Introduction
Materials and Methods
1. Plant materials and cultivation conditions
2. Multispectral image analysis
3. Plant growth measurements
4. Statistical analysis
Results and Discussion
1. Growth of kimchi cabbage seedlings as affected by cell size of plug tray
2. PCS-Based prediction of growth and grading of kimchi cabbage seedlings
3. Growth of kimchi cabbage after transplanting by seedling grade
Conclusion
Introduction
Kimchi cabbage (Brassica rapa L. ssp. pekinensis) is a major vegetable crop in Korea, widely consumed as the primary ingredient in kimchi. In 2023, the average annual consumption per person of kimchi cabbage was 52 kg, the highest among all vegetable crops (MAFRA, 2024). More than 90% of kimchi cabbage is cultivated in open fields, yielding 1,958,500 tons in 2023, which accounted for 93% of total production (Statistics Korea, 2024). However, climate change strongly affects the productivity of open-field kimchi cabbage, with high temperatures during cultivation reducing yield and leading to sharp price increases (Choi et al., 2011; Kim et al., 2013; Lee et al., 2016; Wi et al., 2020). Therefore, the production of vigorous seedlings capable of maintaining stable growth under extreme weather conditions is of critical importance.
Seedling production is a crucial stage in vegetable cultivation, and factors such as nutrient supply, growing medium, and plug tray cell size significantly influence seedling growth and quality (Kim et al., 2019; Leskovar and Othman, 2016; Qin and Leskovar, 2020; Tremblay and Senécal, 1988). Seedling growth and quality are largely determined by environmental conditions and cultivation practices during the nursery stage, and these differences also affect subsequent growth after transplanting. Although commercial nurseries have established cultivation practices and seedling quality standards based on accumulated experience, there is an increasing demand for objective, data-driven techniques for seedling evaluation.
Imaging technology provides a rapid, efficient, and non-destructive approach for predicting crop growth and monitoring plant health. Numerous studies have reported the application of various imaging techniques—including RGB imaging, chlorophyll fluorescence imaging, multispectral imaging, and 3D multispectral scanning—to detect growth responses and physiological disorders in stressed vegetable crops such as tomato (Rossi et al., 2022), basil (Lazarević et al., 2021), watermelon (Nabwire et al., 2022), and lettuce (Zhang et al., 2020). Imaging technology is recognized as an effective tool for plant growth monitoring and trait analysis, but most previous studies have focused on the transplanting stage. Despite these advances, research on its application in commercial seedling production remains limited, highlighting the need for practical approaches to improve seedling growth and quality.
Therefore, the objectives of this study were (i) to evaluate the applicability of imaging technology using projected canopy size (PCS) for predicting the growth of kimchi cabbage seedlings cultivated in plug trays with different cell numbers, and (ii) to compare post-transplant growth among seedling grades classified by PCS.
Materials and Methods
1. Plant materials and cultivation conditions
The experiment was conducted in a glass greenhouse at the University of Seoul, Seoul, Republic of Korea (37°34′57.1″ N, 127°03′37.7″ E) from October 21, 2024 to January 3, 2025. Kimchi cabbage ‘Cheongmyeonggaeul’ (Nongwoobio Co., Ltd., Suwon, Republic of Korea) was used in this study. Seeds were sown and grown in plug trays with 72, 105, 128, 162, and 200 cells filled with commercial growing media (Biosangto, Nongwoobio Co., Ltd., Suwon, Republic of Korea). Seedlings were sub-irrigated daily with tap water until 6 days after sowing (DAS) and then with a standard nutrient solution for vegetable seedlings (Lee et al., 2000). The elemental composition of the standard nutrient solution was as follows: macronutrients N 8.0, P 2.4, K 2.4, Ca 4.8, and Mg 1.6 me·L-1 and micronutrients Fe 2.0, B 0.5, Mn 0.5, Zn 0.05, Cu 0.02, and Mo 0.01 mg·L-1. The nutrient solution was adjusted to pH 6.2 and EC 1.1 dS·m-1 using a pH meter (LAQUAtwin pH-33, HORIBA Advanced Techno Co., Ltd., Kyoto, Japan) and an EC meter (LAQUAtwin EC-33, HORIBA Advanced Techno Co., Ltd., Kyoto, Japan). In the greenhouse, environmental conditions were monitored using an agricultural environmental monitoring system (aM-31, WISE Sensing Inc., Yongin, Republic of Korea). During the seedling production period, the average air temperature, relative humidity, and daily light integral (DLI) were 21.6 ± 3.0°C, 55.8 ± 15.6%, and 8.7 ± 4.1 mol·m-2·d-1, respectively.
On November 15, 2024, 20 seedlings per treatment were transplanted into pots (32 cm in diameter) and cultivated for 49 days. During the cultivation period, a nutrient solution formulated for Brassicaceae plants (RDA, 2021) was supplied by drip irrigation and maintained at pH 6.2 and EC 1.5 dS·m-1. The nutrient solution used for cultivation after transplanting contained N 15.2, P 3.0, K 6.8, Ca 4.8, and Mg 2.0 me·L-1, and Fe 3.0, B 0.5, Mn 0.5, Zn 0.05, Cu 0.02, and Mo 0.01 mg·L-1. During the cultivation period, the average air temperature, relative humidity, and daily light integral were 21.3 ± 2.9°C, 37.0 ± 10.3%, and 5.8 ± 1.9 mol·m-2·d-1, respectively.
2. Multispectral image analysis
At 25 DAS, multispectral images of individual seedlings and plug trays were acquired using a Plant Image Measurement System (PIMS, Podo Inc., Seongnam, Korea) equipped with LEDs and a multispectral camera. The LEDs provided light in the range of 400-1000 nm to measure spectral reflectance in the visible (VIS) and near-infrared (NIR) regions. The multispectral camera was mounted at the top of the black box and captured images at five wavelengths: blue (450 nm), green (550 nm), red (650 nm), NIR1 (750 nm), and NIR2 (830 nm). Images at these five wavelengths were calibrated using a reflectance standard bar and merged into a single composite image. The projected canopy size (PCS) was then extracted from the images using ENVI software (version 5.3, L3Harris Geospatial, Broomfield, CO, USA). The PCS of each individual seedling was calculated based on the pixel count of leaf regions.
3. Plant growth measurements
After image acquisition, leaf length, number of leaves, leaf area, SPAD value, and shoot fresh and dry weights were measured. SPAD values and leaf area were obtained using a chlorophyll meter (SPAD-502Plus, Konica Minolta, Inc., Tokyo, Japan) and a leaf area meter (LI-3100, LI-COR Inc., Lincoln, NE, USA), respectively. Shoot fresh and dry weights were determined using a scale (EWJ 300-3, Kern & Sohn GmbH, Balingen, Germany). The correlation between PCS and growth parameters (leaf area and shoot fresh weight) was analyzed to confirm the applicability of PCS for detecting seedling growth. Based on PCS values, kimchi cabbage seedlings were classified into three grades (A-C) and transplanted according to their grade. At 7, 21, 35, and 49 days after transplanting (DAT), kimchi cabbage growth among seedling grades was assessed by measuring the number of leaves, leaf area, and shoot fresh weight.
4. Statistical analysis
The experimental data were analyzed using SAS software (version 9.4, Enterprise Guide 8.5; SAS Institute Inc., Cary, NC, USA), and mean differences among treatments were evaluated by Duncan’s multiple range test at p < 0.05. Linear regression analysis was performed with SigmaPlot software (version 15.0, Systat Software Inc., Palo Alto, CA, USA).
Results and Discussion
1. Growth of kimchi cabbage seedlings as affected by cell size of plug tray
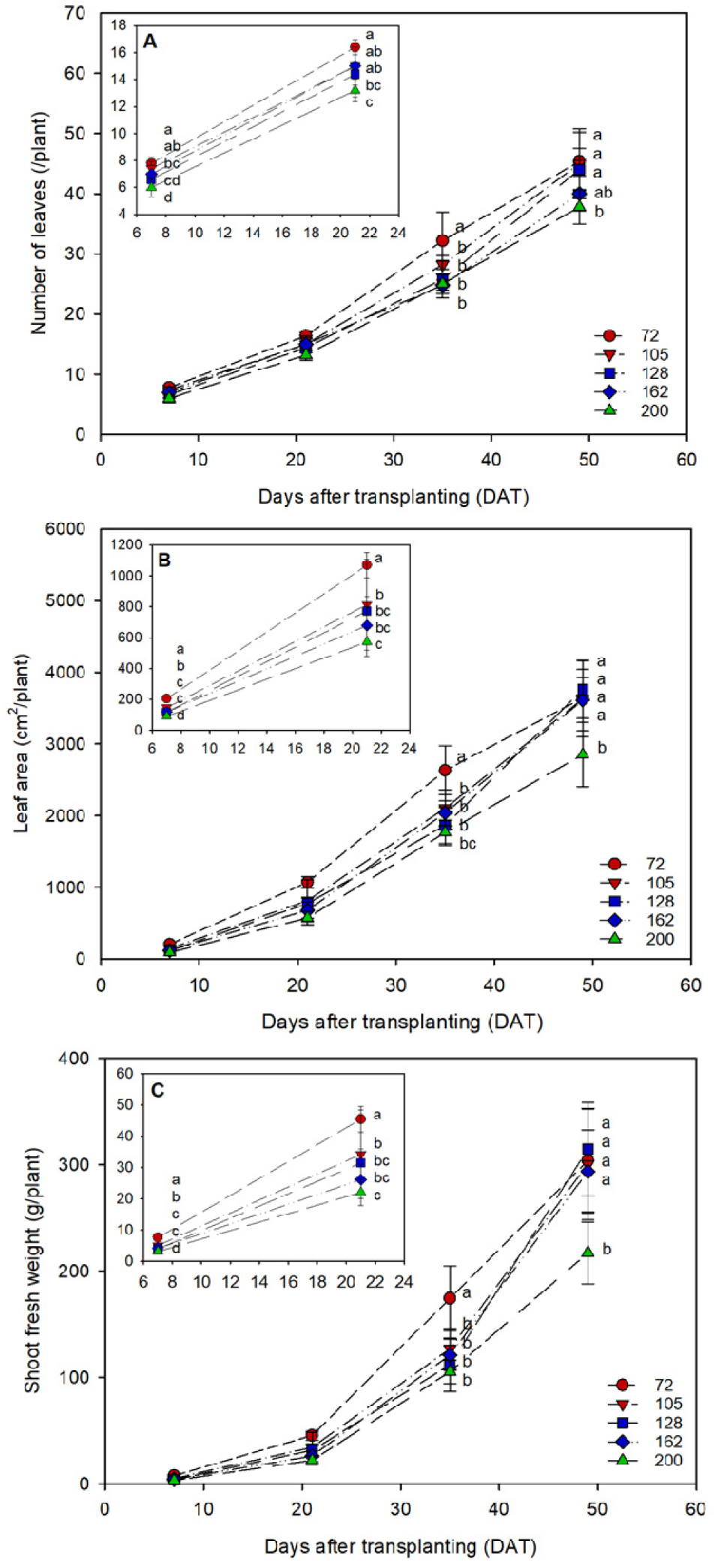
The growth of kimchi cabbage seedlings was significantly affected by plug tray cell size (Table 1, Fig. 1). Growth generally increased with increasing cell size, whereas SPAD values were not significantly influenced. Among the measured parameters, leaf area exhibited the greatest sensitivity to cell size. Growth responses were statistically similar between the 72- and 105-cell trays and between the 128- and 162-cell trays, while seedlings in the 200-cell trays showed significantly lower values across all parameters.
Table 1.
Growth parameters of kimchi cabbage seedlings grown in different plug cell trays at 25 days after sowing.
|
Cells of plug tray |
Leaf length (cm) |
No. of leaves (/plant) |
SPAD value |
Leaf area (cm2/plant) |
Shoot fresh weight (g/plant) |
Shoot dry weight (g/plant) |
| 72 | 12.68 az | 5.0 a | 26.48 ab | 91.72 a | 3.339 a | 0.205 a |
| 105 | 12.06 ab | 5.0 a | 28.18 a | 82.25 b | 3.120 a | 0.186 a |
| 128 | 11.38 ab | 4.0 b | 25.16 b | 53.82 c | 1.988 b | 0.121 b |
| 162 | 10.54 c | 4.0 b | 26.98 ab | 51.68 c | 2.003 b | 0.120 b |
| 200 | 9.90 c | 3.4 c | 25.28 b | 33.52 d | 1.414 c | 0.080 c |
Plug tray cell size is recognized as a key factor influencing seedling growth. Variation in cell size alters rooting volume, thereby inducing physiological and morphological changes in seedlings (NeSmith and Duval, 1998). In general, smaller cells delay seedling development due to restrictions on root growth. Previous studies have reported that pepper seedlings grown in larger cell volumes exhibited greater shoot and root dry weight (Bar‐Tal et al., 1990), whereas those raised in trays with more cells under the same medium conditions showed reduced plant height and total dry weight (Nair and Carpenter, 2016). Similarly, reductions in leaf number, leaf area, shoot dry weight, and root dry weight have been observed in watermelon seedlings as cell volume decreased (Liu and Latimer, 1995), and muskmelon seedlings grown in larger cells exhibited enhanced growth (Walters et al., 2005). In cucumber, seedlings produced in 128-cell trays were of lower quality than those in 50-cell trays (Wenneck et al., 2022). Consistent with these findings, our results demonstrate that the growth of kimchi cabbage seedlings is also strongly influenced by plug tray cell size.
2. PCS-Based prediction of growth and grading of kimchi cabbage seedlings
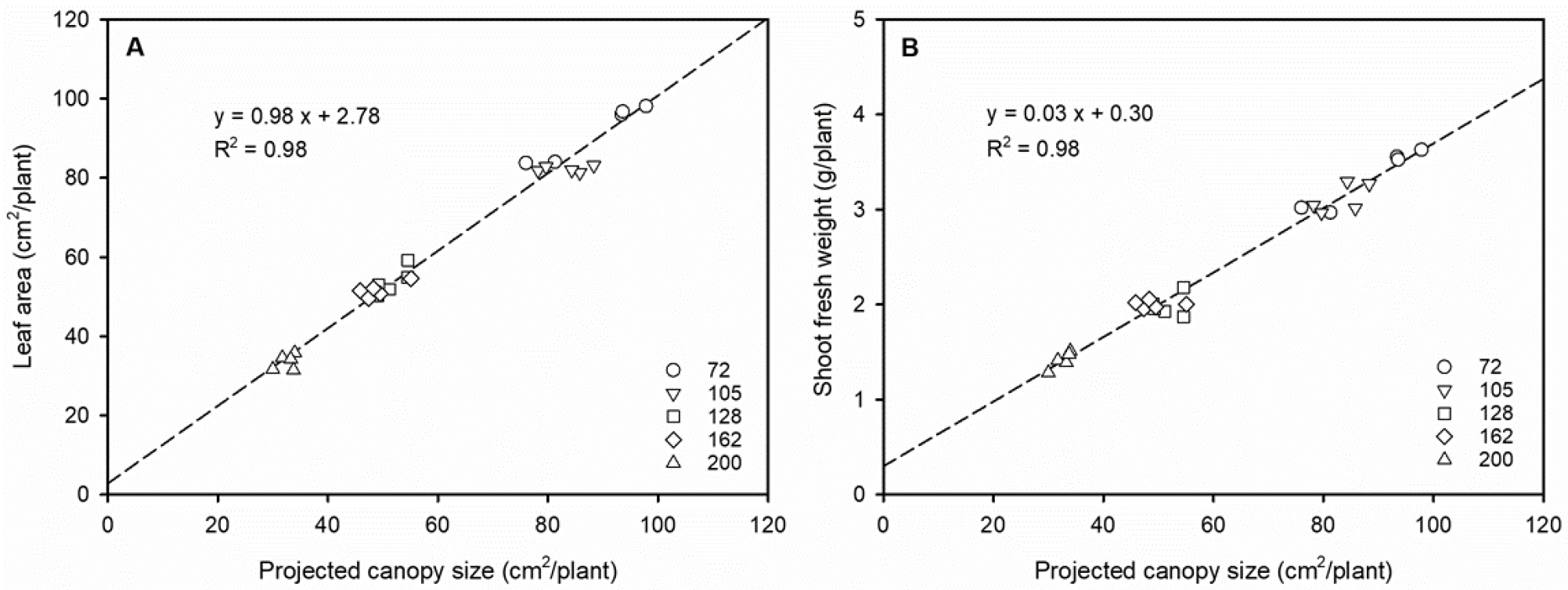
The correlation between PCS and growth parameters (leaf area and shoot fresh weight) was examined at 25 DAS (Fig. 2). PCS exhibited a strong correlation with both leaf area and shoot fresh weight (R2 = 0.98), indicating that these parameters can be estimated with high accuracy from image analysis. To further assess the applicability of PCS for predicting seedling growth, kimchi cabbage seedlings were classified into three grades according to PCS values (Table 2). Grade A was defined as PCS ≥ 70 cm2 per plant and included seedlings grown in 72- and 105-cell trays. Grade B corresponded to PCS values of 50-70 cm2 per plant, encompassing seedlings from 128- and 162-cell trays. Grade C was assigned to seedlings in 200-cell trays with PCS < 50 cm2 per plant.
Table 2.
Projected canopy size (PCS) and predicted shoot fresh weight of kimchi cabbage seedlings classified into three grades.
| Grade |
PCSz (cm2/plant) |
Predicted shoot fresh weighty (g/plant) |
| A | 70 ≤ X | 2.4 ≤ Y |
| B | 50 ≤ X < 70 | 1.8 ≤ Y < 2.4 |
| C | X < 50 | Y < 1.8 |
Previous studies have demonstrated that plant growth can be non-destructively estimated using canopy area extracted from images. For instance, digital canopy area obtained through image processing showed a strong correlation with leaf dry weight in sugar beet (Joalland et al., 2016). During the early growth stage (6-26 DAS), the projected area from maize RGB images correlated strongly with leaf area, shoot fresh weight, and shoot dry weight (Ge et al., 2016). In lettuce cultivars with green leaves, PCS was highly correlated with shoot fresh weight from 32 days after germination (Kim and van Iersel, 2022). Similarly, the leaf fresh weight of Chinese chives was modeled as a power function of the photographic leaf area index, enabling non-destructive estimation of growth (Nomura et al., 2022).
The accuracy of growth estimation can also depend on image position and crop morphology. Ma et al. (2020) reported that projected leaf area extracted from hyperspectral side-view images of maize had a stronger correlation with shoot fresh weight than top-view images. In this study, PCS values from top-view images at the seedling stage of kimchi cabbage accurately predicted leaf area and fresh weight, demonstrating its utility as a non-destructive growth indicator. Unlike maize, which shows substantial height increase, kimchi cabbage exhibits limited vertical growth and broad canopy expansion, reducing bias in top-view imaging. Thus, PCS is particularly suitable for growth prediction in leafy vegetables where horizontal expansion dominates seedling development.
Seedling grading is an essential process in commercial nurseries to improve the efficiency of grafting and transplanting. However, conventional classification is time-consuming and labor-intensive, relying heavily on expert experience (Tong et al., 2013). To address this limitation, rapid and accurate image-based classification techniques have recently been developed for seedling quality evaluation. For example, muskmelon and pumpkin seedlings were graded based on stem diameter extracted through image processing, achieving a high classification success rate (Tian et al., 2017). Leaf area estimated from RGB-D images of artichoke seedlings was associated with quality labels and used to construct a grading system (Otoya and Gardini, 2021). More recently, with the integration of machine learning and deep learning, seedling grading systems have further advanced (Xiao et al., 2019; Yuan et al., 2025).
3. Growth of kimchi cabbage after transplanting by seedling grade
The growth of kimchi cabbage after transplanting was evaluated according to plug tray cell number and PCS-based seedling grade (Figs. 3, 4). Up to 21 DAT, growth increased with larger cell volume, consistent with the trends observed during the seedling stage. By 35 DAT, seedlings grown in 72-cell trays exhibited the greatest growth. However, at 49 DAT, only seedlings classified as grade C from the 200-cell trays showed delayed growth, whereas no significant differences were observed between grades A and B (72-162-cell trays).
The growth and quality of vegetable seedlings strongly influence subsequent growth and yield after transplanting (Gallegos‐Cedillo et al., 2024; Jeong et al., 2020). Lee et al. (2012) reported that paprika seedlings with superior growth characteristics achieved better initial growth and earlier harvest after transplanting. Similarly, when cucumber seedlings were cultivated under different environments, those produced in plant factories exhibited the highest seedling quality and the most rapid post-transplant growth (Hyeon et al., 2024). Korean melon seedlings with a low T/R ratio and high compactness also showed greater shoot growth and higher flowering rates during the early stages after transplanting (Lee et al., 2016).
In the present study, seedlings with greater initial size and growth exhibited faster early growth after transplanting. However, these differences tended to diminish over time. Similar trends have been reported in lettuce (Jeong et al., 2024) and tomato seedlings (Bae et al., 2025). In the early stages after transplanting, seedlings with greater initial growth exhibited faster growth. However, as the cultivation period progressed under favorable conditions, growth differences among seedlings above a certain quality threshold became less pronounced. By contrast, seedlings below this threshold continued to show delayed growth throughout the cultivation period. Therefore, selecting kimchi cabbage seedlings above a defined threshold (PCS ≥ 50 cm2 per plant in this study) through image-based, non-destructive analysis could contribute to stable production after transplanting. Such standards may be further refined and their accuracy improved with the application of machine learning and advanced modeling algorithms.
Conclusion
To produce kimchi cabbage seedlings of different sizes and growth levels, seedlings were grown in plug trays with varying cell numbers, and larger cell volumes resulted in greater seedling growth and size. Image analysis of seedlings with diverse growth characteristics confirmed that PCS values could accurately predict leaf area and fresh weight. Based on PCS, seedlings were classified into three grades, and selecting those above a defined threshold ensured stable growth after transplanting. These findings suggest that PCS-based grading provides a practical and non-destructive approach for selecting robust seedlings, thereby improving efficiency in commercial nursery operations.