.
3. Statistical analysis
Fruit quality attributes and incidence and severity of storage disorders were analyzed according to a completely randomized experimental design. Fifteen fruit were used for the assessment of fruit quality attributes with five fruit per replicate. All statistical analyses were conducted using SAS version 9.3 (SAS Institute Inc., Carry, NC, USA). Fruit quality attributes and incidence rate and severity of storage disorders were subjected to analysis of variance (ANOVA) using a general linear model (Proc GLM) to evaluate main and interaction effects and means compared using a least significant difference (LSD) test at P=0.05.
Results
Ethylene production was highly affected by storage temperature (Fig. 1A), where higher storage temperature (3°C) inhibited ethylene production, while lower storage temperatures (-1 or 0.5°C) caused a slight increase of ethylene (P<0.0001). In this study, ethylene production gradually increased by 2 month and then remained unchanged or slightly decreased at -1°C or 0.5°C, respectively. However, respiration rate was not affected by storage temperatures for 2 months but higher at -1°C than at 0.5°C at 3 month cold storage (Fig. 1B, P<0.0001).
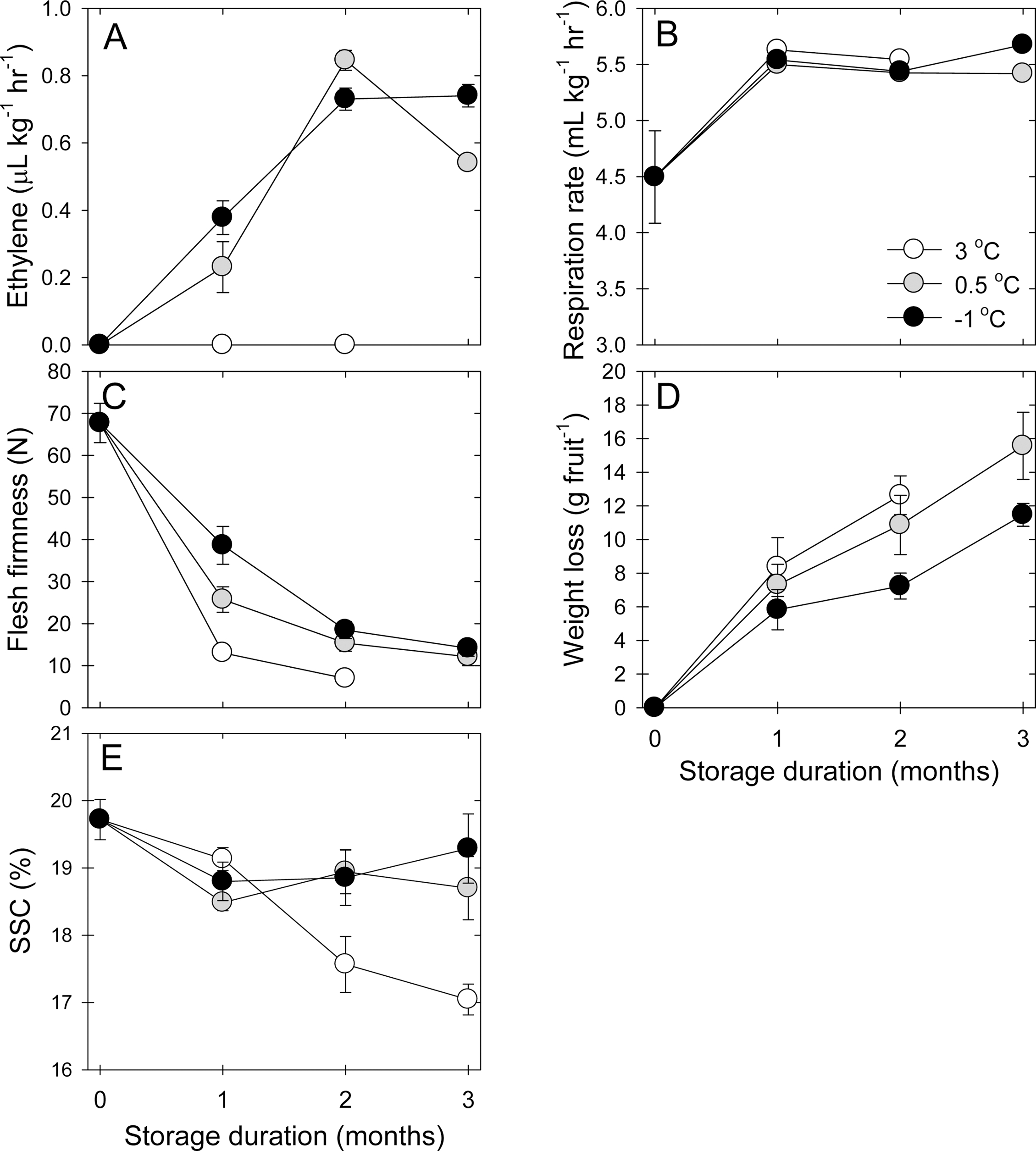
Fig. 1.
The responses of ethylene production (A), respiration rate (B), flesh firmness (C), fresh weight loss (D), and soluble solids content (SSC, E) of ‘Sangjudungsi’ fruit stored in air at -1, 0.5, or 3°C for up to 3 months. Each data point is the mean of fifteen fruit replicates ± standard error (n=15), but ethylene and respiration rate are the mean of three replicates (n=3), where larger than the symbols.
Decrease in flesh firmness was higher at higher storage temperatures than at lower storage temperatures at 1 month (Fig. 1C, P<0.0001). However, after 2 months of air storage, flesh firmness was not different. At the same time, the loss of fresh weight gradually increased as storage duration advanced (Fig. 1D, P<0.01). Furthermore, the increase in fresh weight loss was higher at higher storage temperature than at lower storage temperature (P=0.0436). SSC was not affected by storage temperature after 1 month cold storage. After 1 month, SSC levels were better maintained at lower storage temperature, compared with higher storage temperature (Fig. 1E, P=0.0227).
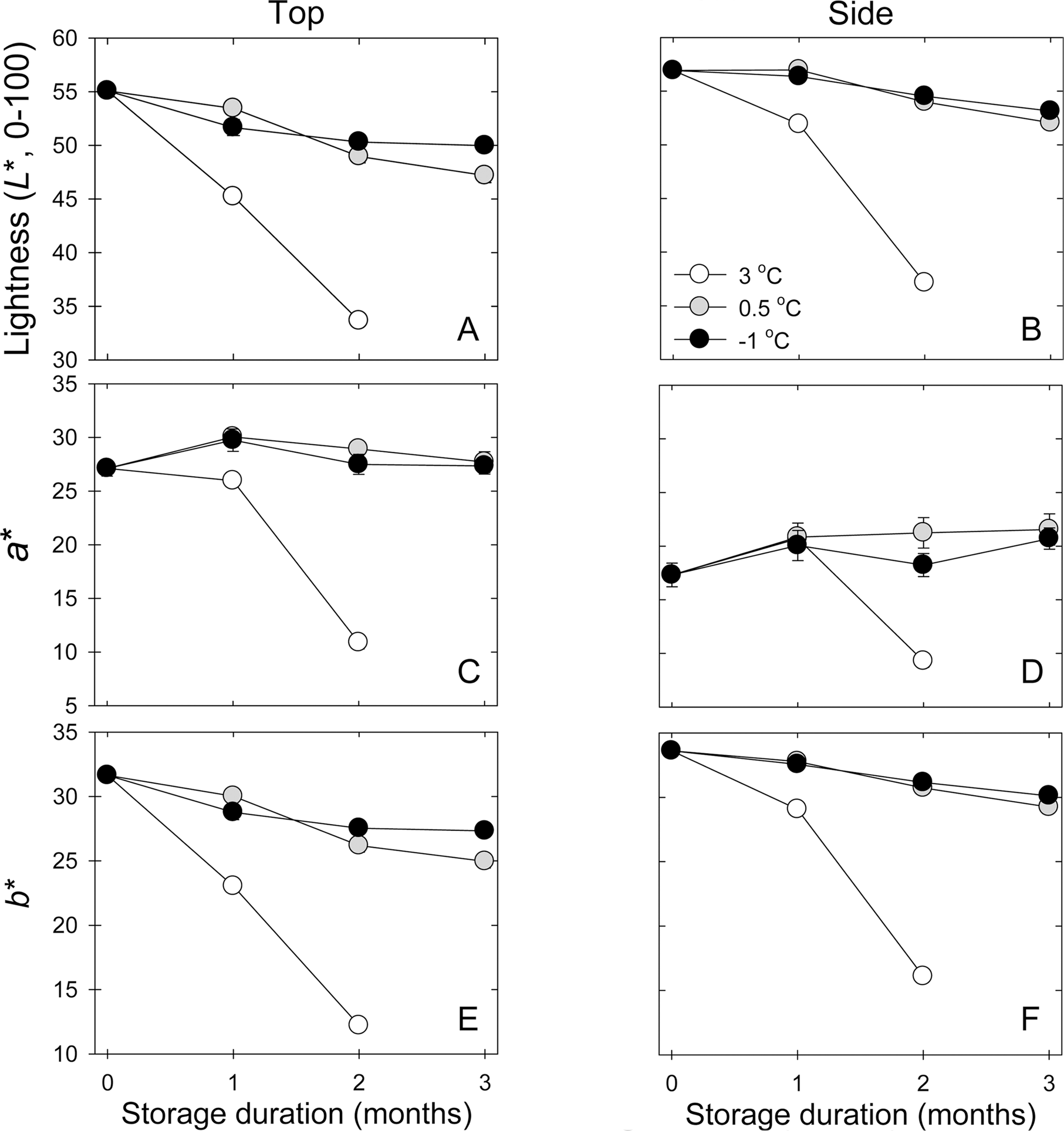
The response of color variables to cold storage temperatures in top and side regions of peel tissues (Fig. 2), lightness (L*) value at the top (calyx-end) region of ‘Sanjudungsi’ persimmon fruits decreased only slightly during air storage at -1 or 0.5°C, but were sharply reduced at 3°C (P<0.0001). At 3 month, L* value was still higher at -1°C than 0.5°C (Fig. 2A and 2B, P<0.0001). Furthermore, L* value was higher at side regions of peel tissue than at top regions of peel tissue (P<0.01). Lower (-1 and 0.5°C) storage temperature did not affect a* value during air storage but higher storage temperature highly decreased a* value at 2 months in both regions of peel tissues (Fig. 2C and 2D, P<0.0001). Value of a* was lower at side regions of peel tissue of persimmon fruit than at top regions, regardless of storage temperatures (P<0.0001). In addition, higher storage temperatures gradually decreased b* value at top and side regions of peel tissues of persimmon fruit but lower storage temperature sharply reduced b* value between 1 and 2 month cold storage (Fig. 2E and 2F, P<0.0001). At the higher storage temperatures, b* value was not affected at side regions of peel tissues but the reduction in b* value was slightly less at -1°C than at 0.5°C at top regions of peel tissues (P<0.0001).
Regarding the incidence of physiological disorders, blackening incidence rate was relatively higher at lower storage temperature than at higher storage temperature, except for at the 2 month evaluation (Fig. 3A). The incidence rate of fruit softening increased as storage temperature increased at 1 month. However, after 2 months air storage, there was no significant difference detected according to storage temperature (Fig. 3B). The incidence of fruit rot was only detected at 3 months with a lower storage temperature, compared with higher storage temperature (Fig. 3C). In addition, the incidence severity of peel blackening increased with the increase in storage temperature at 1 and 2 months but not at 3 months (Fig. 3D). On the other hand, the incidence severity of fruit softening increased with the increase in storage temperature. Furthermore, fruit softening was extreme at 3°C at 2 month cold storage (Fig. 3E). The incidence severity of fruit rot was negligible, compared with the other incidence severity of storage disorders (Fig. 3F). Peel blackening increased with the increase at storage temperature at 1 month but the severity was significantly lower at 0.5°C, compared with the other storage temperatures at 2 month (Fig. 3G). The degree of fruit softening increased with the increase in storage duration and storage temperature (Fig. 3H).
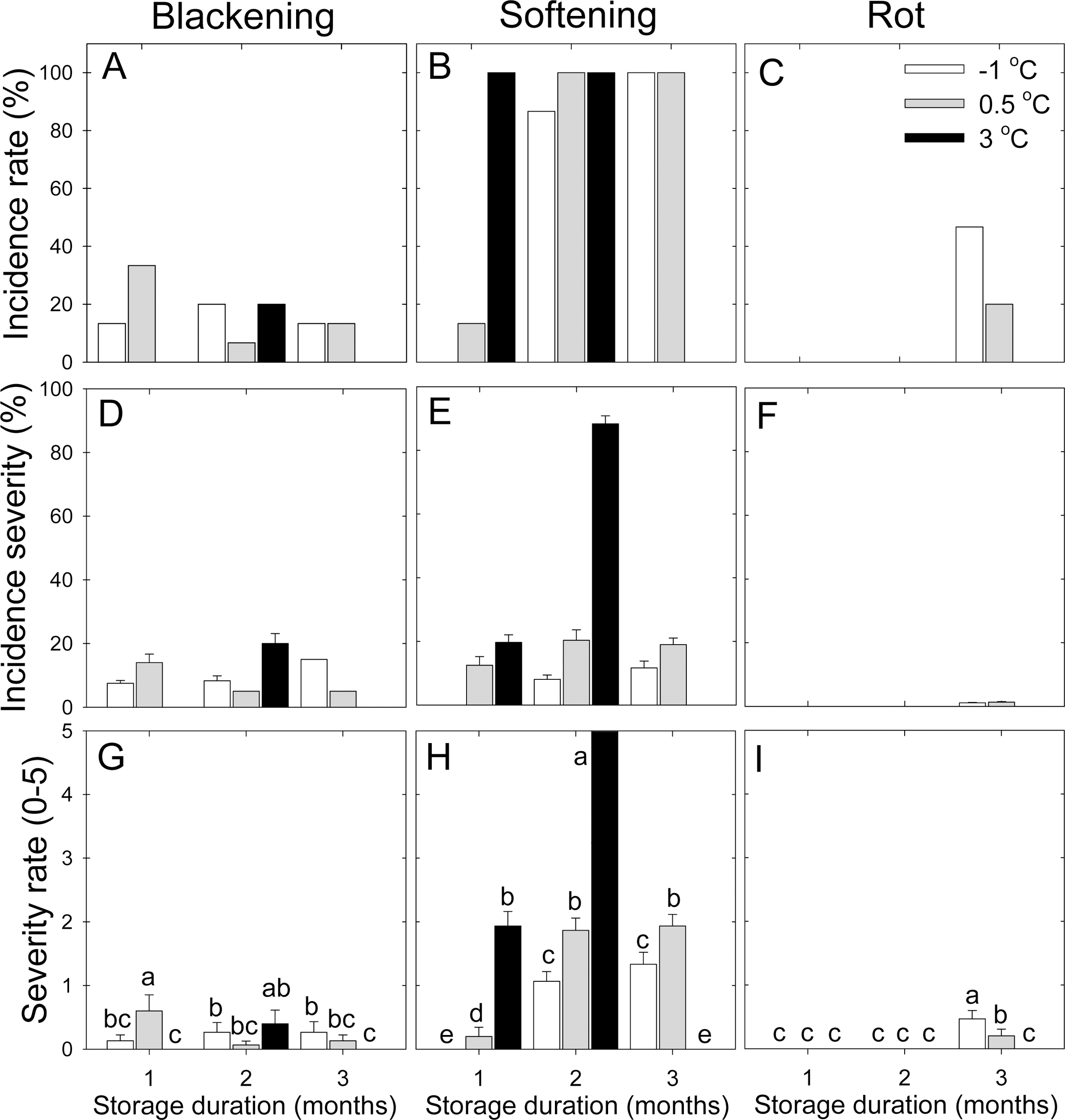
Fig. 3.
The incidence rate (A, B, C), incidence severity (D, E, F) and severity rate (G, H, I) of peel blackening (A, D, G), fruit softening (B, E, H), and rot (C, F, I) of ‘Sangjudungsi’ persimmon fruit stored in air at -1, 0.5, or 3°C for up to 3 months. Each data point is the mean of fifteen fruit replicates ± standard error. Disorder incidence was expressed as percent fruit affected (n=15). The severity of storage disorders was subjectively scored as 0=0%, 1=1% to 10%, 2=11% to 25%, 3=26% to 50%, 4=51% to 75%, and 5=76% to 100% area of the peel with the largest area with the corresponding symptoms.
Discussion
Astringent persimmon fruit are more widely utilized for producing a range of persimmon products, such as semidried or dried persimmon, compared with non-astringent fruit in which are mostly consumed as fresh fruit. Thus, this study was mainly focused on how to manage astringent persimmon fruit quality during cold storage. Of fruit quality attributes, respiration rate was not affected by storage temperatures but ethylene production rate was steadily enhanced at both 0.5°C and 3°C, compared with that at - 1°C. As storage temperature increased, internal ethylene concentration was also differently responded to the given storage temperature in cold stored apple fruits (Johnston et al., 2001). The reduction of respiration and ethylene production rates was decreased with the increase in storage temperature in Chinese bayberry fruit (Yang et al., 2010). However, ethylene production rate increased more significantly at -1°C than at 1°C in ‘Anjou’ pear (Wang, 2016). In addition, lower storage temperature significantly delayed the decrease in flesh firmness of ‘Sangjudungsi’ persimmon, compared with a higher storage temperature. In ‘Rojo brillante’ persimmon fruit, a lower storage temperature highly affected the delay in the loss of flesh firmness (Arnal and Del Rio, 2004). At the same time, lower storage temperature delayed the reduction of fresh weight loss, compared with a higher storage temperature. These results indicated that a lower storage temperature could be involved in delaying in ethylene production and then postpone the decrease in flesh firmness and fresh weight loss as well. Like the responses of the other fruit quality attributes, SSC was greatly influenced by higher storage temperature, in which it was significantly reduced, compared with lower storage temperature. This result indicated that the alteration in SSC level corresponded to storage temperature. Similarly, in ‘W. Murcott’ mandarins, SSC level linearly responded to the increase in storage temperature during cold storage (Obenland et al., 2011).
Peel color variables are also one of main factors to be considered for fruit quality evaluation. Peel tissue color responses in cold stored ‘Sangjudungsi’ fruit were assessed at top (calyx-end) and side regions of persimmon fruit peel tissues. As shown in Fig. 2, lower storage temperatures resulted in less change in all the color variables, irrespective of tissue regions, compared with those at 3°C. Furthermore, these results indicated that top regions of persimmon fruit are much more quickly changed than side regions of fruit skin, regardless of the responses of color variables, suggesting that top region of persimmon fruit ripens earlier than side region. In ‘Navelina’ oranges, lower storage temperature delayed the peel color change during cold storage, compared with higher storage temperature which reduced greenness, thereby less contributing to accumulating the level of carotenoids in which is highly playing an active role in peel pigmentation (Carmona et al., 2012). The color change on peel tissue is likely to be highly associated with carotenoid responses to cold storage, based on physiological responses of β-carotene and β-cryptozanthin in sliced persimmons (Wright and Kader, 1997).
During cold storage, numerous physiological disorders, such as peel blackening, flesh browning, fruit softening and water soaking can appear during cold storage in astringent persimmon fruit. In this study, peel blackening appeared even at the first month at lower storage temperature, compared with higher storage temperature. This kind of storage disorder can highly affect fresh fruit for producing processed items, although some of these products would be relatively less affected by the incidence of peel blackening because some processing procedures remove peel tissues. Conversely, the incidence and severity of fruit softening were significantly associated with the increase in storage temperature. In order to provide relatively fresh and firm fruit, fruit softening should be the main issue to be considered. Furthermore, fruit softening is highly linked with flesh firmness. Immediately after one month cold storage, flesh firmness declined sharply after the first half of storage duration. This result is consistent with the incidence rate and severity of fruit softening. Therefore, at least, lower storage temperature should be reasonable for maintaining fruit firmness during cold storage, compared with relatively higher storage temperature, with the primary objective being to control the incidence of fruit softening. In addition, fruit rot was relatively less detected at lower storage temperature than at higher storage temperature. This result suggested that fruit rot would be negligible for persimmon fruit storage. Although the response of SSC level was highly linked with the increase in storage temperature, fruit rot was by and large relatively less active. This response would be driven by the other factors rather than SSC responses.
In conclusion, some physiological responses of ‘Sangjudungsi’ astringent persimmon fruit were consistently affected by a range of storage temperatures during air storage, while others were not. Flesh firmness, fresh weight loss, and peel tissue color responses were strongly and consistently associated with storage temperature. Ultimately, these physiological characteristics would also be linked with the incidence rate and severity of fruit softening to higher storage temperature. Also, the differential response of peel color variables would be involved in the progress of fresh fruit ripening, thereby contributing to aggravating fruit softening during cold storage. By contrast, peel blackening was highly associated with lower storage temperature. Nonetheless, the incidence rate and severity of peel blackening was relatively slight, compared with fruit softening. Therefore, these results suggested that lower storage temperature would be invaluable to maintain fruit freshness and quality attributes during cold storage.