Introduction
Interest in the use of grafted vegetable seedlings has increased in many countries during recent years in order to improve resistance to biotic and abiotic stresses (Lee and Oda, 2003; Rivero et al., 2003). The successful production of grafted plants requires highly technical grafting skills and environmental control during healing and acclimatization process. Proper acclimatization is critical for grafted plants to survive (Lee et al., 2010). After grafting, it is important to control the environment around grafted seedlings for the robust joining of a scion and rootstock. In conventional culture, acclimatization may be achieved simply by enclosing the rootstock and scion in a plastic film shaded by cloth to avoid heat buildup, keep the high relative humidity and reduce light intensity until the union is formed (Oda, 1999; Davis et al., 2008). However, it is quite difficult to control optimal environment for healing and the acclimatization of grafted seedlings under natural condition.
Once grafting is performed, it is important to increase the chances for vascular bundles of the scion and rootstock to come into contact (Oda et al., 1994). During formation of the graft union, callus proliferation in both the rootstock and the scion is very important for vascular connection in the rootstock-scion interface (Ogata et al., 2005). A low or incorrect callus formation between the rootstock and scion could lead to defoliation, reduction of scion growth and low survival of grafted plants (Johkan et al., 2008; Johkan et al., 2009; Oda et al., 2005). In addition, some researchers report that callus induction and growth required light condition (Afshari et al., 2011; Moon et al., 1997). Therefore, light is one of important factors that are necessary for grafted plants to survive (Nobuoka et al., 2005).
Light quality regulates a variety of plant development pathways from germination to flowering induction and fruit development (Jiao et al., 2007). Light-emitting diodes (LEDs) are the new fourth-generation light source with good spectral characteristics and spectral width, and can be assembled to match light quality which plants need (Goins et al., 1997). Many studies were carried out to evaluate effect of LEDs on growth and development of plants, however, most studies have focused on growth, photosynthesis, metabolism and gene expression (Neff et al., 2000; Yu and Ong, 2003; Wang et al., 2009; Zhang et al., 2003), while less is known about how LEDs affects the survival rate and quality of grafted seedlings during the healing and acclimatization process. Regarding irradiation by different LEDs during healing and acclimatization period, Vu et al. (2014) reported that there were no significant differences between red LEDs and white fluorescent lamps treatments with survival rate and growth parameters, but they decreased in far-red LEDs, blue LEDs, or darkness treatments. Therefore, the objective of this study was conducted to confirm the effects of red LEDs during healing and acclimatization process on the survival rate and seedling quality of different tomato cultivars.
Materials and Methods
1. Plant materials and grafting method
Both scion and rootstocks of tomato seeds were sown in the 128-cell plug trays (Bumnong. Co., Ltd., Jeongeup, Korea) that was filled with commercial growing substrate (BM2, Berger Group Ltd., Quebec, Canada). ‘Choice’ cultivar was used as scion while ‘B-Blocking’, ‘Kanbarune’, ‘High-power’ cultivars were used as rootstocks. During the plastic house, seedlings were watered daily or as required. One week after sowing, seedlings started to receive a fertilization based on Wonder Grow fertilizers (Wonder Grow Fertilizers, Chobi Co., Ltd., Seoul, Korea) twice a week through an overhead irrigation. Twenty-two days after sowing, the seedlings were used for grafting.
Grafting was done by splice grafting method. Plants were grafted by first making approximately 45o cuts on both the rootstock and scion seedlings using a sharp razor blade. After placing the scion on the rootstock, ordinary grafting clips were used to fix the grafted position tightly together (Lee and Oda, 2003). Rootstock and scion plants with similar stem diameters (2.3~2.4 mm) were chosen to increase the grafting success rate.
2. Light quality treatments
Red LEDs (the light intensity was approximately 15 µmol · m−2 · s−1 PPFD) and no light (darkness) were used for treating three rootstock cultivars in healing room for 10 days. The healing room was equipped with an auto-controlled air conditioning system for healing the grafting. Relative humidity was maintained 85~90%, temperature were set up 23°C. After 10 days in the healing room, grafted seedlings were transferred to plastic house in natural condition for 5 days.
3. Data collection and analysis
All grafted seedlings were evaluated for signs of graft failure on the 10th day after grafting. Infected seedlings were evaluated on the 15th day after grafting. Grafted seedlings on the 15th day after grafting were harvested for analysis of growth characteristics. Plant height and number of leaves of grafted seedlings were measured, leaf area estimated by leaf area meter (Delta-T Device Ltd., Burwell, Cambridge, UK), leaf chlorophyll content was measured by using a chlorophyll meter (SPAD-502 Plus, Konica Minolta Sensing Inc., Osaka, Japan). The fresh and dry weights of shoot and root were measured. Dry weights of shoot and root were taken through oven-dry method at 80°C for 72 h until constant weight was achieved. T/R ratio and compactness were calculated according to Kim et al. (2008). For the root system morphology, the WinRHIZO Pro 2009c (Regent Instruments, Inc, Quebec, Canada) images analysis system was used, coupled with professional scanner Epson 10000XL (Seiko Epson Corporation, Nagano, Japan) according to Arsenault et al. (1995). The roots were detached from their shoots and then placed in a tray (15 × 30 × 2 cm) with water and placed on the scanner. Scanned images were analyzed by the WinRHIZO program for total root surface area, total root length, average root diameter, and number of root tips.
The experimental design was a split-plot. Light was the main plot and rootstock cultivars were the sub plot in this experiment. Six treatments were designed by combination of two levels of light (red LEDs and no light) and three cultivars. For the statistical analysis of survival rate, one 128-cell plug tray with 64 plants in each replication was measured. For the statistical analysis of growth parameters, ten seedlings per treatment from each replication were randomly selected. Data were analyzed using SAS v.9.3 software (SAS Institute Inc., Cary, NC, USA). Mean separations were calculated using Duncan’s multiple range tests.
Results
1. Effect of red LEDs on survival rate of grafted tomato seedlings
On the 10th day after grafting, the survival rates of grafted seedlings in red LEDs were higher than the survival rate of grafted seedlings in no light treatment. Significant variation on survival rates of rootstock cultivars was observed in no light treatment but there was not significant variation in red LEDs treatment. Light treatment also reduced percentage of infected plants, except for the ‘Kanbarune’ cultivar. Final survival rates (87.9~95.4%) were considerably high in all rootstock cultivars when red LEDs irradiation was applied during the healing and acclimatization process whereas final survival rates (34.9~67.7%) were low in no light treatment. Red LEDs had greatly influenced the survival rate of ‘Kanbarune’ cultivar which ‘High-Power’, ‘B-Blocking’ cultivars followed in order (Table 1).
Table 1.
Effect of red LEDs and darkness during healing and acclimatization process on the survival rate and infected plants of grafted tomato seedlings.
| Treatments | Cultivars | Survival rate after 10 days (%) | Infected plants (%) | Final survival rate (%) |
|---|---|---|---|---|
| No light (Darkness) | ‘B-Blocking’ | 72.6 bz | 4.8 a | 67.7 b |
| ‘Kanbarune’ | 35.7 d | 1.8 c | 34.9 c | |
| ‘High-Power’ | 50.8 c | 4.9 a | 48.5 c | |
| Red LEDs | ‘B-Blocking’ | 90.2 a | 3.3 b | 87.9 a |
| ‘Kanbarune’ | 98.2 a | 1.8 c | 95.4 a | |
| ‘High-Power’ | 93.4 a | 3.3 b | 90.5 a | |
| Significance | ||||
| Light (L) | **y | * | ** | |
| Cultivar (C) | * | * | * | |
| L × c | * | * | * | |
2. Effect of red LEDs on growth characteristics of grafted tomato seedlings
Analysis of the variance of growth characteristics in red LEDs and no light of three rootstock cultivars are presented in Table 2 and 3. Significant differences on plant height, leaf length, leaf width, leaf area, fresh and dry weight of shoot and root of grafted seedlings were observed between red LEDs and no light treatments. Plant height of seedlings in red LEDs was higher than that in no light treatment. Stem diameter was not significant difference on light treatments but significant different on rootstock cultivars. Leaf number and leaf chlorophyll value did not exhibit any significant difference between no light and red LEDs treatment in all rootstock cultivars. However, leaf length, leaf width, and leaf area of seedling in red LEDs were higher than those in no light treatment. Fresh and dry weights of shoot and root of seedlings in red LEDs were greater than those in no light treatment. Different light treatments and different rootstock cultivars did not affect on T/R ratio of grafted seedlings but seedlings in red LEDs were significantly more compact than those in no light treatment.
Table 2.
Effect of red LEDs and darkness during healing and acclimatization process on growth characteristics of grafted tomato seedlings.
| Treatments | Cultivars | Plant height (cm) | Stem diameter (mm) | No. of leaves | Leaf chlorophyll value (SPAD) | Leaf length (cm) | Leaf width (cm) | Leaf area (cm2) |
|---|---|---|---|---|---|---|---|---|
| No light (Darkness) | ‘B-Blocking’ | 6.88 cz | 2.57 ab | 4.20 c | 45.05 a | 6.56 c | 4.50 b | 15.10 b |
| ‘Kanbarune’ | 6.58 c | 2.32 c | 4.40 bc | 44.11 a | 6.09 c | 4.28 b | 13.50 b | |
| ‘High-Power’ | 6.67 c | 2.45 bc | 4.50 bc | 43.74 ab | 6.48 c | 4.28 b | 13.20 b | |
| Red LEDs | ‘B-Blocking’ | 8.93 a | 2.73 a | 5.00 a | 40.95 b | 9.03 a | 5.18 a | 19.20 a |
| ‘Kanbarune’ | 7.64 b | 2.49 bc | 4.30 bc | 44.84 a | 7.59 b | 5.25 a | 17.70 a | |
| ‘High-Power’ | 7.39 b | 2.54 b | 4.70 ab | 42.93 ab | 8.19 a | 5.33 a | 19.30 a | |
| Significance | ||||||||
| Light (L) | **y | NS | NS | NS | ** | ** | ** | |
| Cultivar (C) | NS | * | NS | NS | NS | NS | NS | |
| L c | * | * | NS | NS | * | NS | * | |
yNS indicates Non-significant; significant at P ≤ 0.05 and P ≤ 0.01, respectively.
* indicates Non-significant; significant at P ≤ 0.05 and P ≤ 0.01, respectively.
** indicates Non-significant; significant at P ≤ 0.05 and P ≤ 0.01, respectively.
Table 3.
Effect of red LEDs and darkness during healing and acclimatization process on fresh weight, dry weight, T/R ratio and compactness of grafted tomato seedlings.
| Treatments | Cultivars | Fresh weight (g) | Dry weight (g) | T/Rz ratio | Compactnessy (mg · cm–1) | ||
|---|---|---|---|---|---|---|---|
| Top | Root | Top | Root | ||||
| No light (Darkness) | ‘B-Blocking’ | 0.987 cx | 0.228 ab | 0.094 b | 0.016 b | 4.21 ab | 11.78 bc |
| ‘Kanbarune’ | 0.820 d | 0.148 b | 0.081 c | 0.011 c | 4.96 ab | 10.75 c | |
| ‘High-Power’ | 0.802 d | 0.177 b | 0.081 c | 0.012 c | 4.75 ab | 11.87 bc | |
| Red LEDs | ‘B-Blocking’ | 1.383 a | 0.274 a | 0.119 a | 0.021 a | 5.05 a | 13.69 a |
| ‘Kanbarune’ | 1.089 bc | 0.286 a | 0.123 a | 0.021 a | 4.13 b | 14.23 a | |
| ‘High-Power’ | 1.181 b | 0.244 a | 0.118 a | 0.020 a | 4.81 ab | 13.30 ab | |
| Significance | |||||||
| Light (L) | *w | * | ** | * | NS | * | |
| Cultivar (C) | * | NS | NS | NS | NS | NS | |
| L × c | * | * | * | NS | NS | * | |
y Compactness is the values of the dry weight divided by the plant height.
x Mean separation within columns by Duncan’s multiple range test at P = 0.05.
wNS indicates Non-significant; significant at P ≤ 0.05 and P ≤ 0.01, respectively.
* indicates Non-significant; significant at P ≤ 0.05 and P ≤ 0.01, respectively.
** indicates Non-significant; significant at P ≤ 0.05 and P ≤ 0.01, respectively.
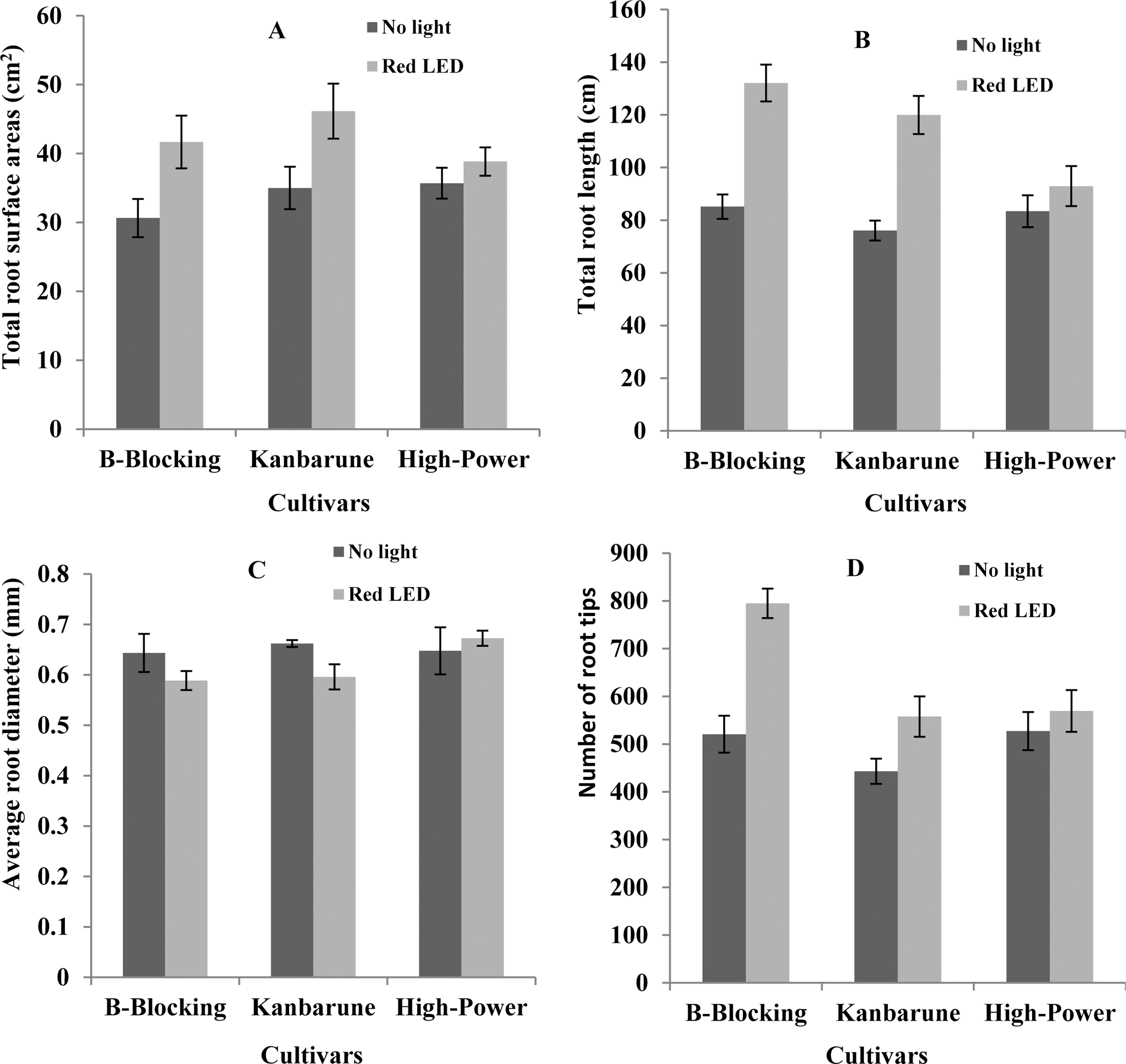
3. Effect of red LEDs on root morphology of grafted tomato seedlings
Root morphology of grafted seedlings responses to red LEDs and no light treatments are given in Fig. 1. Total root surface area, total root length, average root diameter, and number of root tips were significantly different in both light treatments and rootstock cultivars. Total root surface area, total root length, and number of toot tips of seedlings in red LEDs were higher than those of seedlings in no light treatment, except of ‘High-Power’ cultivar. However, average root diameter of ‘B-Blocking’ and ‘Kanbarune’ cultivars in red LEDs was lower than those in no light treatment.
Discussions
Light is one of the important factors that influence the survival rate of grafted seedling during healing and acclimatization process (Nobuoka et al., 2005). Therefore, in this study survival rates of grafted seedlings in red LEDs were higher than survival rates of grafted seedlings in no light treatment. This result agreed with results of Nobuoka et al. (2005) who suggested that the survival rate and growth of grafted tomatoes were enhanced by light during healing and acclimatization process. In addition, no significant difference was observed on survival rate in red LEDs treatment. Our result agreed with results of Oda et al. (1993) who did not find difference in survival rate between cucumber grafted onto C. maxima and C. moschata. Salehi et al. (2009) suggested that survival rate was not affected by the number of vascular bundles in hypocotyls of rootstocks. Vu et al. (2013) also reported that survival rate was not affected by different rootstock cultivars. On the other hand, light treatment also reduced percentage of infected plants, except ‘Kanbarune’ cultivar. This result agreed with the result of Buck et al. (2010) who reported that germination and germ tube elongation of fungi decreased with increasing light intensity. However, the survival rates of grafted seedlings in no light treatment were very low. Moreover, there was significant different among rootstock cultivars with survival rate in no light treatment. Therefore, darkness during healing and acclimatization process was not a suitable environment for grafted seedlings after grafting.
Some papers reported that the plant height of seedlings decreased with increasing light intensity during healing and acclimatization process (Jang et al., 2011; Nobuoka et al., 2005). However, in our study seedlings in red LEDs were higher than seedlings in no light treatment. A suitable environmental condition for grafted seedlings in red LEDs treatment was the cause that increased the chances for vascular bundles of the scion and rootstock to come into contact. Therefore, the growth recovery of grafted seedlings in red LEDs was faster than those in no light treatment. So, plant growth parameters in red LEDs were higher than those in no light treatment. Moreover, the grafted seedlings in red LEDs were significantly more compact than grafted seedlings in no light treatment. This result agreed with result of Jang et al. (2011) who reported an increase of photosynthetic photon flux during healing and acclimatization led to the improvement of growth and quality of grafted plants. In addition, the root morphology were also enhanced in seedlings treated with red LEDs through improving total root surface, total root length, and number of root tips. In relation to this, Li et al. (2010) and Moon et al. (2006) indicated that the root activity of seedlings was greatest under red LEDs light compared with no light conditions in tissue culture. However, the rapid development of root by increasing total root length and number of root tips in red LEDs of ‘B-Blocking’ and ‘Kanbarune’ cultivars could be the cause of reduced average root diameter in these cultivars.
In conclusion, short-term red LEDs irradiation is a suitable environmental condition during healing and acclimatization process for grafted seedlings. Red LEDs promoted the survival rate and quality of grafted tomato seedlings.