Introduction
Although silicon is the second most abundant element both on the surface of the Earth’s crust and in soils, but it has not yet been listed among the essential elements for higher plants. However, the beneficial role of silicon in stimulating the growth and development of many plant species has been generally recognized (Epstein, 1999; Liang, 1999; Ma, 2004; Liang et al., 2005). Silicon is also known to effectively mitigate various biotic and abiotic stresses. Silicon enhances resistance of plants to diseases and suppresses insect pests (Seebold et al., 2001; Fauteux et al., 2005). Silicon plays an important role in alleviating various abiotic stresses such as salt stress, metal toxicity, drought stress, radiation damage, nutrient imbalance, high temperature, freezing (Ma, 2004).
Chinese cabbage is one of the most important vegetable crops with world-wide production (Lee et al., 2010). In Korea its estimate annual production was 2.93 millions of tons (Cheigh and Park, 1994). Chinese cabbage is using as major raw material for Kimchi as well as preparing various foods. Chinese cabbage can be grown year-round by using the different varieties. Unfortunately, many environmental factors affect plant growth and development. Plants are able to modify their growth and physiology according to surrounding environment. The ability of plants to do this plays a key role in determining their tolerance to stress and their maintaining efficient growth (Murchie and Horton, 1997; Walters et al., 2003). Therefore, this study was conducted to investigate the effect of silicate fertilizer on growth, physiology and abiotic stresses tolerance of Chinese cabbage seedlings.
Materials and Methods
1. Plant material and growing conditions
Chinese cabbage (Brassica campestris L.spp. Pekinensis Pupr.) seeds ‘Asia Alpine F1’ (Asia seed Co., Ltd., Korea) were sown in the 128-cell plug trays (Bumnong. Co., Ltd, Korea) that were filled with commercial growing substrate (BM 2, Berger Group Ltd, Canada). Twenty-five days after sowing, the seedlings were transplanted to plastic pots (with top and bottom diameters of 10cm and 6.5cm, depth of 9.5cm and 8 bottom perforation) which filled with the commercial growing substrate. One week after sowing, seedlings were fertilized at overhead irrigation twice a week with Wonder Grow fertilizers (Chobi Co., Ltd, Korea). The experiment was carried out in a glasshouse at Kangwon National University from July to September 2014.
2. Silicate application and abiotic stress treatment
Effect of silicate concentrations on growth and physiology of Chinese cabbage seedlings was studied. Five silicate concentrations (8, 16, 32, 64, and 128mM, as ‘Keunson’ silicate acid fertilizer (Saturn Bio Tech Co., Korea) with the control (non-treatment) were applied to Chinese cabbage seedlings twice a week for 3 weeks. After the end of last treatment, seedlings were evaluated for physiology and growth characteristics. For abiotic stresses, seedlings were treated with high (50±1°C) and low (0±1°C) temperature. One day after the imposition of high and low temperatures, the leaf of cabbage seedlings were cut for analysis of the cell membrane integrity estimated by relative electrolyte leakage. Three days after imposition of stresses, seedlings were estimated heating and chilling injury index. On the other hand, for drought stress, twenty pots per treatment were irrigated with full water before transfer to the growth chamber, and then the irrigation was withheld until temporary wilting point was observed. Growth chamber conditions: relative humidity was maintained in 40-50%; light intensity was approximately 100μmol·m-2·s-1 provided by fluorescent lamps; temperature were set up 25±1°C.
3. Data collection and analysis
The number of leaf, leaf area, and leaf chlorophyll content were measured, leaf area (cm2) was measured by leaf area meter (Area meter, Delta-T, UK), leaf chlorophyll content was measured by using a chlorophyll meter (Minolta, SPAD-502, Japan). Fresh and dry weights of shoot and root were measured. Dry weight of shoot and root were taken through oven-dry method at 80°C for 72 hrs.
For the root morphology, the WinRHIZO Pro 2009c (Regent Instruments, Inc, Quebec, Canada) images analysis system was used, coupled with professional scanner Epson 10000XL (Seiko Epson Corporation, Nagano, Japan) according to Arsenault et al. (1995). The roots were detached from shoots and then placed in a tray (30x15x2 cm) with water and placed on the scanner. Scanned images were analyzed by the WinRHIZO program for the total root surface area, total root length, average root diameter, and number of root tips.
For physiological characteristics, stomatal diffusive resistance and transpiration rate were assessed with an LI-1600, steady state porometer (LIP) (LI-COR, Lincoln, Nebraska, USA) at 4th leaf from the top of 5 plants of each treatment. Data were collected between 11-13 hrs.
Relative ion leakage was also assessed by the electrolytes leakage from the leaf of ten plants for each treatment with similar sizes. Leakage of electrolytes was determined using conductivity meter (Mettle Toledo AG). The leaf segments (disks of leaves with d = 1 cm) were strictly washed, blotted dry, weighted and put in stopped vials filled with the exact volume of deionized water. The vials were then incubated for 2 hrs in the dark with continuously shaking and were then measured conduction (C1). The vials were heated at 80°C for 2 hrs and were measured conduction again (C2). The electrolyte leakage was expressed as a percentage of relative ion leakage, which was calculated according to the following equation (Zhao et al., 2007): Relative electrolyte leakage (%) = C1/C2 × 100.
Chilling and heating injury was assessed after treating 3 days at low and high temperatures, using the following scale: (1) 0~20% of leaf area damage; (2) 21~40% of leaf area damage, (3) 41~60% leaf area damage, (4) 61~80% of leaf area damage, and (5) 81~100% of leaf area damage.
Percentage of wilted plant in drought stress were calculated when 75% of leaves per seedling withered.
The experiment was arranged in completely randomized design. For the statistical analysis of growth and physiology parameters, ten seedlings per treatment were randomly selected. Data were analyzed using SAS v.9.3 software (SAS Institute Inc., Cary, NC, USA). Mean separations were calculated using Duncan’s multiple range tests at P ≤ 0.05.
Results
1. Effect of silicate on growth and physiology characteristics of Chinese cabbage seedlings
Plant growth parameter responses to different concentrations of silicate are given in Table 1. Compared to control, the growth parameters such as number of leaves, leaf area significantly increased in 8mM treatment, but they were not statistically different with 16, 32 and 64mM treatments. The unsatisfactory value of growth parameters was performed in 128mM treatment. Leaf chlorophyll value was advancement in all treatments. Silicate treatments notably improved fresh and dry weight of shoot and root compared with control. The fresh weight of shoot and root significantly increased in 8, 16 and 32mM treatment, but it obviously decreased in 128mM treatment. There was a similar result of shoot dry weight in 16, 32, 64mM treatments except 8mM. Dry weight root remarkably increased in 8, 16, and 32mM of silicate treatments. The lowest value of fresh and dry weight of shoot and root were obtained in 128mM of silicate treatment.
Table 1.
Effect of silicate concentrations on growth characteristics of Chinese cabbage seedlings.
| Silicate concentrations (mM) | Number of leaf | Leaf chlorophyll value (SPAD) | Leaf area (cm2) | Fresh weight (g) | Dry weight (g) | ||
|---|---|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | ||||
| Control | 11.2 bz | 26.8 c | 422.3 b | 22.65 c | 1.18 c | 1.42 b | 0.15 b |
| 8 | 12.3 a | 29.0 b | 477.0 a | 26.38 a | 1.30 b | 1.61 a | 0.17 a |
| 16 | 11.5 b | 30.2 b | 449.3 b | 24.85 b | 1.50 a | 1.45 b | 0.17 a |
| 32 | 11.3 b | 29.6 b | 433.8 b | 23.53 b | 1.47 a | 1.46 b | 0.17 a |
| 64 | 11.3 b | 30.5 b | 419.0 b | 22.18 c | 1.12 c | 1.44 b | 0.15 b |
| 128 | 10.3 c | 32.0 a | 375.0 c | 18.89 d | 1.03 d | 1.20 c | 0.14 c |
There was an expanded value of the total root surface area, total root length, and number of root tips in 8, 16 and 32mM treatments, but it did not show in 64 and 128mM treatments. The highest of total root surface area, total root length, and number of root tips was also obtained in 8mM of silicate treatment (Table 2).
Table 2.
Effect of silicate concentrations on root morphology of Chinese cabbage seedlings.
| Silicate concentrations (mM) | Total root area (cm2) | Total root length (cm) | Average root diameter (mm) | Number of root tips |
|---|---|---|---|---|
| Control | 92.8 cz | 446.2 cd | 0.67 ab | 2575.5 bc |
| 8 | 112.2 a | 535.5 a | 0.71 a | 3141.3 a |
| 16 | 108.0 ab | 509.1 ab | 0.69 ab | 2866.5 ab |
| 32 | 100.3 bc | 472.5 bc | 0.67 ab | 2589.0 bc |
| 64 | 82.2 d | 392.7 d | 0.64 ab | 2254.7 c |
| 128 | 73.9 e | 318.9 e | 0.61 b | 2198.8 c |
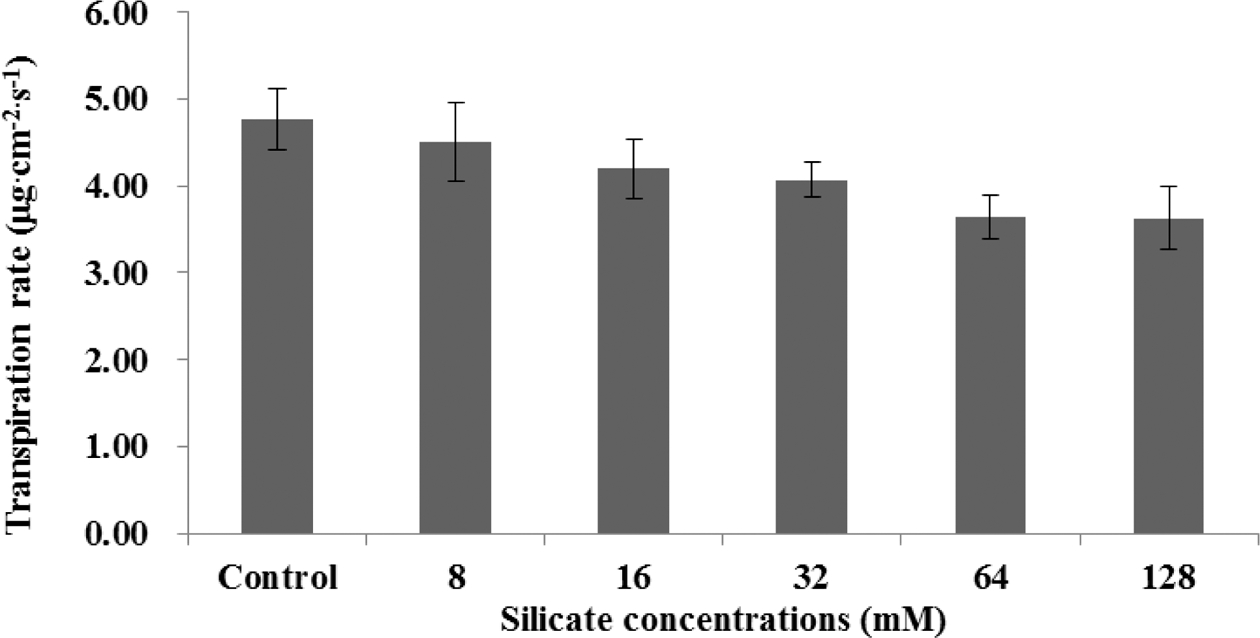
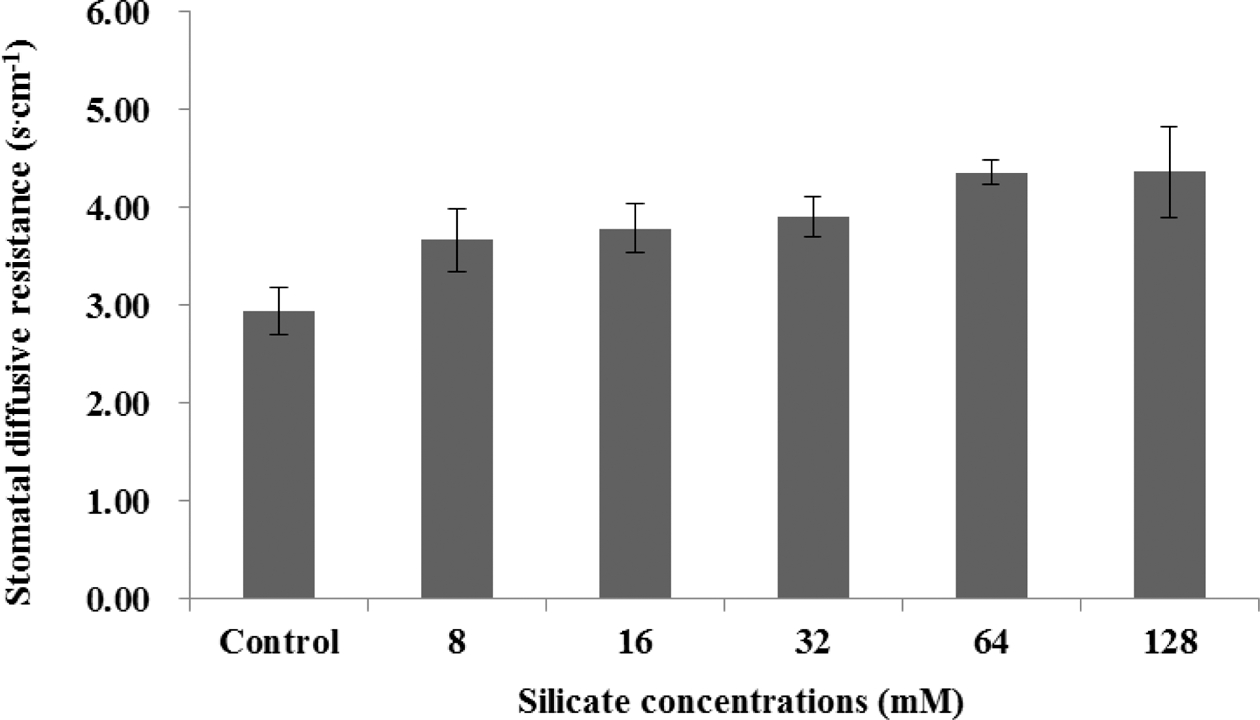
Transpiration rate and stomatal diffusive resistance had a vice versa relationship to treatments application including control. Transpiration rate was higher in control treatment compared to silicate treatments and it had decreased trends with increasing silicate concentration (Fig. 1). On the other hand, stomatal diffusive resistance increased with increasing silicate concentration (Fig. 2).
2. Effect of silicate on abiotic stress of Chinese cabbage seedlings
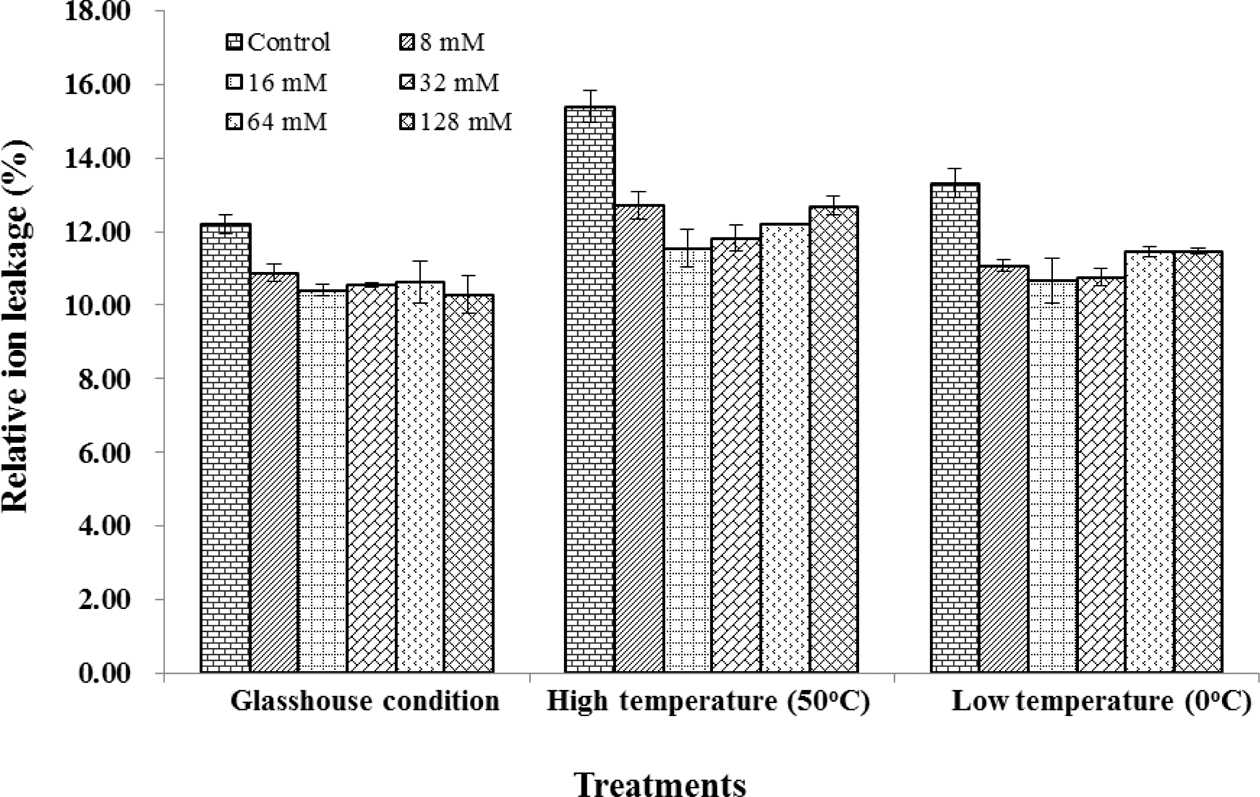
Electrolyte leakage is another component connected with different stresses. In this study we found that silicate treatments led to significantly decrease in the electrical conductivity under all conditions (glasshouse, high and low temperature) compared to control. The values of relative ion leakage of all treatments in high temperature were higher than those in low temperature condition. Similar values of relative ion leakage were observed in all silicon treatments in the glasshouse condition, but they were found significantly different among silicate treatments at high and low temperature conditions. The lowest value of relative ion leakage was observed in treated seedlings with 16mM of silicate at both high and low temperatures. However, relative ion leakage increased with increasing silicate concentration over 32mM (Fig. 3). The application of silicate also reduced the heating and chilling injury index at high and low temperatures by reducing the damage to the leaf. The low values of heating or chilling injury were observed in treated seedlings with 16 and 32mM of silicate concentration (Table 3).
Table 3.
Effect of silicate concentrations on heating and chilling injury index of Chinese cabbage seedlings (measured at 3 days after treating in high (50±1°C) and low (0±1°C) temperatures).
| Silicate concentrations (mM) | Heating injury index | Chilling injury index |
|---|---|---|
| Control | 3.3 a | 3.5 a |
| 8 | 3.0 b | 2.5 c |
| 16 | 2.0 d | 1.2 d |
| 32 | 2.2 d | 1.5 d |
| 64 | 2.5 c | 2.4 c |
| 128 | 2.6 c | 2.7 b |
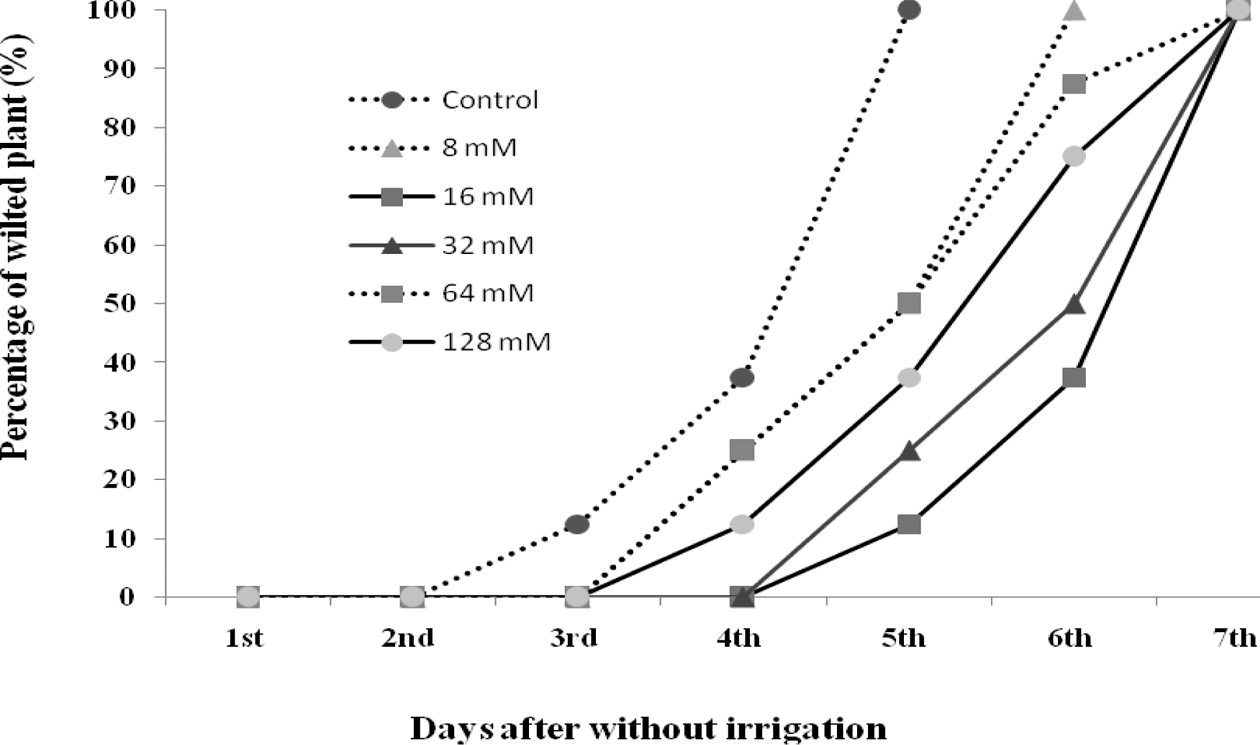
Silicate enhanced drought tolerance of Chinese seedlings by delaying the starting time of wilting point. The starting time of wilting point in the control was observed 3 days after discontinuation of irrigation, while in the 8, 64 and 128mM of silicate treatments the starting times were 4 days, and the 16 and 32mM of silicate treatments the starting times were 5 days. In the control, 100% of wilted plants were observed after 5 days without irrigation, while in the 8mM of silicate treatment after 6 days, in the 16, 32, 64, 128mM of silicate treatments after 7 days (Fig. 4).
Discussion
In this experiment growth parameters significantly increased in treated seedlings with 8, 16 and 32mM of silicate compared to control. The highest growth parameters were observed in treated seedlings with 8mM of silicate. Although silicon has not been considered as an essential element for higher plants, nevertheless, application of silicon can improve the growth and yield of various crops (Adatia and Besford, 1986; Anderson et al., 1987; Edward et al., 1982). Gong et al. (2003) observed that silicon increased plant height, leaf area and dry mass of wheat even under drought. Singh et al. (2006) also suggested that silicon application increased dry matter and yield in rice. In addition, silicon has also been proved to be beneficial for the healthy growth and development of many plant species, particularly graminaceous plants such as rice and sugarcane and some cyperaceous plants (Epstein, 1999; Liang et al., 2005). However, growth parameters of Chinese cabbage seedlings decreased in 128mM of silicate treatment.
Transpiration rate decreased and stomatal diffusive resistance increased with increasing silicate concentrations. This result agreed with results of Lu and Cao, (2001) who reported that melon (Cucumis melo L.) showed benefits of Si supplementation with higher chlorophyll levels and reduced transpiration rates compare to untreated plants. Gao et al. (2006) found that silicon application of 2mM significantly decreased transpiration rate and conductance for both adaxial and abaxial leaf surface, but had no effect on transpiration rate and conductance from the cuticle in corn subjected to polyethylene glycol osmotic stress in solution culture. However, in our study transpiration rate had declined trends with increasing silicate concentration. This finding may have been due to an excessive silicon supply, causing the formation of silicate polymers on root surfaces.
Marschner et al, (1990) also pointed out that silicon enhances freezing tolerance of many crops. In this study, we found silicate treatments led to significantly decrease in the electrical conductivity under glasshouse, high and low temperature conditions. Silicate could migrate to reduce heating and chilling injury index during high and low temperature by reducing damage of leaf. At two levels of temperature, chilling injury index values of seedlings in silicate treatments were lower than those in non- silicate treatment. The low value of heating or chilling injury was observed in treated seedlings with 16 and 32mM of silicate. On the other hand, silicate enhanced drought tolerance of Chinese seedlings by delaying the starting time of wilting point. This result agreed with results of Trenholm et al. (2004) who reported that under severe drought stress, silicon-amended plants had better responses than non-amended plants and little improvement was seen under moderate drought stress.
In conclusion, low concentration of silicate stimulated the growth of Chinese cabbage seedlings by increasing growth parameters and root morphology. Transpiration rate decreased and stomatal diffusive resistance increased with increasing silicate concentrations. Silicate could migrate to reduce heating and chilling injury index during high and low temperature by reducing relative ion leakage and damage of leaf. On the other hand, silicate enhanced drought tolerance of Chinese seedlings by delaying the starting time of wilting point.