Introduction
Light is an essential energy source that acts as a signal for survival and reproduction for plants. Since 89% of greenhouse areas rely only on sunlight, the yields and quality of products change according to drastic fluctuations in light intensity and day length with season (Jeong et al., 2009a). Low irradiation can lead to early falling flowers and fruits or irregular fruit settings (Heuvelink and Koner, 2001;Kim et al., 2011). Consequently, the yield and quality of greenhouse crops decrease during winter season in middle-latitude regions such as Korea or high-latitude regions such as the Netherlands. To overcome the low light intensity in winter season, high-pressure sodium lamps have been used to compensate the lack of light and to increase the yield of fruit vegetables such as cucumber, tomato, and paprika in Europe (Hao and Paadopoulos, 1999;Aistė at al., 2015;Hao at al., 2016).
In particular, the prevalent cropping season of paprika starts from the end of August and ends in June of the next year in Korea. Therefore, low light conditions from October to February induce longer harvest days after fruit setting, lower fruit setting rates, and higher incidence of malformed fruits at longer cloudy days. Since the fruit setting rate of paprika is susceptible to the change in the amount of light when light intensity widely fluctuates (as in Korea), low irradiation decreases productivity of paprika (Jeong et al., 2009b). Many studies indicated that supplemental lightings improved the productivity and quality of crops (Hogewoning et al., 2010a, 2010b;de Visser et al., 2014;Cocetta et al., 2017). Particularly in fruit vegetables, such as tomato, cucumber, and paprika supplemental lighting can be an essential method for compensating the deficit of sunlight (Kim et al., 2011;de Visser et al., 2014;Hernández and Kubota, 2014;Guo et al., 2016b). Recently, various supplemental lights, such as light-emitting diode and high-pressure sodium lamp, have been used for greenhouse crop production (Gomez et al., 2013;Kim et al., 2013;Lee et al., 2014). Because individual wavelengths differently affect the production and quality of fruits (Li and Kubota, 2009;Hogewoning et al., 2010b;Hikosaka et al., 2013;Guo et al., 2016a;Hao et al., 2016;Yang et al., 2017), it is important to choose appropriate light sources for crop production.
For greenhouse cultivation, numerous studies have focused on the enhancement effects of supplemental lighting on the productivity and quality of fruits at low-light seasons. However, high-pressure sodium lamps have been mostly used due to the high efficiency of PAR spectrum (400-700nm) in greenhouse and the low cost (Dorais et al., 1991;Demers et al., 1998), while LED was often applied to intra-canopy lighting due to its low output (Trouwborst et al., 2009;Gomez et al., 2013). Hogewoning (2010a) reported that an artificial solar lamp using plasma lamp which closely resembles sunlight spectrum showed higher growth in cucumbers. As artificial lamps with a light spectrum similar to sunlight, plasma lamps were used for cucumbers and tomatoes at seedling and vegetative stages in greenhouses (Hogewoning et al., 2012). Therefore, plasma lamps should be applied to other crops except cucumber until harvest stage for evaluation of its effectiveness. The objective of this study was to compare the supplemental lighting effects of sulfur plasma and high pressure sodium lamps on the growth, productivity, and physiological characteristics of paprika in growth chamber and greenhouse.
Materials and Methods
1. Plant and Cultivation Conditions
Seeds of paprika (Capsicum annuum L. ‘Cupra’) were sown on 25 September, 2015 and transplanted on 8 December, 2015 for greenhouse experiment and were sown on 27 March, 2016 and transplanted on 21 April, 2016 for growth chamber experiment. Nutrient solutions were maintained at an electrical conductivity of 2.5 ~ 3.0 dS·m-1, a pH of 5.8 ~ 6.0, and an air temperatures of 25°C/18°C (day/night) with ventilation and heating in greenhouses. Nutrient solutions of Dutch PBG were composed as N 12.75, P 3.75, K 6.0, Ca 7.5, and Mg 2.5 mg·L-1. The experiment was conducted in a Venlo-type greenhouse and growth chambers at National Institute of Horticultural and Herbal Science of Rural Development Administration (RDA) located at Haman, Korea.
2. Spectral Characteristics of Supplemental Lights
A 1000 W high-pressure sodium lamp (HPS; E-papillon 1000 W, Light Interaction, Eindhoven, Netherland) and 1030 W sulfur plasma lamp (SP; SPLS-1000, LG Electronics, Seoul, Korea) were used as supplemental light sources. Portions of ultraviolet (UV, 300-400 nm), photosynthetic photon flux density (PPFD, 400-700 nm), and near infrared (NIR, 700-1100 nm) lights in HPS and SP were measured at the vertical distance of 4 m from the light sources by using a radio spectrometer (Li-1800, Li-COR, Lincoln, NE, USA). R/FR ratio was obtained by dividing the portion of red light (655-665 nm) to far-red light (725-735 nm) by Smith’s definition (1982).
3. Growth Index and Photosynthesis at HPS and SP in Growth Chamber
To identify the effect of HPS and SP on paprika growth and development, cultivation experiments were conducted in two growth chambers (L 1.6 m × W 1.4 m × H 1.8 m, DS-59M, Dasol, Hwaseong, Korea) with each two HPSs and two SPs at a photoperiod of 14 h/10 h (day/night), respectively. Air temperature and relative humidity were maintained at 25°C and 50% during the day and at 18°C and 50% at night, respectively. The maximum leaf length, plant height, number of nodes, diameter of stem, SPAD, and leaf area were measured on 27 June, 2016; in addition, fresh and dry weights were measured every two weeks. At 8 weeks after transplanting in the growth chamber, photosynthetic rates and leaf temperature were measured by using a photosynthesis measuring apparatus (Li-6400, Li- COR, Lincoln, NE, USA). The temperature and relative humidity in the leaf chamber were maintained at 25ºC and 45-55%, respectively. Net CO2 assimilation rate as a function of light curves was determined at each step every 3-4 min. Variable PPFDs were beamed from an internal LED light source ranging from 0 to 1500 μmol·m-2·s-1 at a CO2 concentration of 400 μmol·mol-1.
4. Growth Index under Supplemental Lights in Greenhouses
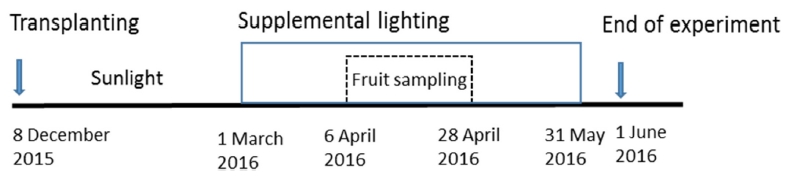
The greenhouses (W 9.6 m × L 45 m × H 5.5 m) were divided to three plots for comparative experiments: with the sunlight with two supplemental lighting treatments of Sun + HPS and Sun + SP, and the sunlight without supplemental lighting (Sun). Supplemental lighting started from 1 March to 31 May, 2016 and fruits were sampled from 6 April to 28 April, 2016 in order to reflect the pure effect of supplemental lighting (Fig. 1). From 07:00 to 21:00, the supplemental lights installed at the height of 4 m from the ground turned on when outside solar radiation was below 104.2 W·m-2 (in the average of 10 min) until 1 June, 2016. From 07:00 to 21:00 in a day, supplemental lights in the greenhouses turned on for about 3 hours a day (1 hour in the morning and 2 hours in the evening) on a sunny day and about 14 hours a day on a rainy or cloudy day under the outer irradiation of 100 W·m-2.
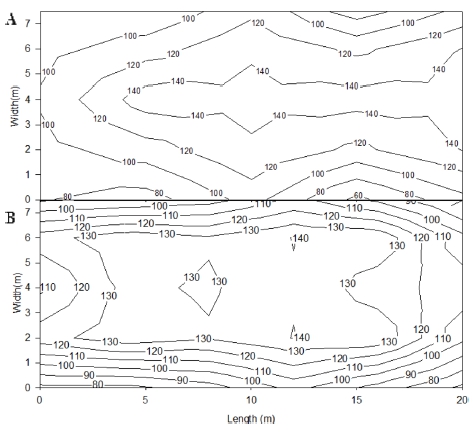
In order to make the same PPFD, eighteen SPs were installed at an interval of 3 m × 3 m and fifteen of HPSs were installed at an interval of 3 m × 4 m. The PPFDs at the floor (15 areas) under lamps were measured at night by using a quantum sensor (Li-250A, Li-COR, Lincoln, NE, USA). The average PPFDs were 113.1 and 112.1 μmol·m-2·s-1 at HPS and SP, respectively (Fig. 2). To assess the difference in light environment in the greenhouses under supplemental lights, the light spectra were measured at Sun, Sun + HPS, and Sun + SP in the greenhouses on a clear day and a cloudy day by using the radio spectrometer. The number and weight of fruits were measured weekly from 6 April to 28 April. The plant height, number of nodes, diameter of stem, and leaf area were measured at 1 June, 2016.
5. Statistical Analysis
Data were analyzed by one-way analysis of variance (ANOVA) using the SAS program (Statistical Analysis System, 9.2 Version, SAS Institute, Cary, NC, USA). Treatment means were compared using LSD test at p < 0.05 for greenhouse experiment and t-test at p < 0.05. Graphs were generated with Sigmaplot 11 (Systat software Inc., CA, USA).
Results
1. Spectral Characteristics of Light Sources
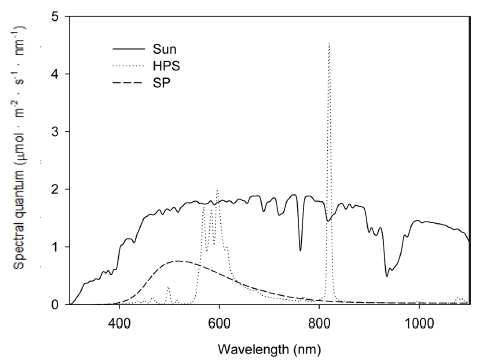
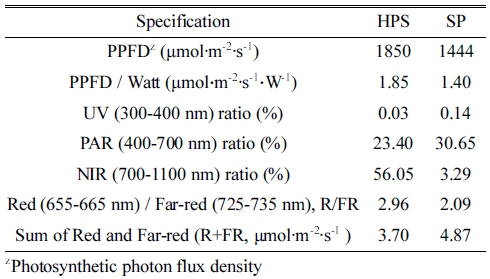
The spectral characteristics of HPS and SP were measured in the growth chambers. SP has a more similar light spectrum to the sunlight than HPS which has two peaks around 550 ~ 650 nm and 800 ~ 820 nm (Fig. 3) and showed 1.3 and 4.7 times higher in PAR and UV ratios, and 17.0 and 1.3 times lower in NIR and R/FR ratios than HPS (Table 1). SP showed the R/FR ratio closer to sunlight than HPS. The photon flux sum of R and FR (Myers, 1971) in SP was 1.3 times higher than that in HPS.
In addition, light spectra in the greenhouses were measured before sunrise, after sunset, on a cloudy day, and at night on 2 March, 2016 (Fig. 4). Regardless of different solar radiation conditions after sunrise, before sunset, and on cloudy days, the light intensities were very low at that time and shown as similar spectra patterns to those observed in the growth chambers.
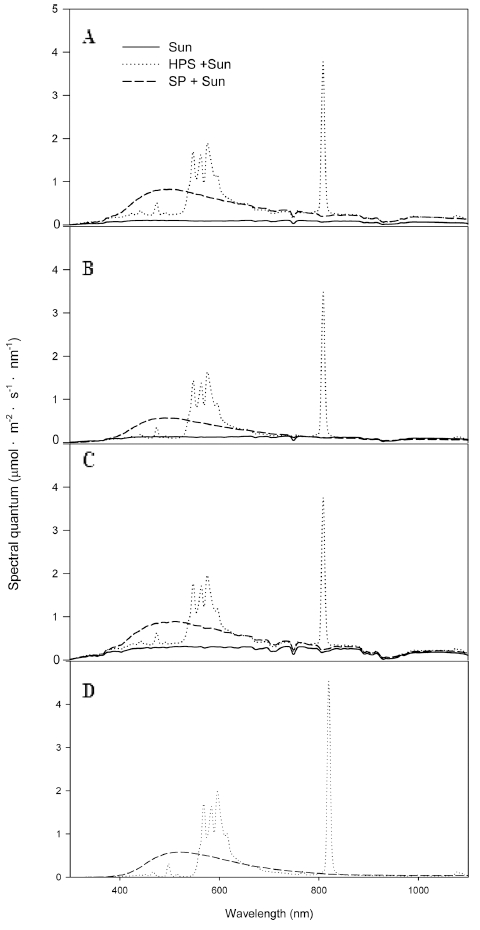
Fig. 4
Spectral quantum distributions with time in the greenhouses under sunlight with no supplemental lighting (Sun), sunlight with high-pressure sodium lamp (Sun + HPS), and sunlight with sulfur plasma lamp (Sun + SP): (A) before sunrise, (B) after sunset, (C) cloudy day, and (D) midnight on 2 March, 2016.
2. Growth and Photosynthesis at SP and HPS as Main Lights in the Growth Chamber
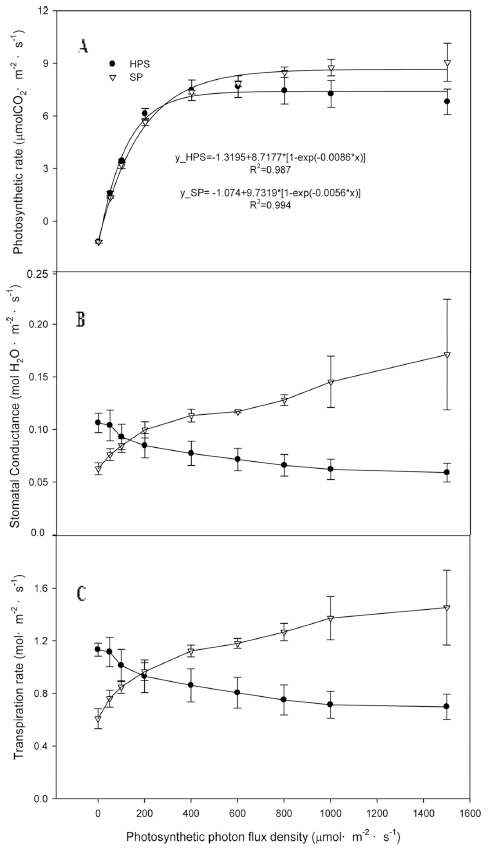
The plant height, leaf area, leaf fresh weight, and leaf dry weight at SP in the growth chambers were significantly higher than those at HPS, while the stem diameter, number of leaves, SPAD, stem fresh weight, and stem dry weight were not significantly different on 27 June, 2016 (Table 2). In particular, SP significantly enhanced the leaf area by 29% as compared to HPS and also induced a higher plant height, leaf area, and dry weight than HPS during the growth period in the growth chamber (Fig. 5). In particular, the plant height and dry weight were significantly higher at SP than HPS all the time on 27 June (Figs. 5A, 5C). The leaf areas at SP and HPS showed similar patterns until 12 June, 2016; however, the rate of leaf area at HPS decreased afterwards (Fig. 5B). The photosynthetic rate was significantly higher at SP than that at HPS on 16 June (Fig. 6). Below the PPFD of 300 μmol·m-2·s-1, the photosynthetic rate was similar between SP and HPS; however, afterwards it became significantly higher at SP (Fig. 6A). The stomatal conductance and transpiration rate showed similar tendencies to the photosynthetic rate, showing increases at SP and decreases at HPS with increase of PPFD (Figs. 6B, 6C).
Table 2. Growth indexes and chlorophyll contents of paprika (n = 5) grown under high-pressure sodium lamp (HPS) and sulfur plasma lamp (SP) in the growth chamber (on 27 June, 2016).
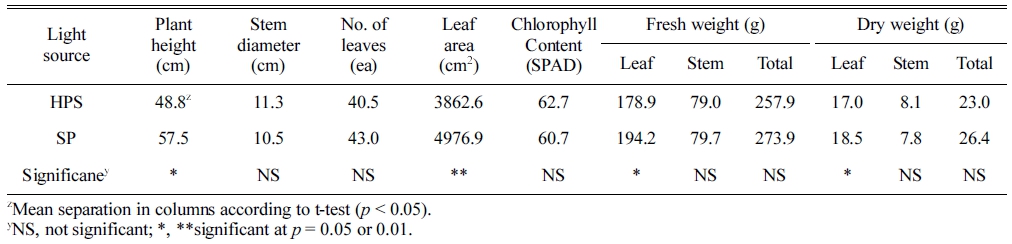 |
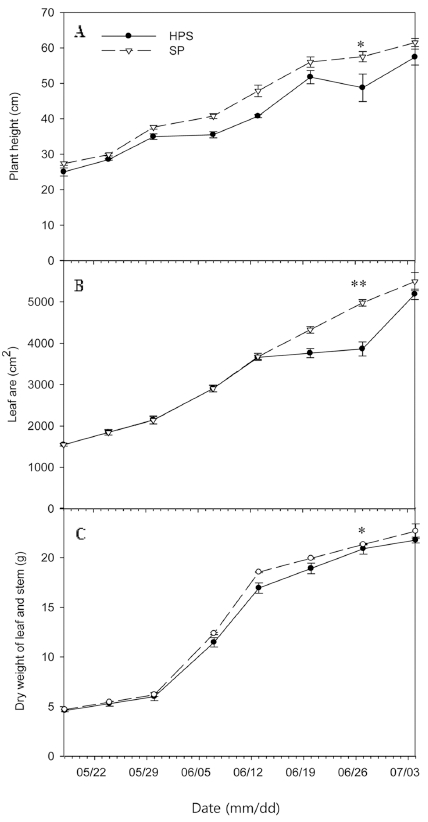
Fig. 5
Changes in plant height (A), leaf area (B), and dry weight (C) of paprika grown under high-pressure sodium lamp (HPS) and sulfur plasma lamp (SP) in the growth chamber (from 18 May to 5 July, 2016). Vertical bars represent the standard error of the mean (n = 5). *, ** show significance at p = 0.05 or 0.01 according to t-test on 27 June, respectively.
3. Yield and Fruit Number of Paprika at SP and HPS as Supplemental Lights in the Greenhouses
In greenhouse, the stem diameter was significantly bigger at Sun + HPS and Sun + SP treatments than at Sun (Table 3). The accumulated yield and number of fruits were significantly higher at Sun + HPS and Sun + SP than at Sun (Fig. 7). Specifically, there were 79% and 227% higher in accumulated yields (Fig. 7A) and 81% and 309% higher in numbers of fruits, respectively, at Sun + HPS and Sun + SP than at Sun from 6 April, 2016 (DAT 119) to 28 April, 2016 (DAT 147) when supplemental lighting affected the growth of the plants (Fig. 7B). Consequently, the number of fruits and weight of fruits were higher at SP than at HPS. Considering the difference in amount of fruits, the yield of paprika per energy was higher at SP than at HPS.
Table 3. Growth indexes of paprika (n = 3) grown under sunlight with no supplemental lighting (Sun), sunlight with the high-pressure sodium lamp (Sun + HPS), and sunlight with the sulfur plasma lamp (Sun + SP) in the greenhouses on 1 June, 2016.
 |
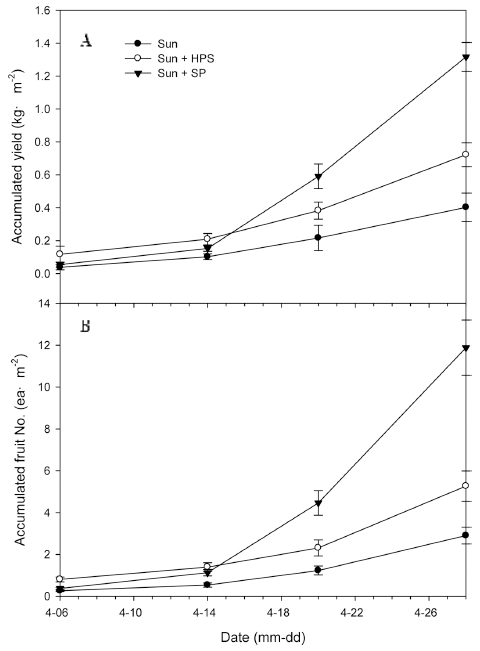
Fig. 7
Accumulated yields (A, kg·m-2) and fruit numbers of paprika fruits (B, ea/m2) grown under sunlight with no supplemental lighting (Sun), sunlight with high-pressure sodium lamp (Sun + HPS), and sunlight with sulfur plasma lamp (Sun + SP) in the greenhouses from 6 to 28 April, 2016. Vertical bars represent the standard error of the mean (n = 3).
Discussion
1. Growth and Photosynthesis at SP and HPS as Main Light in the Growth Chamber
The growth indices of paprika was stimulated by SP than HPS and the individual leaf area increased at SP in the growth chamber. Active phytochrome activated by red light, such as supplemental lamps like SP and HPS, increased plant growth as a result of increased photosynthetic rates. The ratio and R and FR in the sunlight on clear days was calculated as 1.09 (Smith, 1982). In this study, the ratio at SP showed closer to that on clear days than at HPS. It means that SP stimulated photosynthesis than HPS and active photosynthesis transferred growth signal by active phytochrome (Paul, 2016).
The higher photon flux sum of R and FR at SP than HPS, the more stimulated the improvement of photosynthesis by Emerson effect (Myers, 1971). In particular, SPs have widely spectral characteristics that stimulated the plant height due to more red wavelengths and smaller R/RF ratio like the sunlight. Furthermore, these results may come from higher photosynthetic rates of paprika grown at SP than HPS lamp in the growth chamber due to the high stomatal conductance and transpiration rate of paprika grown at SP. Light quality had no impact on the initial slope of the CO2 response curve in our results, while the temperature could influence the initial slope (Sage and Sharkey, 1987;Sager et al., 1989; Sharkey et al., 2017). Since the initial slope was Pepindetermined by the Rubisco activity (von Caemmerer and Farquhar, 1981), this indicated that the plant treated with high light limited the Rubisco activity. Photoreceptors, such as phytochromes, phototropins, and cryptochromes, are involved in physiological responses, such as stomatal density or size of aperture and photosynthesis characteristics, and these are affected by light quality (Hogewoning et al., 2010a, 2010b;Amoozgar et al., 2017). The blue wavelength has been reported to increase stomatal opening and improve gas exchange for photosynthesis (Kinoshita et al., 2001). The higher blue band spectrums found in the plasma lamps can have a greater effect on the light saturation and compensation point (Fig. 6A).
2. Yield and Fruit Number of Paprika at SP and HPS as Supplemental Light in the Greenhouses
Previous research reported that the yield of tomato increased with the increase in the amount of light received from flowering to harvest (McAvoy et al., 1989). When the light level on the top of the canopy or within the canopy was low, application of supplemental lighting at the top of the canopy or within the canopy enhanced the yield of tomatoes and other vegetable species (McAvoy et al., 1989; Hovi et al., 2004;Pettersen et al., 2010). The results of the present study are consistent with these findings.
In this study, supplemental lighting was applied from 1 March, 2016 to 1 June, 2016 for three months because the supplemental lighting effect drastically decreased after 1 May, 2016 due to longer day length. Therefore yield data were collected from one month after the treatment to before May, 2016 as pure results of three-month supplemental lighting because the yield was somewhat affected by previous sunlight condition for one month after 1 March, 2016 and was little affected by the supplemental lighting after 1 May, 2016. With supplemental lightings, the growth and yield were enhanced than those without supplemental lighting (Fig. 6). In the greenhouse, the yield of paprika was higher at the supplemental lightings than in the control.
Many studies have reported that individual wavelengths can differentially affect fruit production (Li and Kubota, 2009;Hogewoning, 2010a, 2012;Hikosaka et al., 2013;Guo et al., 2016a, 2016b;Hao et al., 2016;Amoozgar et al., 2017;Yang et al., 2017). For example, Pepin et al. (2014) suggested that tomato crop yield may be achieved by providing supplemental LED lighting within the plant canopy under low natural light conditions. Also, application of plasma emitting lights produced a 5.5% increase in dry matter with a 13% increase in fruit number of paprika (Lee et al., 2014). In addition, tomato plants under plasma lighting also had more consistent fruit size with a higher nutritional value and better taste (data not shown). Since plasma light has more ultra violet wavelengths and less yellow and red light than HPS, this can impact the biochemical metabolites in fruits.
The supplemental lighting is an effective method of enhancing crop growth and yield. Considering that our experiments started from March to April, 2016, the cultivation conditions rapidly improved with the increase of solar radiation and air temperature. As the supplemental lighting promoted the physiological activities of the plants, adequate fertigation might be adjusted to meet the increased requirements. Despite this condition, the results indicate that supplemental lighting, particularly SP, could a proper artificial light source. SP showed a light spectrum similar to sunlight, higher PAR and photon flux sum of red and far-red wavelengths than HPS, which increased the photosynthesis and yield of paprika. Further studies are needed to investigate the fertigation system control adapted to supplemental lighting.