Introduction
Sprouts have excellent nutritional and functional value because they contain protein, dietary fiber, minerals, vitamins, and health-promoting phytochemicals that are synthesized during seed germination (Cevallos-Casals and Cisneros-Zevallos, 2010; Waje and Kwon, 2007). Recent research has demonstrated that sprouts contain antioxidant and anticancer properties, which has caused them to receive more attention as healthy, plant-based foods (Gawlik- Dziki et al., 2012; Yuan et al., 2010).
Despite these benefits, sprout consumption is occasionally associated with disease (Alexander and Hammes, 2003). When sprouts are produced using nonsterilized or incompletely sterilized seeds, high humidity and temperature generated by irrigation and seed respiration during germination promote the growth of microorganisms that remain on the seed coat (Peñas et al., 2010; Taormina et al., 1999). At least 40 cases of foodborne disease related to consumption of sprouts are reported to have occurred from 1973 to 2006. Escherichia coli O157:H7, Salmonella sp., and Bacillus cereus were the major bacteria implicated in these diseases, and various treatments (e.g., high pressure, heat, electron-beam and gamma irradiation) have been evaluated for their ability to reduce bacterial growth during postharvest processing of sprouts (Alexander and Hammes, 2003; Cevallos-Casals and Cisneros-Zevallos, 2010; Waje et al., 2009).
Air ions are classified as cations, which gain electrons, and anions, which lose electrons (Kataka, 1978). These molecules are formed by energy generated by colliding neutrons or molecules with elements in soil (e.g., radium and thorium), the radioactive gas radon, ultraviolet light, and cosmic rays. Air anions promote human health by activating cell metabolism and by causing acidic blood to become more alkaline, which confers strong immunity (Chee, 2009). Air anions have been applied in industrial areas because of their sterilizing effects (Yun and Seo, 2013). Two major methods, the high-voltage and spray-ionization methods, are used to generate air anions. The high-voltage method produces negatively charged oxygen molecules by colliding atmospheric oxygen with anions generated by a high-voltage pulse with direct current (Chee, 2009). The spray-ionization method generates oxygen anions by combining electrons from ionized water molecules with oxygen, called the Lenard effect (Park, 2006). A typical example of the Lenard effect in action occurs in waterfalls, where unstable electrons are produced when microdroplets of water strike rock, generating oxygen anions.
Air anions have been known to have positive effects on plant growth. Exposure of barley to air anions caused a significant increase in height, fresh weight, and dry weight (Krueger et al., 1963b). Growth promotion in plants exposed to excessive air anions has been attributed to increased respiration and photosynthesis, and to the synthesis of cytochromes and iron-binding proteins (Elkiey et al., 1985). To date, research relating to air anions has been limited to a few crops, such as barley and oats (Krueger et al., 1962; Krueger et al., 1963a; Krueger et al., 1965); examination of this effect should be conducted in additional crops, including vegetables such as edible sprouts. The objective of this study was to assess the effects of air anions generated by the spray-ionization method on growth and sterilization of edible sprouts.
Materials and Methods
1. Plant Materials and Growth Conditions
Seeds of four plant species were used in this study: lettuce (Lactuca sativa, Asia Seed Co., Ltd., Seoul, Korea); red cabbage (Brassica oleracea var. capitata f. rubra, Onsem Co., Ltd., Anseong, Korea); tatsoi (Brassica campestris var. narinosa; Asia Seed Co., Ltd., Seoul, Korea); and kale (Brassica oleracea var. acephala, Koregon Co., Ltd., Anseong, Korea). Seeds were sown into twelve plastic trays (18 × 9 × 3 cm, L ×W× H) in one layer and cultured in two auto-sprouters (EasyGreen Mikro-FarmTM, Seed and Grain Technologies, LLC. Albuquerque, USA). The auto-sprouters were placed in a growth chamber equipped with red and blue LEDs (red:blue = 78:22, 85 ± 3 μmol m–2 s–1, 12-h photoperiod) and the temperature was maintained at 20°C. One autosprouter was used as a control and was provided irrigation water by spraying for 15 min every 4 h; the second sprouter was equipped with an ultrasonic fogging system (DH-014, NIT Co. Ltd., Suwon, Korea) for spray-ionization, and was supplied with mist and anions for 37 min every 4 h (Fig. 1). Both treatments supplied same amount of water per day (1.35 L sterile distilled water d–1), sufficient for seed germination. The range of anion concentration applied to the sprouts was 28 × 104~40 × 104 ions cm–3 and was measured by an anion counter (COM-3600, Com Systems Ltd., Tokyo, Japan). The range of anion concentration in the control was 27~30 ions cm–3. The trays were rotated daily to minimize differences in light intensity, moisture, and anion concentration according to position.
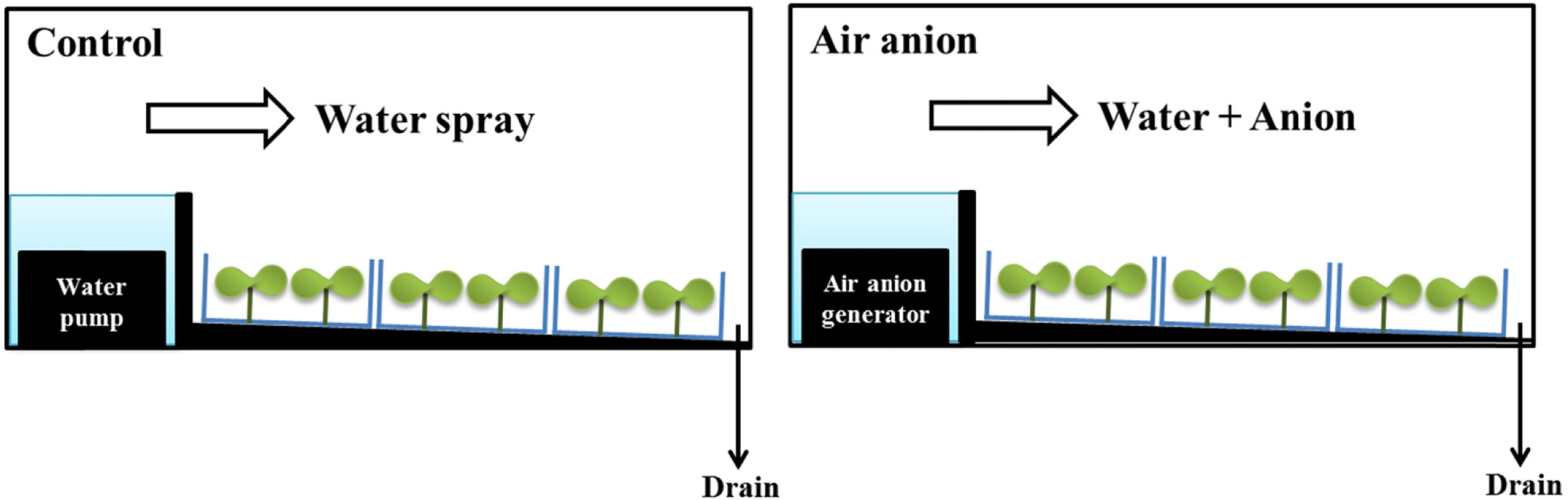
Fig. 1.
Two sprout-growing systems for control and air-anion treatment placed on a growth chamber. Sterile distilled water in control was supplied to sprouts by water pump. In air anion treatment, both the distilled water and air anion were applied to sprouts by an air anion generator. Irrigation amount of control and air anion treatment was the same as 1.35 L per day.
2. Sprout Growth
Ten sprouts of each species were sampled from the control and spray ionization treatments for measurement of hypocotyl and radicle length, fresh weight, and dry weight at 7 d after seeding. An electronic scale (SI-234, Denver Instruments, Bohemia, USA) was used to determine fresh weight, after which the sprouts were dried at 70°C in an oven (VS-1202D2, Vision Scientific Ltd., Daejeon, Korea) for 72 h to determine dry weight.
3. Radicle Activity
Radicle activity was analyzed in four sprouts of each species prior to cotyledon formation. Samples (about 0.5 g, 10 sprouts/repeat) were placed into 15-mL tubes, and 1% TTC (2,3,5-triphenyl tetrazolium chloride, Sigma-Aldrich, St. Louis, USA) solution, 0.1 M sodium phosphate buffer (pH 7.0), and distilled water were then added (1:4:5 v/v/v), bringing the volume to 10 mL. The tubes were incubated for 4 h at 37°C in a water bath (MSB-2011D; Mono-tech Eng. Co. Ltd., Siheung, Korea) in the dark. The reaction was then stopped by adding 3 mL of 2 N H2SO4. The solutions were passed through a filter paper (No. 2; Whatman International Ltd., Maidstone, UK); radicles remaining on the filter paper were dried with gauze and ground with a mortar and pestle, and 10 mL ethyl acetate and sea sand were added to extract formazan. The supernatant containing extracted formazan was collected, and optical density was measured at 470 nm using a spectrophotometer (UV-1800, Shimadzu Co. Ltd., Kyoto, Japan). A standard curve was prepared using 1,3,5-triphenyltetrazolium formazan (TPF, Sigma-Aldrich, St. Louis, USA) standard solution (0, 2.5, 5, 10, 20, 40 ppm) dissolved in ethyl acetate. Radicle activity was expressed by mg formazan generated h–1 g radicle–1.
4. Bacterial Counts
Test solutions were prepared by homogenizing sprouts (about 5 g) with 50 mL sterile distilled water; drainage water from the auto-sprouters was sampled for bacterial counts at 7 d after seeding. One milliliter of test solution was diluted with sterile distilled water in a series of 10 dilutions. The diluted test solution (50 μL) was smeared on nutrient agar medium (DifcoTM, Becton, Dickinson and Company, Franklin Lakes, USA) and incubated at 28°C for 2 d. Bacterial colonies were counted and presented as the product of the number of colonies and number of dilutions.
Results and Discussion
1. Growth
Hypocotyl and radicle length were influenced by spray ionization in each of the four tested species (Fig. 2). Hypocotyl length increased significantly in red cabbage and kale sprouts exposed to air anions, by 26% and 27% respectively, compared with the control; hypocotyl length of lettuce and tatsoi sprouts did not differ between the control and anion treatments. Radicle length in lettuce, red cabbage, and kale sprouts subjected to spray-ionization was 60%, 64%, and 45% higher than that of the control, respectively, while radicle length of tatsoi did not differ significantly.
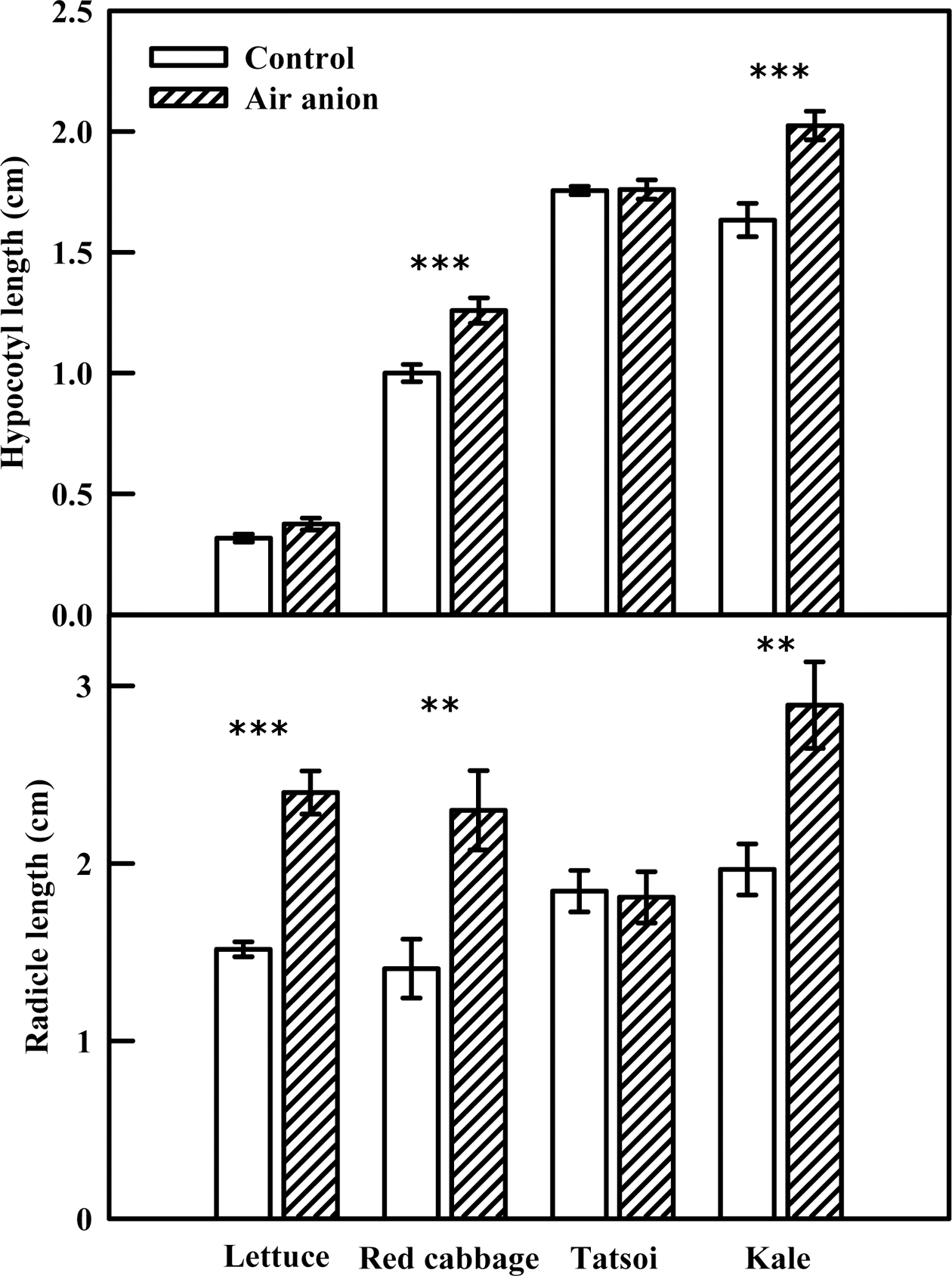
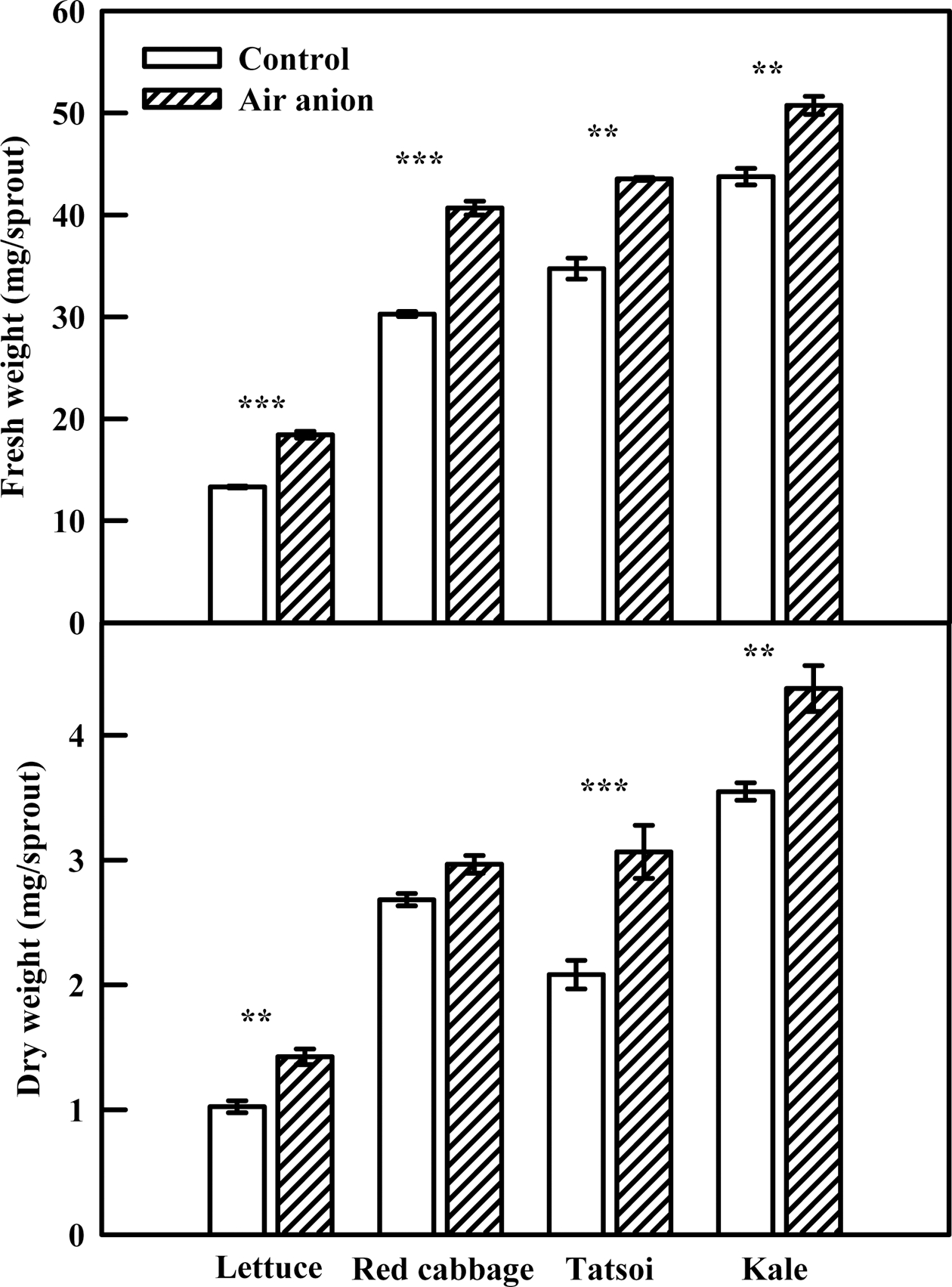
Fig. 2.
Hypocotyl and radicle length of various edible sprouts exposed to air anion at 7 days after seeding. The concentration of air anion treated to sprouts was 28 × 104~40 × 104 ion·cm–3. The data are the means and the bars indicate standard errors (n=10). Significant at **P = 0.01 and ***P = 0.001.
Air-anion treatment had significant positive effects on sprout FW and DW (Fig. 3). The FW of lettuce, red cabbage, tatsoi, and kale sprouts exposed to spray-ionization was 38.5%, 34.3%, 25.3%, and 16.0% higher than that of the control, respectively. Dry weights of lettuce, tatsoi, and kale sprouts in the air-anion treatment were significantly higher than DW of the respective control treatments. Hypocotyl and radicle lengths of tatsoi sprouts were not influenced by air-anion treatment, but hypocotyl diameter was large, causing apparent increases in FW and DW (data not shown). Spray ionization generally had a positive effect on seed germination and sprout growth, although the growth response differed among the species. Lim et al. (2011) reported that germination and growth rate of brown rice were improved by air-anion treatment. A previous study also reported that air anions improved lettuce growth (Song et al., 2014; Wachter and Widmer, 1976). Air anions may stimulate respiration in seeds by promoting oxygen uptake (Krueger et al., 1963b). Since we controlled for environmental factors, including moisture condition in relation to seed germination, our findings may be a result of promotion of seed respiration by air anions.
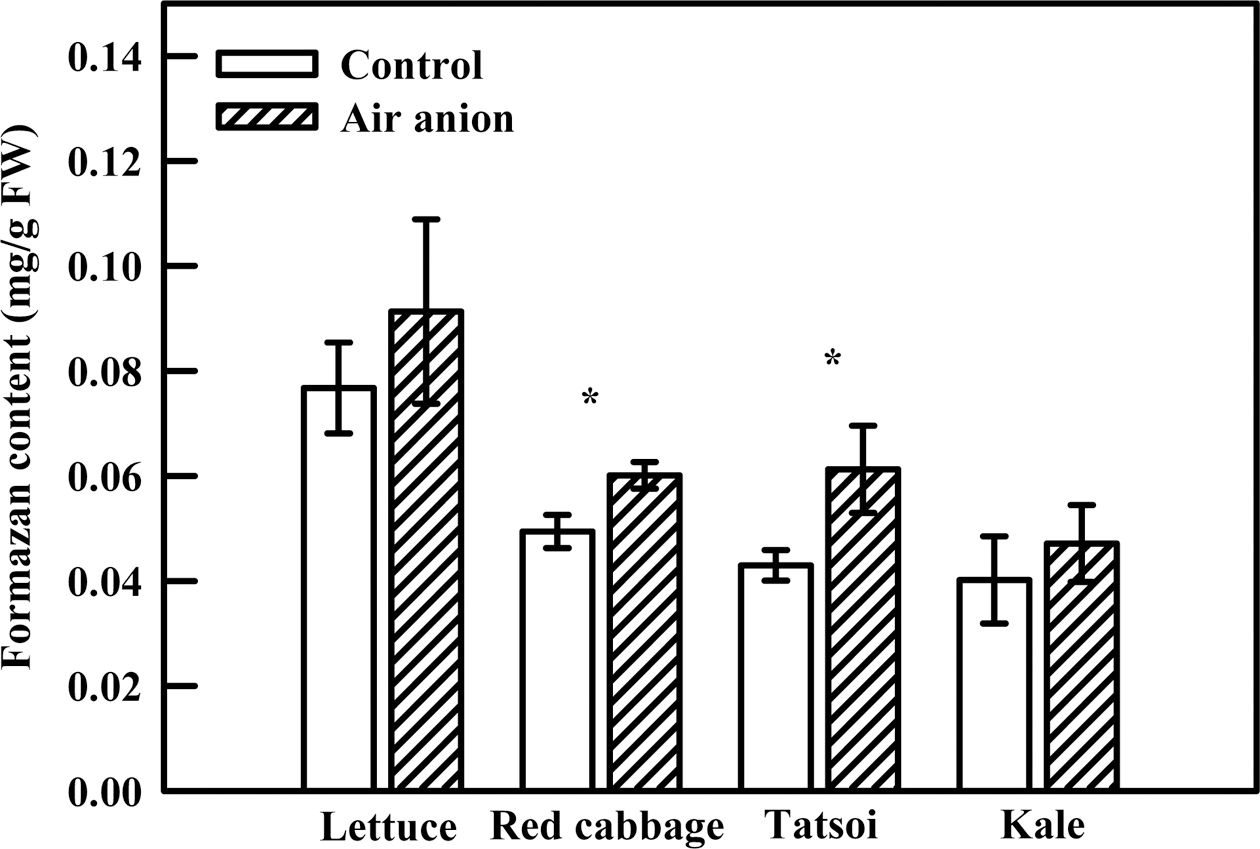
2. Radicle Activity
Spray-ionization treatment increased radicle activity prior to cotyledon generation in all sprout species (Fig. 4). Tatsoi showed the highest rate of increase (42.6%), followed by red cabbage (21.7%), lettuce (19.0%), and kale (17.2%). When colorless tetrazolium combines with hydrogen ions generated by dehydrogenase, tetrazolium is converted to formazan (red), with the quantity of red representing the level of radicle activity (Jones and Prasad, 1969). Thus, the increased radicle activity observed here may have represented increased respiration due to vigorous dehydrogenase activity suggesting that spray-ionization may stimulate respiration of seeds during germination.
3. Bacterial Counts
Spray-ionization of sprouts had a sterilizing effect (Fig. 5). With the exception of kale, the number of bacterial col- onies associated with each sprout species in the anion treatment was lower than that in the control at 7 d after seeding. Anion treatment caused a significant (66.1%) reduction in bacterial counts in red cabbage sprouts. Bacterial colonies in spray-ionization-treated lettuce and tatsoi sprouts were significantly reduced, by 40.8% and 18.9%, respectively. Numbers of colonies were reduced by 78%, 11%, 36% in the drainage water of lettuce, tatsoi, and kale, respectively. These results suggest that air anions suppressed the propagation of bacteria on seed coats. Arnold et al. (2004) reported that outbreaks of bacterial pathogens such as Campylobacter jejuni, E. coli, Salmonella enteritidis, and Listeria monocytogenes, and spores of Bacillus stearothermophilus were nearly eliminated by exposure to air anions. A sterilization effect of air anions was also observed in Candida albicans (Sharagawi et al., 1999). Kellogg et al. (1979) reported that superoxide radicals generated by air anions enhanced the surface charge of Staphylococcus albus, causing de-esterification that weakened bacterial cell membranes and resulted in cell death. The effects of spray ionization on sterilization can be interpreted similarly in the present study. Sprouts can be produced more safely by applying spray ionization, which reduces the threat of infection by bacteria such as E. coli O157:H7, Salmonella sp., and Bacillus cereus, which are commonly present on edible sprouts.
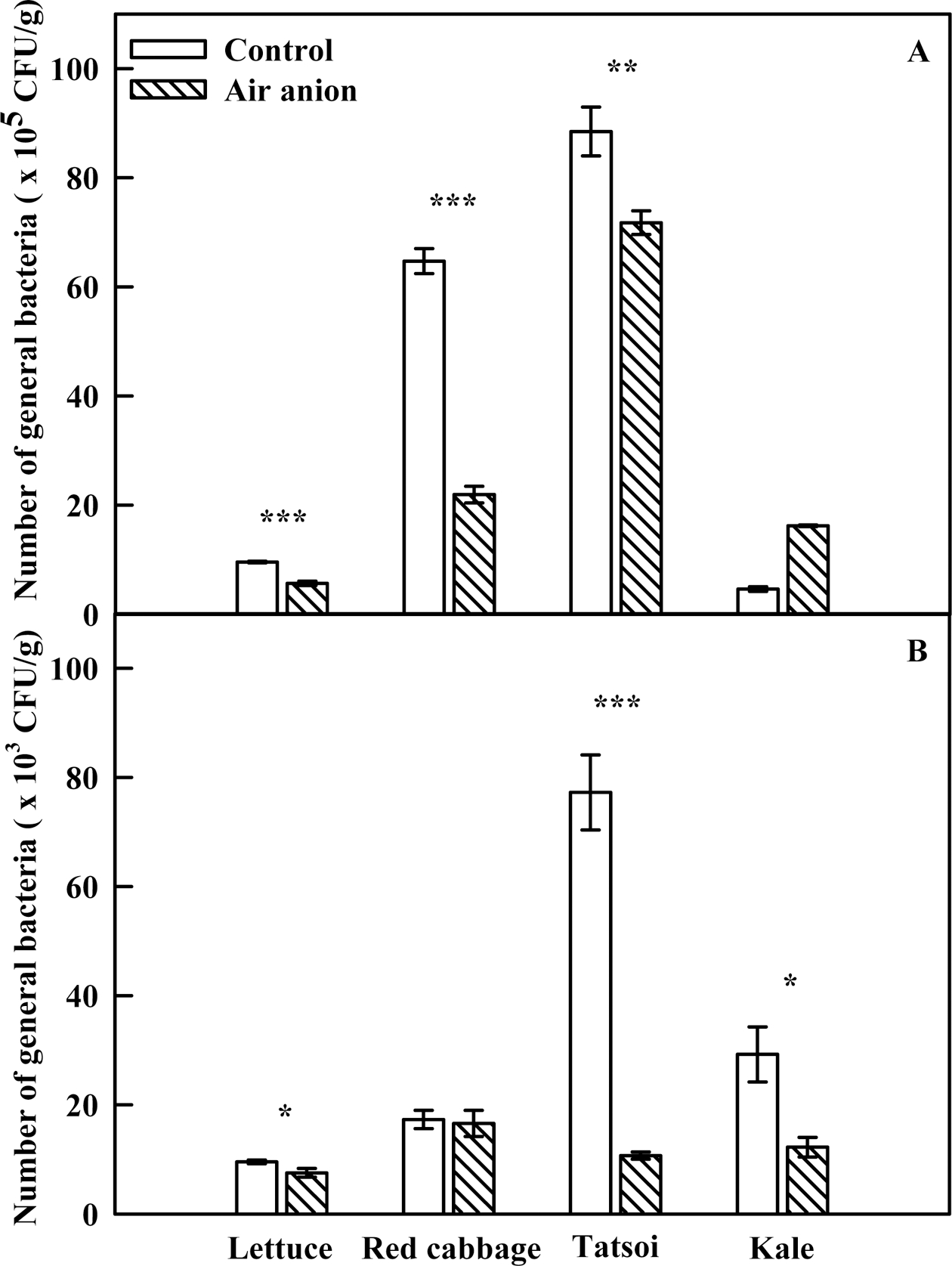
Fig. 5.
Bacterial count of various edible sprouts (A) and drained water (B) exposed to air anion at 7 days after seeding. The concentration of air anion treated to sprouts was 28 × 104~40 × 104 ion·cm–3. The data are the means and the bars indicate standard errors (n = 3). Significant at* P= 0.05, **P = 0.01 and ***P = 0.001.
Conclusion
Spray-ionization treatment enhanced the growth of edible sprouts of four plant species by increasing respiration, although sensitivity to air-anion treatment differed among the species. Anion treatment also demonstrated a sterilization effect, reducing the generation of bacteria that present major drawbacks to sprout production. Incorporation of airanion technology into sprout production should provide positive effects on yield and safety.