Introduction
Lettuces and Chinese cabbages can be grown successfully either in spring or fall. However, there is a risk of environmental stress associated with exposing young plants to cold weather after field setting (Palada et al., 1987). It is well known that temperature and CO2 are the two conclusory factors controlling the growth of plants (Berghage, 1998;Heins et al., 2000). Lettuce and Chinese cabbage species has an optimum temperature for rapid growth rate, however lower temperatures below the optimum range allow the plant to grow, but at a considerably reduced rate (Kalisz and Cebula, 2006). Nam et al. (1995) reported that the response of plants to unfavorable temperatures results in a modification of many physiological and biochemical processes leading to change in the chemical composition. The degree of these changes is mainly dependent on the temperature level, the temperature exposure duration and the stage of plant development. It is found that seedling stage of Chinese cabbage generally more sensitive to unfavorable thermal conditions than more developed plants (Daly and Tomkins, 1995). Low temperatures have been reported to reduce the growth of Chinese cabbage seedlings (Wiebe, 1990) and to influence their chemical composition (Moe and Guttormsen, 1985;Sasaki et al., 1996). Likewise temperature, elevated CO2 may directly or indi-rectly have an effect on plant production and development. It has been observed that exposure of plants to elevated CO2 resulted in a higher biomass production, an altered morphology, increased water use efficiency, increased photosynthetic activity and an enhanced carbohydrate content (Kimball, 1983;Cure, 1986;Smith et al., 1987;Lawlor and Mitchell, 1991;Woodward et al., 1991). However, the interspecific variation in response to elevated CO2 would be related to the morphology and strategy of different plant species (Poorter, 1993; Loehle, 1995). Differences in growth rate, life span and sink strength between different species have been shown to influence the effect of CO2 (Stulen et al., 1994).
Therefore, with growing concerns of controlling the factors (temperature and CO2) in greenhouse for crop production, DME burner using DME gas may be one alternative technique in sustainable agricultural practice. DME which is clean and considered to be economically alternative fuel is produced from natural gas through synthesis gas (Semelsberger et al., 2005). It is proved that DME and LP gas has the almost similar properties (Ogawa et al., 2003). Nowadays, DME is used widely in various fields as a fuel such as power generation, transportation, home heating and cooking, etc. (Bhattacharya et al., 2013;Olah et al., 2009;Arcoumanis et al., 2008;Kim et al., 2008;Marchionna et al., 2008). In this study, DME gas was used as a fuel for DME burner to increase temperature and CO2 concentration in greenhouse for lettuce and Chinese cabbage cultivation.
DME gas is a conversion form synthesis gas. The synthesis gas is transformed into DME through following twostep synthesis reactions (EBTP, 2011;Yuan and Eden, 2016). DME gas is also known as unpolluted fuel by burning it in agriculture facility, it is not only provide heating but also provide sufficient CO2 without any high concentration of toxic gases.
| $$2{\mathrm H}_2+\mathrm{CO}\leftrightarrow{\mathrm{CH}}_3\mathrm{OH}\left(\triangle\mathrm H0=-90.56\mathrm{kJ}\;\mathrm{mol}^{-1}\right)$$ |
| $$2{\mathrm{CH}}_3\mathrm{OH}\leftrightarrow{\mathrm{CH}}_3{\mathrm{OCH}}_3+{\mathrm H}_2\mathrm O\left(\triangle\mathrm H0=-49.43\mathrm{kJ}\;\mathrm{mol}^{-1}\right)$$ |
| $$\mathrm{CO}+{\mathrm H}_2\mathrm O\leftrightarrow{\mathrm{CO}}_2+{\mathrm H}_2\left(\triangle\mathrm H0=-41.12\mathrm{kJ}\;\mathrm{mol}^{-1}\right)$$ |
To identify optimal amount of DME combustion gas supply to greenhouse, it is essential to quantify the complex crop–climate and phenology analysis. Many researches have been conducted to evaluate the process and application of DME for different sectors (Zhao et al., 2011;Zhang et al., 2016;Liu et al., 2013;Sun et al., 2014). However, a few number of experiments have been conducted to evaluate the impacts of DME on crop yield, especially in greenhouse where crops in winter season face low temperature stress (Basak et al., 2018;Qasim et al., 2018). Therefore, the objectives of this study were to (i) evaluate the changing pattern of temperature and CO2 concentration applying DME in a control greenhouse system and (ii) determine the impacts of DME on chlorophyll content and fresh and dry weight of lettuce and Chinese cabbage.
Materials and Method
1. Experimental Design
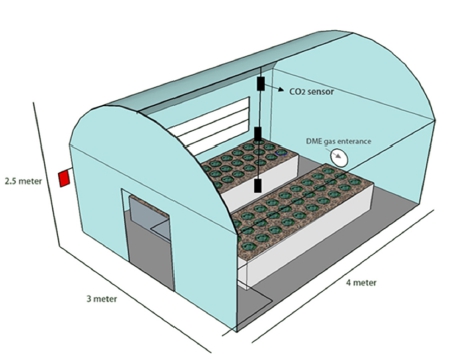
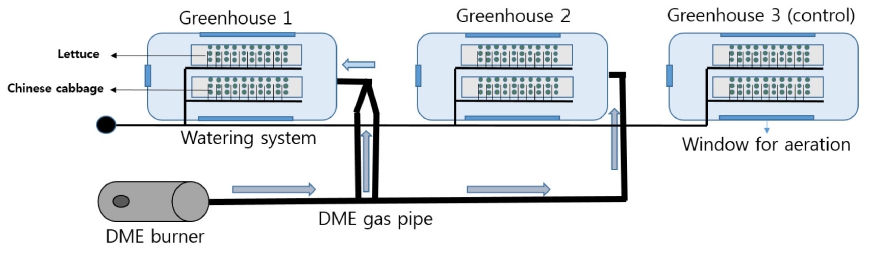
This experiment was performed in three identical greenhouses (width 3 m, length 4 m, height 2.5 m) of Gyeongsang National University (Fig. 1). In all 3 greenhouses, there were 2 beds for planting vegetables. In one bed, lettuce was grown and, in another bed, Chinese cabbage was grown. Three tests were performed to compare and analyze the growth of lettuce and Chinese cabbage and chlorophyll content in greenhouses at different flow quantity of DME. DME supply times were 00.5 hr day-1, 1 hr day- 1, 1:30 hrs day-1 and 2 hrs day-1 on week 1, 2, 3 and 4 respectively in every morning at 6 o’clock. The average DME flow quantity in duct was 17.4 m3 min-1 and 10.2 m3 min-1 to greenhouse-1 and greenhouse-2 and no DME gas was supplied to greenhouse-3 which was left as control (Fig. 2). For this experiment, lettuce and Chinese cab-bage seeds were purchased from market and seeds trays were used for seedling. After 3 weeks, 30 lettuce and 30 Chinese cabbage plants were transplanted to two beds of greenhouses.
The irrigation systems were uniform in all three greenhouses. Water was provided by needles to each separate plant. The irrigation time was 2 min day-1 in all greenhouses. For aeration side windows were opened at 9:00 am and closed at 6:00 pm to keep the temperature in range.
2. Properties of DME
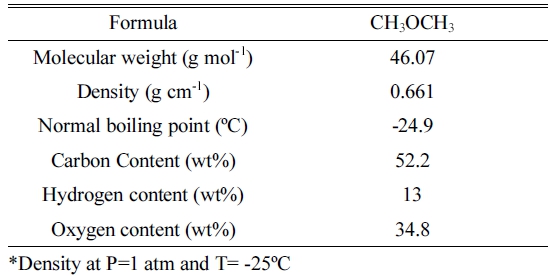
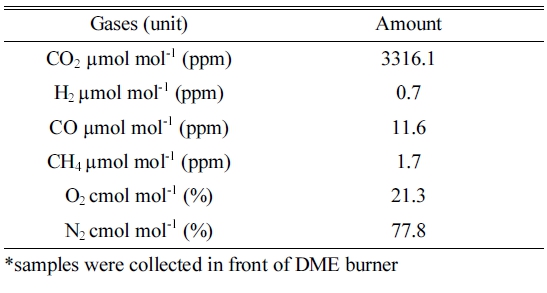
The physical properties of DME gas and the specification of DME burner are described in Table 1 and 2, respectively. For testing the concentration of gases, the samples were collected in air plastic bags (20 litter capacity) during operation of DME combustion. The sample was taken to Korean Testing laboratory on same day and ISO 6974-6 standard method was used for determination of gases concentration. The testing environment was (mean and S.D.) 19.8±5.20°C in temperature and 50.2±19.9% in relative humidity. Table 3 shows the concentration of gases during combustion of DME.
3. Data Collection and Analysis
The plants were periodically examined after the germination stage to observe the changing pattern of plant growth and DME treatment. For measuring carbon dioxide, temperature and humidity “Lutron MCH-383SD (Lutron Electronic Enterprise Co., Ltd., Taipei, Taiwan)” electrochemical sensors were set at three different height of greenhouses. Lutron MCH-383SD is equipped to record the date, time interval and has a memory card to store these data. Data were checked at 10-min intervals and recorded data were averaged for further analysis and interpretation. The lower sensor was equipped at height of 0.3m from floor which was equal with height of plants in bed.
Fresh weight and dry weight of leaves and roots of each plant in greenhouse-1, 2 and 3 were compared at end of the experiment. Fresh weight was measured as whole plant because of high number of leaves and size of lettuce. For Chinese cabbage, 5 big leaves from each plant were taken for measuring fresh and dry weight analysis. Roots were cleaned from soil and separated from whole plants for analysis. The fresh and dry weight of plant was estimated to use digital balance (model- FX-300iWP, A&D Company Limited, Tokyo, Japan) and drying oven (Shelves for 5EDHG6310: 2 Layers, Changsha Kaiyuan Instruments Co., Ltd, China). Number of papers were followed various temperature range and time for measuring the dry weight of plant (e.g., Arshadullah and Zaidi, 2007; Cho et al., 2007; Karimi et al., 2009). In experiment, where different treatments of plants were studied, dry weight of plant was measured to keep temperature at 80°C for 24 hour. Standard statistical methods were used for data evaluation including analysis variance with a significance level of P<0.05. The significance differences between mean values of experimental data were tested with a post-hoc Tukey’s HSD test. All statistical calculations were performed with Statistix10 and Statistical Package for the Social Sciences (SPSS Version: 22.0.0.0).
Results and Discussion
1. Temperature and CO2 pattern
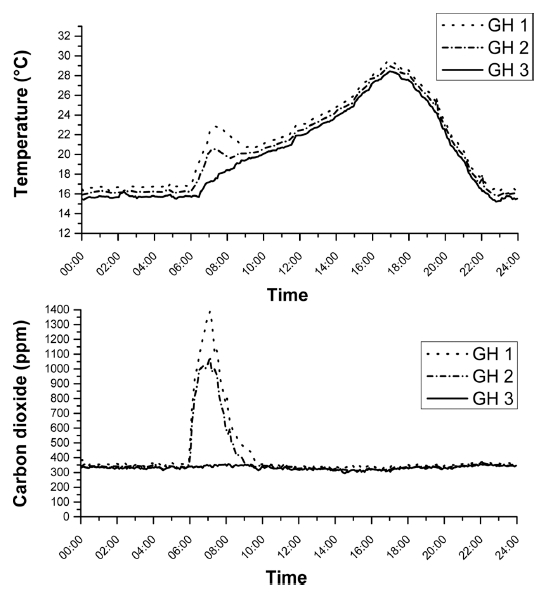
Environmental factors, like temperature and CO2 are important at all stages of plant growth of lettuce and Chinese cabbage. It was examined the changing pattern of temperature and CO2 concentration due to the different levels of DME applications. The result of the study showed that the higher levels of DME application accompanied with a greater diversity CO2 concentration were confirmed the increased of temperature in this period (Fig. 3). Even the application of DME in an amount of 10.2 m3 min-1 was efficient to increase temperature and CO2 content in greenhouse-2. With 17.4 m3 min-1 and 10.2 m3 min-1 DME supply, the maximum CO2 were recorded 1380 ppm and 940 ppm in greenhouse-1 and greenhouse-2 respectively. Average 24 hours data on temperature and CO2 during experimental period showed that CO2 concentration was increased up to 265% and 174% treated with DME at 17.4 m3 min-1 and 10.2 m3 min-1 respectively compared to control condition in every morning between time intervals at 6 to 8 o’clock. Due to increase the level of CO2 in greenhouses in the same way of concentrations of DME, temperature increased up to 4.8°C and 3.1°C in greenhouse 1 and 2 respectively within this same time. On the basis of these results of changing different concentration CO2 and temperature were chosen to examine those variability chlorophyll content and fresh weight and dry weight of plant for further work. Similar results were reported by other researchers which showed that CO2 and temperature are constantly associated with the application of DME gas in greenhouse (Basak et al., 2018;Qasim et al., 2018).
2. Fresh and Dry weight of Lettuce and Chinese cabbage
Twenty five plants of lettuce and Chinese cabbage were collected from each greenhouse and measured the fresh and dry weight. The evaluation of the fresh and dry weight of lettuce and Chinese cabbage under different application rate of DME (DME-1, DME-2 and DME-3) in the experimental period is presented in Fig. 4 and Fig. 5. Fresh weights of lettuce were significantly different among the three treatments and time. It was maximum for DME-1 treatment compared to other two treatments. Mean (±SD) fresh weight in gram after 8 weeks were 76.1±9.7, 74.7±8.9 and 65.1±9.9 for DME-1, DME-2 and DME-3 respectively. The study found that the differences of fresh weight were significant (p<0.05) among DME-1, DME-2 and DME-3. In this current experiment, it was also examined that fresh weight increased up to 17.1% due to application DME at a rate of 17.4 m3 min-1 and 14.8% for 10.2 m3 min-1 compared to control treatment, thus revealing the increase in fresh weight of lettuce due to increase the amount of DME application. Moreover, due to growing time, dry weight increased at a substantial rate. It was also observed that the dry weight plant whose are directly related to fresh weight, did not keep the same rate within this experimental period. Dry weight values following declined gradually 5.01±0.65, 4.8±0.58 and 4.2±0.65 for DME-1, DME-2 and DME-3 respectively after 8 weeks. Thus, higher dry weight was obtained for the treatments, DME-1 and DME-2 whereas the minimum dry weight of plant was observed for DME-3.
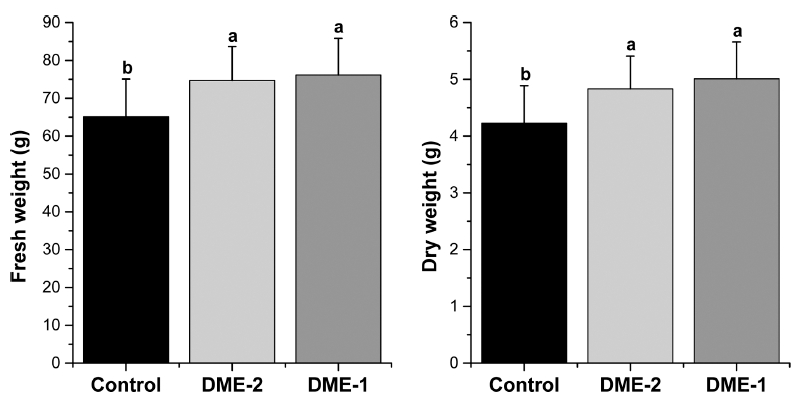
Fig. 4
Data represent means and standard deviation (SD) of fresh and dry weight of lettuce in 8th week in three treatments in greenhouses (n=25). Different letters above the bars (SD) denote significant differences of fresh and dry weight among treatments at p ≤ 0.05 based on Tukey’s HSD post-hoc test.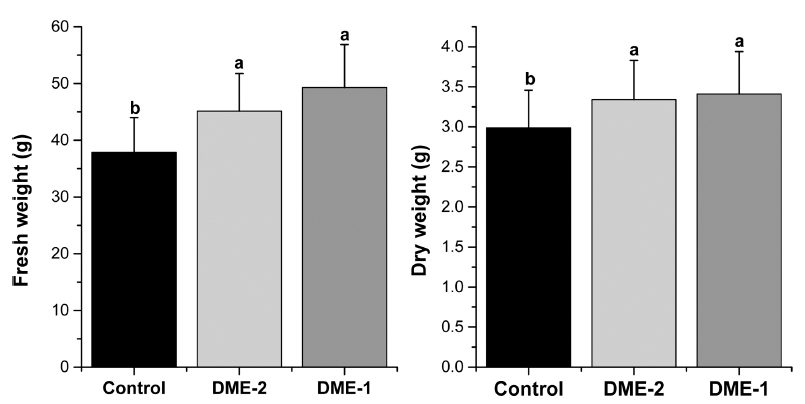
Fig. 5
Data represent means and standard deviation (SD) of fresh and dry weight of Chinese cabbage in 8th week in three treatments in greenhouses (n=25). Different letters above the bars (SD) denote significant differences of fresh and dry weight among treatments at p ≤ 0.05 based on Tukey’s HSD post-hoc test.Likewise, lettuce both different rates of DME application also affect the fresh and dry weight of Chinese cabbage. Five big leaves from each plant of Chinese cabbage were taken for measuring fresh weight and dry weight analysis. When DME was applied at a rate of 17.4 m3 min-1 in greenhouse, fresh and dry weight of Chinese cabbage markedly increased at a range of 30.1% and 14.1% respectively relative to the control treatment. In response to DME at a rate of 10.2 m3 min-1, fresh and dry weight increased up to 19.3% and 11.7% respectively. DME-1 and DME-2 treatments which caused rise in temperature and CO2 in greenhouse was found to have an effect on the fresh and dry matter of lettuce and Chinese cabbage seedlings. Temperature of 18 to 22°C was optimum for Chinese cabbage heading and 10 to 13°C for the final head development, with growth reduction occurring when temperature is below 10°C (Burt et al., 2006). According to Moe and Guttormsen (1985), the increase of temperature in the growing stage resulted in higher dry matter of Chinese cabbage seedlings. A marked increase of fresh matter in Chinese cabbage grown at higher temperatures also observed by Noto and Leonardi (1995). On the other hand, the reduction in growth of cabbage seedlings held in low temperature was observed by Sasaki et al. (1996). Wiebe (1990) mentioned that the number of leaves in Chinese cabbage seedlings raised clearly with higher temperature compared with those subjected to low temperature. Likewise, temperature elevated CO2 may directly, or indirectly, effect on plant production and development. It has been noted that exposure of plants to elevated CO2 resulted in a higher biomass production and an enhanced carbohydrate content (Lawlor and Mitchell, 1991;Woodward et al., 1991). Similar effect was described by Gray (2015) for lettuce plant. The results obtained in this study indicated that DME application during growing stage were more beneficial for plant growth.
3. Chlorophyll content in Lettuce and Chinese cabbage
The chlorophyll content results are presented as an average of the measurement in all 3 greenhouses (Fig. 6). DME application levels had a significant (p<0.05) effect chlorophyll content in lettuce and Chinese cabbage. The mean values of chlorophyll content in lettuce were recorded 36.48±3.7, 33.7±4.8 and 30.4±3.1 for DME-1, DME-2 and DME-3 respectively at the end of 8th week. However, the mean values of chlorophyll content in Chinese cabbage were obtained 59.1±4.5, 49.5±4.9 in DME-1 and DME-2 respectively where it was 40.8±4.8 for DME-3. Thus, chlorophyll content was obtained high for the treatments DME- 1 and DME-2 whereas the minimum chlorophyll content was observed for DME-3. So from these results it is suggested that DME supply to greenhouse occurred high CO2 can be effective for chlorophyll content of leaves. CO2 being the substrate of photosynthesis, the increase in CO2 concentration increases the net photosynthetic rate and crop productivity (Kimball, 1983;Ziska et al. 1997). In reviewing a number of studies of the effects of CO2 on enrichment of leaf chlorophyll content (Evans, 1989;Gifford, 1992;Houpis et al., 1988;Idso and Kimball, 1992;Idso et al., 1991;Idos et al., 1993). Pinter et al. (1994) observed slightly higher per-unit-leaf-area chlorophyll contents in plants exposed to elevated CO2. In another experiment with alfalfa, for example, plants grown at an atmospheric CO2 concentration of 600 ppm actually displayed greater leaf chlorophyll concentrations than those observed in plants grown at 340 ppm (Sgherri et al., 1998).
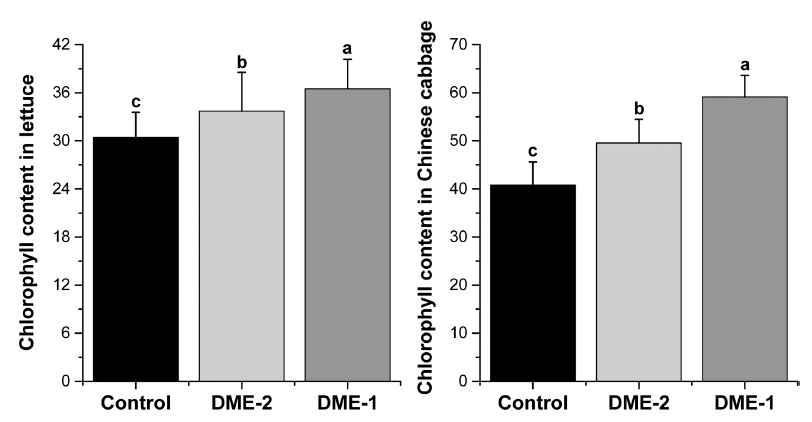
Fig. 6
Data represent means and standard deviation (SD) of chlorophyll content of lettuce and Chinese cabbage in 8th week in three treatments in greenhouses (n=25). Different letters above the bars (SD) denote significant differences of chlorophyll content among treatments at p ≤ 0.05 based on Tukey’s HSD post-hoc test.Conclusions
The current study demonstrates the effects of DME combustion gas on fresh weight, dry weight and chlorophyll content in lettuce and Chinese cabbage, which are important factors for higher yields. Higher concentration of CO2 generated by DME combustion gas in greenhouse showed higher efficiency on fresh weight, dry weight and chlorophyll content of plant. The result of the study showed that, fresh and dry weights of lettuce and Chinese cabbage in greenhouse-1 obtained from DME-1 (17.4 m3 min-1) treatment were higher compared to other two treatments (DME- 2 and DME-3). Moreover, the contents of chlorophyll were better in greenhouse-1 and 2 as compared to control greenhouse. To improve the yield, in response to the above mentioned three parameters, DME gas usage is important in developing new decision making by farmers and researchers for its application under extreme cold condition in greenhouse. Moreover, this study creates a scope for further research using DME combustion gas in greenhouses to measure growth performance of different crops. In addition, it is essential to quantify the accurate rate of DME combustion gas for a specified crop, which can be helpful to improve the plant growth and enhance yield.