Introduction
Materials and Methods
1. Plant materials and cultivation conditions.
2. Ethanol treatments
3. Growth and fruit characteristics measurements
4. Gas exchange measurements
5. Extraction of melon samples
6. Total phenolic content (TPC)
7. DPPH radical scavenging activity
8. Statistical analysis
Results
1. Plant growth characteristics
2. Fruit development and morphology
3. Leaf gas exchange
4. Fruit quality attributes
Discussion
Introduction
Melon (Cucumis melo L.) is an economically important horticultural crop cultivated worldwide, valued for its sweetness, flavor, and nutritional quality. Among the various fruit quality attributes, soluble sugar content is a primary determinant of market value (Kyriacou et al., 2018; Lester, 2006). Enhancing sugar accumulation has thus become a critical goal for both producers and breeders. Controlled water management technique, such as regulated deficit irrigation (RDI), have been widely employed to improve fruit quality. RDI and mild drought stress can enhance water use efficiency and promote the accumulation of sugars by activating abscisic acid (ABA signaling pathways that regulate sucrose phosphate synthase (SPS) and other sugar metabolism enzymes (Grumet et al., 2007; Lim et al., 2021; Ma et al., 2022). However, the modulation of ABA-related responses through external chemical cues to further enhance fruit sweetness remains insufficiently explored.
Ethanol has recently emerged as a potential signaling regulator in plant stress physiology. Exogenous application of ethanol has been reported to enhance tolerance to drought and heat in Arabidopsis thaliana, rice, soybean, and cassava by strengthening ABA signal transduction, reducing stomatal aperture, and conserving water under stress (Bashir et al., 2022; Vu et al., 2022). Ethanol treatment has also been associated with evaluated antioxidant enzyme activity and the accumulation of phenolic compounds, highlighting its role in stress-induced metabolic reprogramming(Rahman et al., 2022). These findings suggest that ethanol not only improves physiological resilience but may also modulate secondary metabolism linked to fruit quality traits.
From a practical standpoint, developing a simple and low-cost strategy to improve both stress tolerance and fruit sweetness is of significant interest to the melon industry, particularly under increasingly variable wqter supply conditions driven by climate change. Ethanol, being inexpensive, environmentally benign, and easily applicable through fertigation, could serve as an alternative or complementary approach to conventional deficit irrigation for enhancing fruit quality without compromising yield stability(Ahmed et al., 2025; Todaka et al., 2024).
Despite these promising reports, research on ethanol application has been largely restricted to leafy vegetables and model species, while fruit-bearing horticultural crops remain understudied. In particular, the effect of ethanol on melon fruit quality (including sugar accumulation and antioxidant capacity) has not been systemically evaluated. This knowledge gap is critical, as fruit crops represent the final sink tissues where carbohydrate allocation, transport, and storage are tightly regulated during development(Dai et al., 2011; Stroka et al., 2024). Moreover, fruit quality outcomes are strongly influenced by seasonal variation in temperature, humidity, and irradiance, necessitation multi- seasonal investigations (Song et al., 2021; Toor et al., 2006).
Therefore, the present study aimed to investigate the effects of exogenous ethanol application on melon plants exposed to drought stress. We evaluated morphological growth, leaf gas exchange parameters, and fruit quality attributes (including soluble solids (°Brix), total phenolic content, and antioxidant capacity) across both spring and summer growing seasons. By integrating physiological and bio chemical analyses, this study provides novel insight into the potential of ethanol as a practical cultivation strategy to enhance sugar accumulation and antioxidant capacity in melon fruit under water-limited conditions.
Materials and Methods
1. Plant materials and cultivation conditions.
Two independent experiments were conducted in a single-span glasshouse at the Korea Institute of Machinery & Materials (KIMM, Daejeon, Korea; 36.3°N, 127.3°E), which had a ridge height of 4.5 m and an overall height of 7.5 m. The first trial was carried out from march 14 to June 17, 2024 (spring season), and the second from July 26 to October 6, 2024 (summer season).
The plant material used was muskmelon (Cucumis melo L. cv. Earl’s PMROK, Kyunong Co., Seoul, Korea). On February 16 and June 27, 2024, 70 seeds per trial were sown into Grodan 240-cell rockwool plugs (Grodan, Roermond, Netherlands). After the emergence of the first true leaf, seedlings were transferred to rockwool blocks (10 × 10 × 6 cm, Grodan, Roermond, Netherlands) and grown under controlled nursery conditions of 25°C/18°C (day/night), 60%/70% relative humidity (day/night), 350 ppm CO2, and a light intensity of 270 ± 10 μmol·m-2·s-1 photosynthetic photon flux density (PPFD) with a 16/8h photoperiod. Seedlings were irrigated with the Yamazaki nutrient solution (1982) adjusted to pH 6.0 ± 0.1 and EC 2.0 ± 0.2 dS·m-1, using bottom watering every four days.
Four weeks after sowing (March 14 and July 26, 2024, for the spring and summer trials, respectively), 36 uniform and healthy seedlings with at least three fully expanded leaves were selected and transplanted into coir slabs (HSonb Co., Daejeon, Korea; 100 × 20 × 10 cm, 20 L; chip:dust = 3:7). To encourage root establishment, irrigation was withheld for three days after transplanting. During the early growth to fruit-setting stages, the drainage ratio was maintained at 25% ± 3% on sunny days and 10% ± 3% on cloudy days. Sixteen days prior to harvest, the drainage ratio was reduced to 3% ± 2% to enhance water-use efficiency and increase fruit sugar content.
Environmental conditions varied between trials. In the spring experiment, maximum and minimum temperatures were 34°C and 16°C, with average daytime and nighttime temperature of 23.96°C and 17.68°C, respectively. Relative humidity ranged from 30% to 72%, and ambient CO2 concentration averaged 330ppm. In the summer experiment, maximum and minimum temperatures were 38°C and 17°C, with average daytime and nighttime temperature of 28.28°C and 22.80°C, respectively. relative humidity ranged from 52% to 86%, and CO2 concentration remained at 330 ppm. Solar radiation inside the greenhouse averaged 15.4 MJ·m-2·day-1 during the spring trail and 20.1 MJ·m-2·day-1 during the summer trail. These values are converted from average cumulative radiation dose measurements (J cm-2) to MJ·m-2·day-1. The Yamazaki nutrient solution was supplied throughout both experiments, with pH maintained at 5.8 ± 0.2 and EC at 2.1 ± 0.2dS·m-1.
2. Ethanol treatments
To evaluate the effects of ethanol on melon growth and responses to drought stress, four ethanol concentrations were established: 0mM (control), 25 mM, 50 mM, and 100 mM. Ethanol (≥99.5%, analytical grade) was directly dissolved into the Yamazaki nutrient solution and applied via the fertigation system. Ethanol treatments were applied during the first two irrigation events conducted 16 and 15 days before harvest, corresponding to the onset of drought stress period. From 15 days before harvest onward, all treatment (Control, E25, E50, and E100) were subjected to the same drought condition by maintaining the drainage ratio below 3%, ensuring identical irrigation volumes among treatment. Treatments were initiated prior to the imposition of drought stress and continued until the end of the stress period. The same irrigation volume was supplied across all treatments to ensure uniform water and nutrient delivery, with ethanol concentration being the only variable.
3. Growth and fruit characteristics measurements
Growth parameters were measured according to the guidelines of the Korea Agriculture Technology Promotion Agency. Plant height, leaf length, and stem thickness were recorded three days after pinching using a vernier caliper (NTD31-AX; Mitutoyo Inc., Kawasaki, Japan) and a measuring tape (DWHT36098-1; DeWalt, Baltimore, MD, USA). Leaf area index (LAI) was calculated following (Wu et al., 2010) using the formula:
Leaf area = 0.73 × Leaf length × Leaf width
Fruit setting was conducted when female flowers appeared between the 11th and 13th nodes. In the first experiment (April 29, 2024), fruit setting was carried out on May 1, and in the second experiment (August 13, 2024), fruit setting occurred on August 16. In both trials, all fruits were thinned except for the fruit with the longest vertical length among those set between the 11th and 13th nodes. Pinching was performed at the 22nd node to regulate plant growth.
Fruit characteristics were measured at harvest. Fruit length, fruit width, and flesh thickness were determined using a long vernier caliper (C-L; Mitutoyo Inc., Kawasaki, Japan), and fruit weight was measured with an electronic scale (SPX2202KR; Ohaus Co., Ltd., Parsippany, NJ, USA). For soluble solid content (SSC), fruits were peeled, and flesh tissue close to the rind was cut into 1 cm slices, while the remaining flesh was sliced at 0.5 cm intervals. The samples were wrapped in sterile gauze, juice was extracted using a manual juicer, and SSC was measured using a glucose refractometer (PR-32a; Atago Co., Ltd., Tokyo, Japan).
4. Gas exchange measurements
Leaf gas exchange parameters were measured two weeks before ethanol treatment and subsequently at 6, 24, and 72h after ethanol application under drought stress. Measurements were conducted on the leaf located one node above the fruit set on the main stem using a portable photosynthesis system (LI-6800; LI-COR Inc., Lincoln, NE, USA). The net photosynthetic rate (Pn), transpiration rate (E), and stomatal conductance (gs) were recorded between 09:30 and 14:00h.
To ensure stable readings, leaves were acclimated for at least 10 min prior to measurement under a photosynthetic photon flux density (PPFD) of 1000 μmol·m-2·s-1 and a reference CO2 concentration of 330 μmol·mol-1. The leaf chamber light source consisted of red and blue LEDs. Air temperature and relative humidity were controlled according to seasonal conditions, with 30°C and 30% RH in spring, and 32°C and 50% RH in summer, respectively, corresponding to the greenhouse environment.
5. Extraction of melon samples
The extraction procedure was adapted and modified from Ismail et al. (2010). Freeze-dried melon flesh was ground into a fine powder using a mortar and pestle and sieved through a 100-mesh sieve. The powdered sample was extracted with methanol at a ratio of 1:10 (w/v). The mixtures were manually swirled for 15min and subsequently sonicated for 60 min. After extraction, the mixtures were filtered through Whatman No. 1 filter paper, and the residue was re-extracted twice using the same procedure. All filtrates were pooled, and methanol was removed under reduced pressure. The resulting methanolic extracts were stored at -80°C until further analysis.
6. Total phenolic content (TPC)
Total phenolic content (TPC) of muskmelon extracts was determined using the Folin–Ciocalteu assay, as described by Singleton and Rossi (1965), with slight modifications. Briefly, 20 μL of methanolic extract was mixed with 250 μL of 10-fold diluted Folin-Ciocalteu reagent and 200 μL of 7.5% (w/v) sodium carbonate (Na2CO3). The mixture was incubated at 40°C for 30 min, and the absorbance was recorded at 760 nm using a spectrophotometer (EPOCH-SN; Agilent Technologies, Inc., USA). Gallic acid was used as a calibration standard, and TPC was expressed as milligrams of gallic acid equivalents per gram of extract (mg GAE·g-1 extract).
7. DPPH radical scavenging activity
The DPPH radical scavenging activity of muskmelon extracts was determined according to the method of Blois (1958), with slight modifications. Briefly, 20 μL of methanolic extract was mixed with 0.49 mL of methanol and 0.39 mL of DPPH methanolic solution (4 mg·100mL-1). The mixtures were vortexed vigorously and incubated in the dark for 60 min. Absorbance was then measured at 515 nm using a spectrophotometer (EPOCH-SN; Agilent Technologies, Inc., USA). Radical scavenging activity was expressed as the percentage of DPPH inhibition relative to the control.
8. Statistical analysis
All data were analyzed using SPSS 29.0 software (IBM, Chicago, IL, USA). Significant differences among treatments were determined by t-test and Tukey’s multiple comparison test at p < 0.05.
Results
1. Plant growth characteristics
Plant growth exhibited distinct seasonal differences (Table 1). The experiments were conducted in a single-span greenhouse oriented north-south.
Table 1.
Growth characteristics of muskmelon during spring and summer.
In spring, plants developed significantly larger leaves, longer stems, and greater leaf area compared with those grown in summer. Specifically, leaf length, leaf width, stem height, petiole length, and leaf area were 20%, 13%, 30%, 22%, and 32% higher, respectively, than in summer-grown plants (Table 1).
In summer, plants maintained smaller leaves and shorter stems. Ethanol application did not cause significant variation in vegetative traits within each season, indicating that exogenous ethanol had little influence on plant morphology.
2. Fruit development and morphology
Fruit size and morphology were significantly influenced by season, with additional moderate effects of ethanol treatment observed in specific traits. Summer-grown fruits were markedly heavier and larger than those produced in spring. Within each season, ethanol treatment significantly affected fruit weight and length, while fruit width and flesh thickness remained statistically unchanged (Table 2).
Table 2.
Characteristics of muskmelon after ethanol treatment during summer and spring.
3. Leaf gas exchange
Leaf gas exchange parameters were strongly affected by ethanol application under drought stress (Fig. 2). In both spring and summer, net photosynthetic rate (Pn), stomatal conductance (gs), and transpiration rate (E) decreased sharply within 6 h as ethanol concentration increased. Partial recovery was observed at 24 h in the 25 mM treatment, whereas recovery in the 50 and 100 mM groups remained limited. By 72 h, all treatments gradually approached control levels. Although the overall patterns were consistent across seasons, absolute gas exchange rates were higher in summer than in spring.
4. Fruit quality attributes
Fruit quality parameters, including soluble solids content (°Brix), total phenolic content (TPC), and antioxidant activity (DPPH), showed clear seasonal and treatment- dependent patterns (Figs. 1, 3).
In spring, °Brix, TPC and DPPH values increased with ethanol concentrations, reaching the highest level at 100mM. In summer, °Brix peaked at 50mM content ethanol, while TPC and DPPH activity showed no significant differences among ethanol treatments except for consistently lowest control group. These findings indicate that the effects of ethanol treatment on fruit quality differed by season.
Discussion
Plant growth and fruit development exhibited clear seasonal differences, which were influenced by both greenhouse orientation and environmental factor. The experiment was conducted in a north-south oriented single-span greenhouse, where light distribution tends to be less uniform than in east-west orientation (Matsumori et al., 2025, Runkle, 2017). Such spatial heterogeneity in light, combined with seasonal variation, likely contributed to the growth differences observed between spring and summer. Larger leaves and elongated stems in spring may reflect an adaptive response to heterogeneous light conditions (Tang et al., 2022, Wang et al., 2022), whereas compact growth in summer could be genotype-specific response to high irradiance and temperature, minimizing water loss (Miccolis and Saltveit Jr, 1991).
Exogenous ethanol treatment significantly influenced both soluble solids content (°Brix) and antioxidant activity (DPPH radical scavenging) in melon fruit, with clear seasonal differences (Figs. 1 and 2). In spring, °Brix increased progressively with ethanol concentration, and the highest values were observed in the 50 mM and 100 mM treatments. Antioxidant activity also rose markedly in treated groups versus control, particularly for E25 and E50 (Fig. 2b). These results suggest that under the relatively mild spring environment, ethanol strengthened drought stress perception and activated sugar metabolism and phenolic biosynthesis, thereby improving both sweetness and antioxidant capacity.
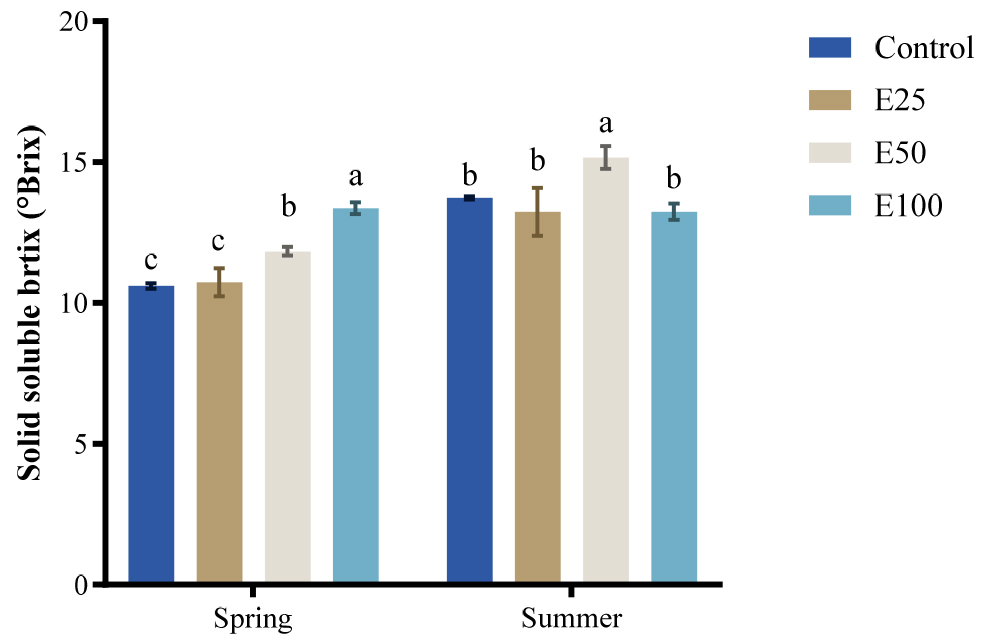
Fig. 1.
Solid soluble content (°Brix) of fruit in spring and summer under different ethanol treatment (Control, E25, E50, E100) in spring and summer. Values are presented as means ± SE (n = 3). Statistical comparisons were made among treatments within the same season, and different letters above the bar indicate significant differences among treatments at p < 0.05 according to Tukey’s HSD test.
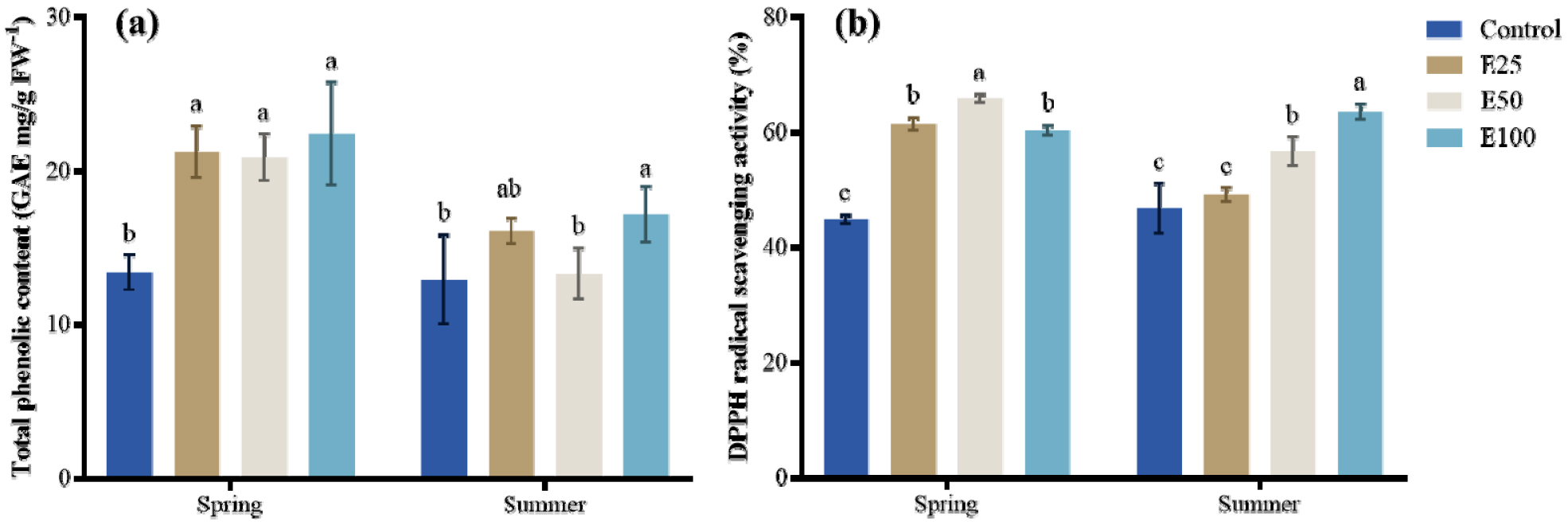
Fig. 2.
(a) Total phenolic concentration and (b) DPPH radical scavenging activity of fruits cultivated under different ethanol treatment (Control, E25, E50, E100) in spring and summer. Values are presented as means ± SE (n = 3). Statistical comparisons were made among treatments within the same season, and different letters above the bar indicate significant differences among treatments at p < 0.05 according to Tukey’s HSD test.
In summer, however, the response pattern differed. °Brix reached its maximum at 50 mM, but declined at 100 mM (Fig. 1), indicating that excessive ethanol may have partially disrupted metabolic balance under high temperature and humidity conditions. In contrast, antioxidant activity continued to increase with ethanol concentration, with the highest value recorded at 100 mM (Fig. 2b). This seasonal discrepancy highlights that ethanol’s effects on fruit quality traits are highly environment-dependent, promoting sugar accumulation under favorable spring conditions, while enhancing antioxidant activity at higher concentrations in summer.
These findings align with previous studies showing that ethanol enhances plant drought tolerance by modulating ABA signaling, reducing stomatal aperture, and strengthening antioxidant defense system (Bashir et al., 2022, Rahman et al., 2022). Studies in tomato also demonstrated that ethanol pretreatment alleviated heat stress damage but with context- dependent efficacy (Todaka et al., 2024). Our results extend these observations to fruit-bearing crops, showing that ethanol not only modulates physiological resilience (Fig. 3) but also impacts fruit quality traits such as sugar accumulation and phenolic-based antioxidant (Figs. 1 and 2).
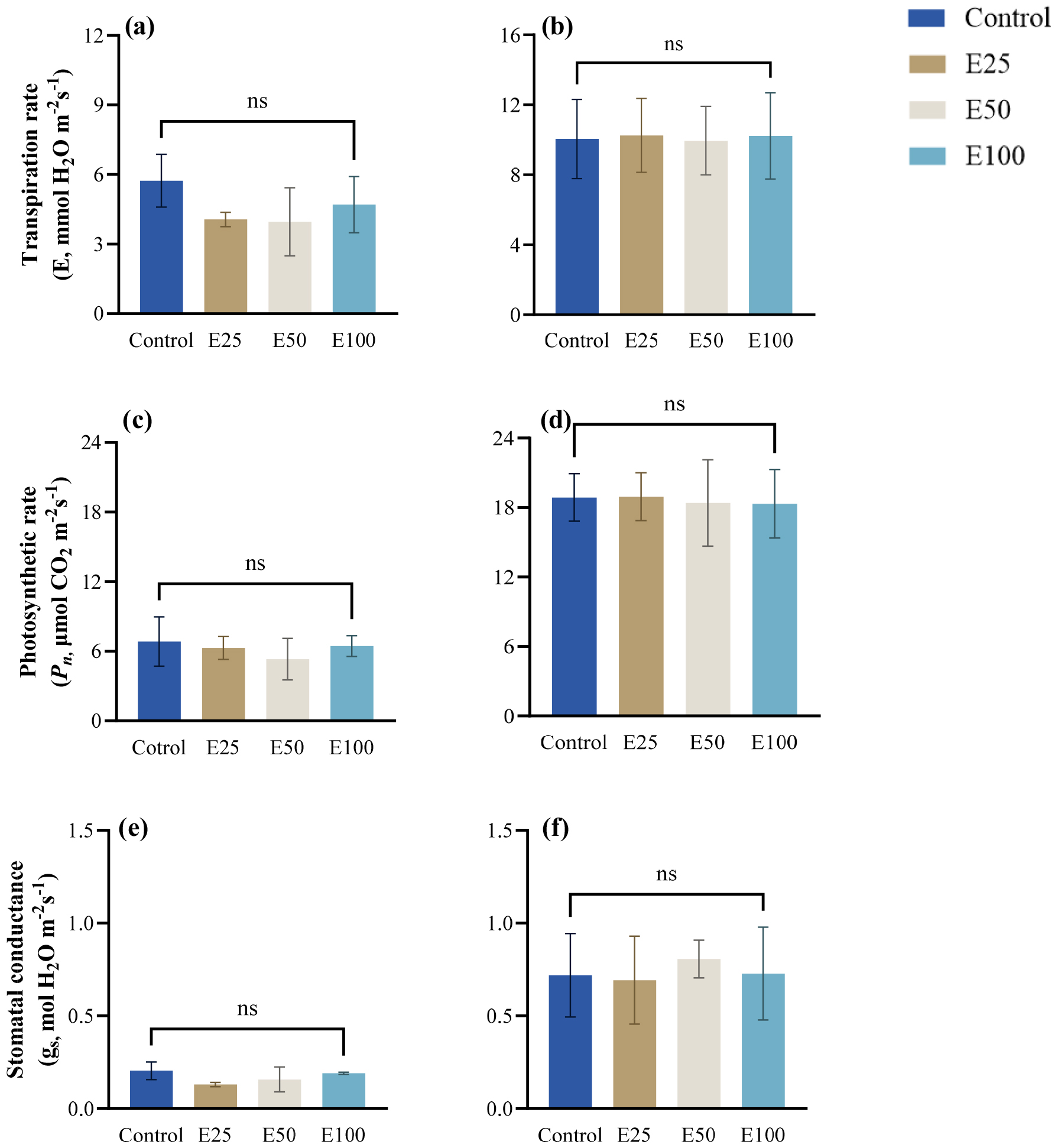
Fig. 3.
Transpiration rate in spring (a) and summer (b), photosynthetic rate in spring (c) and summer (d), stomatal conductance in spring (e) and summer (f) measured 2 weeks before ethanol treatment (Control, E25, E50, E100). Measurements before ethanol treatment were taken on the same individuals as those in the subsequent treatment. Values are presented as mean ± SE (n = 3). Statistical comparisons were made among treatments within the same season, and different letters above the bar indicate significant differences among treatments at p < 0.05 according to Tukey’s HSD test. “ns” denotes no significant difference.
The seasonal divergence in response likely reflects differences in temperature, humidity, and vapor pressure deficit. In spring, moderate stress conditions allowed ethanol to promote both sugar and phenolic accumulation (Table1, Fig. 2), whereas in summer, evaluated stress intensity favored antioxidant enhancement at the expense of sugar accumulation. Similar trade-offs have been observed in melon and watermelon, where abiotic stress altered sugar and phenolic metabolism in opposite direction (Toscano et al., 2019, Song et al., 2021). Moreover, storage studies show that higher temperatures accelerate sucrose breakdown and activate phenylpropanoid metabolism, resulting in elevated phenolic content but reduced sweetness (Wu et al., 2024). Such patterns support our findings that seasonal environment determines the balance between sugar accumulation and antioxidant induction.
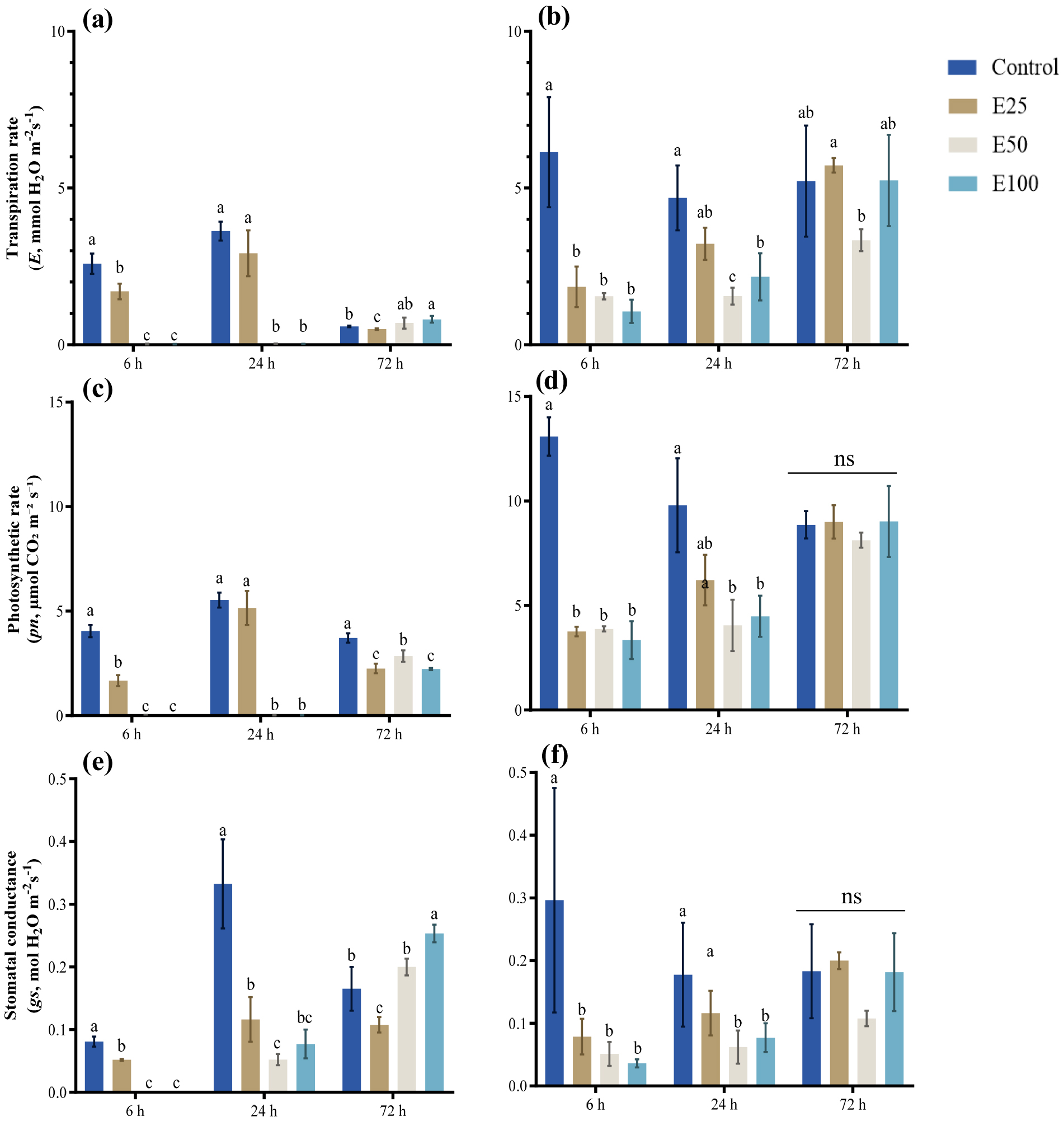
Fig. 4.
Transpiration rate in spring (a) and summer (b), photosynthetic rate in spring (c) and summer (d), stomatal conductance in spring (e) and summer (f) measured at 6 hours, 24 hours, and 72 hours after treatment (Control, E25, E50, E100). Values are presented as mean ± SE (n = 3). Statistical comparisons were made among treatments within the same season, and different letters above the bar indicate significant differences among treatments at p < 0.05 according to Tukey’s HSD test. “ns” denotes no significant difference.
Ethanol application enhanced fruit quality traits in a season-dependent manner: in spring, both sugar accumulation (°Brix) and antioxidant capacity (TPC and DPPH) increased, whereas in summer ethanol mainly promoted antioxidant activity while its effect on sugar accumulation was limited, indicating strong interactions between environmental conditions and ethanol-induced responses. However, the present study was limited by the use of a single cultivar, reliance on a single antioxidant assay (DPPH), and the absence of molecular and metabolomic analyses. Future research should therefore include multiple genotypes, additional antioxidant assays such as ABTS and FRAP, and omics-based approaches, as well as integrate ethanol application with cultivation practices like regulated deficit irrigation or grafting to better evaluate its potential in commercial melon production.




